These Class 8 Science Curiosity Worksheet and Class 8 Science Chapter 7 Particulate Nature of Matter Worksheet with Answers are excellent for board exam practice.
Class 8 Science Chapter 7 Particulate Nature of Matter Worksheet
Class 8 Particulate Nature of Matter Worksheet
Worksheet On Particulate Nature of Matter Class 8 – Particulate Nature of Matter Worksheet Class 8
Let’s Begin
A. Multiple-Choice Questions
Question 1.
Who first proposed the idea of Parmonu (atom) in ancient India?
(a) Aryabhata
(b) Acharya Kanad
(c) Acharya Charaka
(d) Sushruta
Question 2.
Potassium permanganate spreads faster in hot water than in cold water because
(a) Water becomes heavier when heated
(b) Cold water repels particles .
(c) Heat increases particle movement
(d) Hot water changes into gas quickly.
Question 3.
In which state of matter are the particles farthest apart and move freely in all directions?
(a) Solid
(b) Liquid
(c) Gas
(d) None of these
Question 4.
This question consists of an Assertion (A) and a Reason (R). Read the Assertion and Reason and choose the appropriate answer.
Assertion (A): Gases fill the entire container they are kept in.
Reason (R): Gas particles move freely and have large spaces between them.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Question 5.
This question consists of an Assertion (A) and a Reason (R). Read the Assertion and Reason and choose the appropriate answer.
Assertion (A): Liquids have a fixed shape but no fixed volume.
Reason (R): The interparticle attraction in liquids is weaker than in solids.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
![]()
B. State True (T) or False (F).
1. Particles of matter are continuously moving.
2. Water is practically incompressible.
3. Matter is made up of tiny particles called atoms or molecules.
4. The arrangement of particles in solids is irregular and random.
5. Liquids have stronger forces of attraction than solids.
6. Rice grains and flour take the shape of the container.
C. Fill in the blanks.
1. The melting point of ice is ______ while that of urea is ______.
2. A substance that dissolves in water is called a ______ substance.
3. Sand is an example of an ______ substance in water.
4. Soap helps in cleaning because it can mix with both ______ and ______.
5. ______ a substance increases the movement of its particles.
6. The ocean water tastes salty because it contains ______ salts.
D. Assign one word to the following terms.
1. Ability of matter to reduce in volume under pressure. ______
2. Solid particles that remain suspended in air or water. ______
3. The process of solid turning to liquid on heating. ______
4. The process of liquid turning to gas on heating. ______
5. Basic unit that makes up a larger piece of a substance. ______
E. Match the Columns.
Question 1.
Match the statements given in Column I with the correct-terths given in Column II.
| Column I | Column II |
| (i) Space between particles of a substance | (a) Atoms and molecules |
| (ii) Change in state without forming a new substance | (b) Thermal energy |
| (iii) Force of attraction between the particles of a substance | (c) Interparticle space |
| (iv) Energy that increases particle motion | (d) Physical change |
| (v) Basic units of matter | (e) Interparticle attraction |
Question 2.
Unjumble the words given in Column II and write them in the space provided with the help of the images given in Column I. Then match the columns.

Wonder Why?
A. Read the paragraph and answer the following questions.
SPM consists of tiny solid or liquid particles present in the polluted air, like dust, smoke and pollen. These particles vary in size, with some being as small others being much larger. The size of the particles can affect their ability to be inhaled into the lungs and their potential to cause health problems.
Question 1.
What does SPM stand for?
____________________________________
____________________________________
Question 2.
Are SPM particles dissolved in air?
____________________________________
____________________________________
Question 3.
Why are SPM particles harmful?
____________________________________
____________________________________
Question 4.
How are SPM particles different from the constituent particles of matter?
____________________________________
____________________________________
![]()
B. Observe the figures (a) and (b) and answer the following questions.
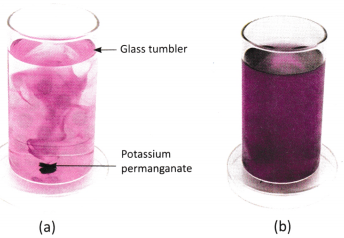
Question 1.
What do you observe initially after adding potassium permanganate to water in figure (a)?
____________________________________
____________________________________
Question 2.
What happens to the colour of water after some time, as shown in figure (b)?
____________________________________
____________________________________
Question 3.
What does this tell us about the movement of particles in liquids?
____________________________________
____________________________________
C. Observe the four pictures representing the stages of grinding chalk-Provide suitable captions for the pictures (a-d) and answer the questions.
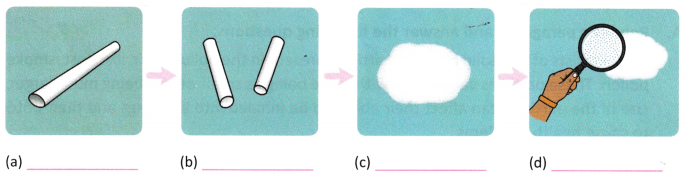
Question 1.
Which stage shows the chalk in its smallest visible form?
____________________________________
____________________________________
Question 2.
Does the chalk change into a new substance after grinding?
____________________________________
____________________________________
Question 3.
What type of change is grinding chalk — physical or chemical?
____________________________________
____________________________________
Question 4.
What does this activity tell us about the nature of matter?
____________________________________
____________________________________
D. Observe the pictures carefully. Provide suitable captions for (a) and (c), and fill in the blanks with the correct words. Observe/Identify

(a) ______ have particles that are ______ (tightly/loosely) packe(d) They have a ______ (definite/indefinite) shape and volume because interparticle ______ (attractions/repulsions) are very strong. These particles cannot move freely but can only ______ (vibrate/flow). When we apply ______ (heat/cold), the particles vibrate more and the (a) ______ starts to ______ (melt/freeze). The temperature at which (a) ______ changes to (c) ______ is called the ______ (melting point/boiling point) of the (a) ______.
Learn By Doing
A. Take three clean and dry containers of different shapes. Label them as A, B, and C. Mark the 350 mL level in Container A using a measuring cylinder and a marker or tape.
Now, fill Container A with water up to the 350 mL mark. Then, carefully pour the water from Container A into Container B. Finally, transfer the water from Container B into Container C. Based on your observations, answer the following questions.
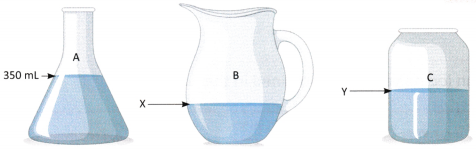
Question 1.
What volume of water does the mark X in Container B indicate?
____________________________________
____________________________________
Question 2.
What happens to the shape and volume of water when it is transferred from Container A to Container B?
____________________________________
____________________________________
Question 3.
What volume of water does the mark Y in Container C indicate?
____________________________________
____________________________________
Question 4.
What happens to the shape and volume of water when it is transferred from Container B to Container C?
____________________________________
____________________________________
Question 5.
What can you conclude about liquids when they are poured into different containers?
____________________________________
____________________________________
B. Take two transparent gas jars, A and B. Take some solid iodine in Gas Jar A and cover its open end with a glass plate for some time to collect iodine vapours. Invert Jar B over the plate, align both jars, and carefully slide out the plate so their openings touch without gaps. Based on your observations, answer the following questions.
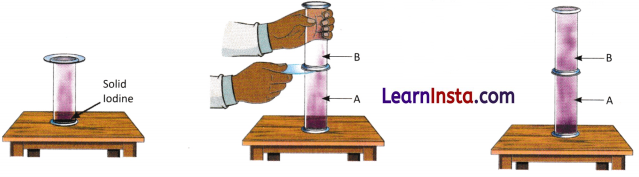
Question 1.
Which property of gases does this activity demonstrate?
____________________________________
____________________________________
Question 2.
How can iodine vapour be obtained in the Gas Jar A?
____________________________________
____________________________________
Question 3.
Why should we be careful while using solid iodine?
____________________________________
____________________________________
![]()
Explore With Curiosity
A. Ankit is playing with a syringe (without a needle) in the science lab. He pulls the plunger outwards in a fully extended position. When he covers the open end of the syringe with his thumb and pushes the plunger in, he feels resistance but can still compress the air. Curious, he repeats the same process with water instead of air. This time, he finds it very difficult to push the plunger in. Why is it easier for Ankit to compress air but not water using the same syringe?
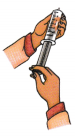
____________________________________
____________________________________
____________________________________
B. In a science experiment, Zara adds one spoon of sugar to one glass of water and one spoon of sand to another. She marks the initial water levels in both glasses.

After stirring:
- The sugar dissolves, and the water level changes only slightly.
- The sand settles, and the water level stays higher.
Why do the two solids behave differently in water? What does this reveal about the spacing between particles in a liquid?
____________________________________
____________________________________
____________________________________
C. At a science museum, Aarav saw three boxes with balls arranged differently. These balls represent the arrangement of particles in solids, liquids, and gases. Later, he is asked to identify different materials and categorise them into three particle models labelled (i), (ii), and (iii).
Help Aarav write the correct number (i, ii, or iii) for the particle arrangement for each material.
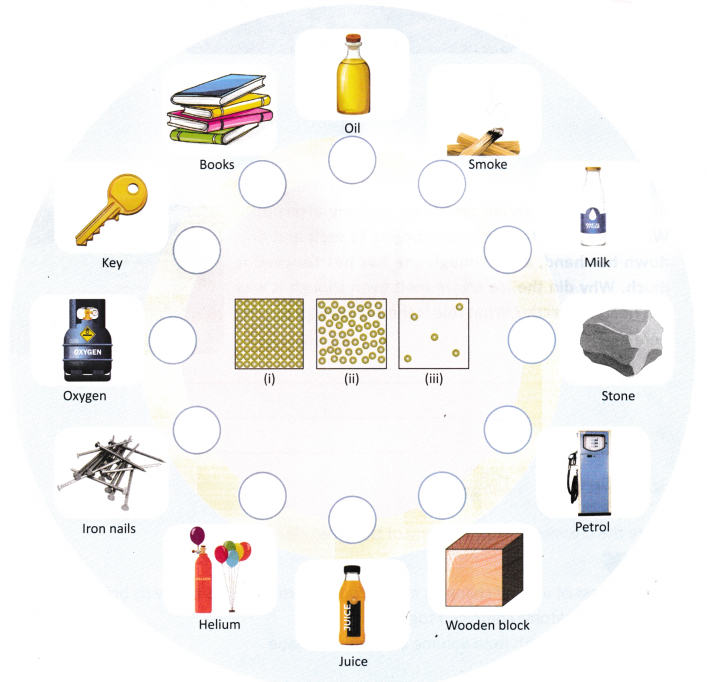
D. Anaya is reading in her bedroom when she suddenly smells something delicious. She walks into the kitchen and sees her mother preparing biryani. The 4dteh6h cloor was closed, yet she could smell the spices from another room. How did the aroma of biryani reach Anaya’s room even though there was no fan or breeze? What does this tell us about the movement of gas particles?

____________________________________
____________________________________
____________________________________
E. Riya is enjoying an ice cream on a sunny afternoon. Within minutes, the ice cream begins to melt and drip down her hand, even though she has not touched it much. Why did the ice cream melt even though it was not heated directly? What role is thermal energy playing here?

____________________________________
____________________________________
____________________________________
Word Puzzle
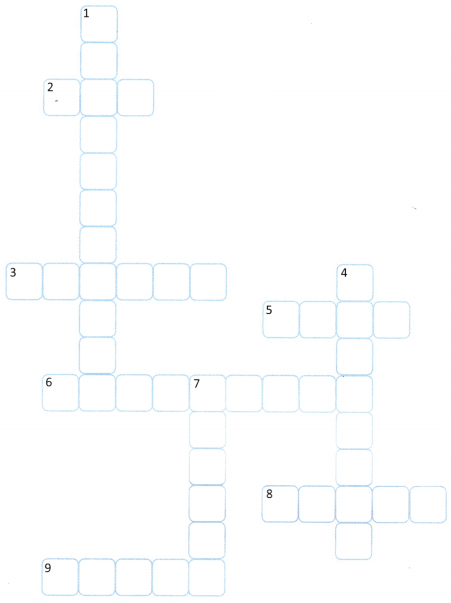
Complete the crossword with the help of the given clues.
DOWN
1. The process of a liquid changing into vapour at temperatures below its boiling point
4. Two or more atoms combined together
7. State of matter with fixed volume but no fixed shape
ACROSS
2. State of matter with no fixed shape or volume .
3. Anything that has mass and occupies space
5. The smallest unit of matter that retains the properties of an element
6. A substance that does not dissolve in a solvent
8. State of matter with fixed shape and volume, tightly 3 packed particles
9. Substances that can flow (liquids and gases)
![]()
Suggested Activities
A. Place ice cubes in a beaker and gently heat them over a flame. Observe the changes that take place as the ice melts into water and then turns into steam. Record the temperature at which each change occurs. Discuss how melting point and boiling point are physical properties of substances.
B. Take two greasy plates—wash one with only water and the other with soap and water. Observe which one gets cleaner and discuss why. Explore how soap makes insoluble substances like oil mix with water, aiding in cleaning.
C. Use clay balls, beads, or thermocol balls to model the arrangement of particles in solids, liquids, and gases. Arrange them on cardboard or place them in jars to represent the spacing and arrangement of particles in each state of matter. Label your models to show interparticle spaces and forces of attraction.