Experts have designed these Class 8 Science Notes Chapter 7 Particulate Nature of Matter Class 8 Notes for effective learning.
Class 8 Science Chapter 7 Particulate Nature of Matter Notes
Class 8 Particulate Nature of Matter Notes
Class 8 Science Chapter 7 Notes – Particulate Nature of Matter Notes Class 8
→ All matter is made up of a large number of extremely small particles. These particles are so small that they cannot be seen even through an ordinary microscope.
![]()
→ A constituent particle is the basic unit that makes up a larger piece of a substance or material.
For example,
- The grains of sand and clay are made up of a large number of their constituent particles.
- When sugar dissolves in water, it breaks up into its constituent particles which cannot be broken down further. Each grain of sugar is made up of millions of such constituent particles.
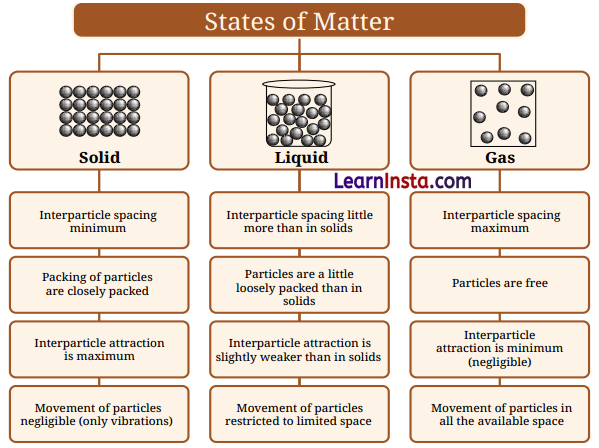
→ Particles of matter have spaces between them called interparticle spaces. These spaces are smallest in solids, larger in liquids, and largest in gases.
→ This is why the substances like sugar dissolve in water – sugar particles fill the spaces between water particles.
→ The constituent particles of matter are held together by interparticle forces of attractions. The strength of these attractions depends on the nature of the substance and the interparticle distance. Even a slight increase in the distance decreases the interparticle forces drastically. The strength of these forces decides the physical state of the substances.
- The interparticle attractions are the strongest in solids, a little weaker in liquids, and the weakest in gases.
→ Acharya Kanad, an ancient Indian philosopher, first spoke about the idea of a Parmanu (atom). He believed that all matter is made up of tiny, indivisible eternal particles called Parmanu. This idea was written in his work called Vaisheshika Sutras.
![]()
→ Arrangement of Particles in Solids:
- Solids have a definite shape and volume because their particles are tightly packed. The strong interparticle forces of attraction in solids keep the particles in fixed positions. These particles can only vibrate in place and cannot move freely past each other.
- When a solid is heated, its particles start to vibrate faster. At a certain temperature, these vibrations become so strong that the particles start leaving their position. The interparticle forces of attraction get weakened and the solid gets converted into the liquid state.
- The minimum temperature at which a solid melts to become a liquid at the atmospheric pressure is called its melting point.
- Generally, particles in a solid state are closer together as compared to those in the liquid state. (Exception: In ice, particles are farther apart than in liquid water.)
- Solids with weaker interparticle forces have low melting points, while those with stronger forces have high melting points.
→ Melting Points of Some Solids:
| S. No. | Material | Melting point |
| 1. | Ice | 0 °C |
| 2. | Urea | 133 °C |
| 3. | Iron | 1538 °C |
→ Arrangement of Particles in Liquids:
- Liquids have a definite volume but no fixed shape. They take the shape of the container they are kept in. The particles in a liquid are not fixed and can move freely, but only within a limited space.
- The interparticle attractions in liquids are weaker than in solids, but still strong enough to keep the particles close together.
- When a liquid is heated, its particles gain energy and move faster. At a certain temperature, the liquid starts to boil. The movement of particles becomes so vigorous that they move apart from each other, resulting in a decrease in the interparticle forces of attraction. Eventually, the constituent particles escape from the liquid and change into vapour or gas.
→ When sugar dissolves in water, the final volume of sugar solution is less than the expected sum of the volumes of water and sugar taken. This is because sugar particles occupy the empty spaces between water particles.
![]()
→ The word ‘particle’ can have different meanings depending on the context. For example, in air pollution, SPM (Suspended Particulate Matter) refers to the tiny dust particles suspended in the air. These are not the same as the extremely small constituent particles that make up all matter, like atoms and molecules. Even dust particles are also made up of many atoms and molecules.
→ Water particles are always moving. When potassium permanganate is added, its particles spread through the water as moving water particles push them around. This turns the water evenly pink without stirring.
→ Water particles move faster in hot water compared to water at room temperature, and even slower in ice-cold water. Hence, the movement of particles increases when heat is provided.
→ The particles of air are in constant motion. For example, when an incense stick is burnt in one corner of the room, the fragrance spreads throughout the room.
![]()
→ The particulate nature of matter affect many daily activities. For example, when we wash oily stains in clothes with soap, it helps to remove the stains. Tiny soap particles surround the oil particles on the fabric. One end of each soap particle sticks to the oil, while the other end mixes with water. This allows the soap to lift the oil off the fabric and wash it away.
→ Particles of Matter Depends on Thermal Energy:
- Matter is made up of tiny particles held together by forces of attraction. The strength of these forces depends on the distance between particles, which is affected by their thermal (heat) energy.
- In solids, particles have low thermal energy. They stay close together and can only vibrate in place because the attractive forces are strong.
- At the melting point, heat energy helps particles overcome these forces, turning the solid into a liquid. In liquids, particles are a bit farther apart and can move around, but still stay close.
- In gases, particles have high thermal energy. They move freely in all directions because the forces of attraction are very weak.
![]()
→ The tiny particles that make up all matter are called atoms and molecules. For example, gold is made of gold atoms. Some elements, cannot exist as single atoms. Instead, their atoms join together to form molecules. For example, two hydrogen atoms join to form a hydrogen molecule. A water molecule has two hydrogen atoms and one oxygen atom.
→ Summary of Particle Arrangement in Different States of Matter