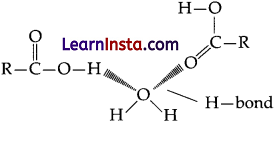
Students can access the CBSE Sample Papers for Class 12 Chemistry with Solutions and marking scheme Set 1 will help students in understanding the difficulty level of the exam.
CBSE Sample Papers for Class 12 Chemistry Set 1 with Solutions
Time Allowed: 3 hours
Maximum Marks: 70
General Instructions :
1. There are 33 questions in this question paper with internal choice
2. SECTION A consists of 16 multiple-choice questions carrying 1 mark each.
3. SECTION A consists of 16 multiple-choice questions carrying 1 mark each.
4. SECTION C consists of 7 short answer questions carrying 3 marks each.
5. SECTION D consists of 2 case-based questions carrying 4 marks each.
6. SECTION E consists of 3 long answer questions carrying 5 marks each.
7. All questions are compulsory.
8. Use of log tables and calculators is not allowed.
SECTION – A (16 Marks)
The following questions are multiple-choice questions with one correct answer. Each question carries 1 mark. There is no internal choice in this section.
Question 1.
Which of the following solutions will have the highest conductivity at 298 K?
(a) 0.01 M HCl solution
(b) 0.1 M HCl solution
(c) 0.01 M CH3COOH solution
(d) 0.1 M CH3COOH solution
Answer:
(b) 0.1 M HCl solution
Explanation: HCl is very strong acid when mixed in water its ions completely dissociates in water to form H+ and Cl–. Conductivity is higher for strong electrolyte and it decreases with dilution.
Question 2.
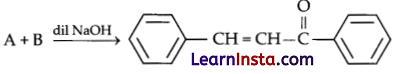
Identify A and B.
(a) A = 1-phenylethanal, B = acetophenone
(b) A = Benzophenone, B = formaldehyde
(c) A = Benzaldehyde, B = Acetophenone
(d) A = Benzophenone, B = Acetophenone
Answer:
(c) A = Benzaldehyde, B = Acetophenone
Explanation:
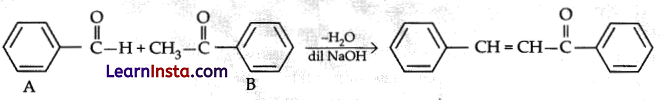
This reaction is an example of cross aldol condensation.
Question 3.
The vitamins which can be stored in our body are:
(a) Vitamin A, B, D and E
(b) Vitamin A, C,D and K
(c) Vitamin A, B, C and D
(d) Vitamin A, D, E and K
Answer:
(d) Vitamin A, D, E and K
Explanation: Vitamin A, D, E and K are fat soluble vitamins and they are stored in liver and adipose (fat storing) tissues.
![]()
Question 4.
What is IUPAC name of the ketone A, which undergoes iodo form reaction to give CH3CH = C(CH3)
COONa and yellow precipitate of CHI3?
(a) 3-Methylpent-3-en-2-one
(b) 3-Methylbut-2-en-one
(c) 2, 3-Dimethylethanone
(d) 3-Methylpent-4-one
Answer:
(a) 3-Methylpent-3-en-2-one
Explanation: Chemical reaction is:
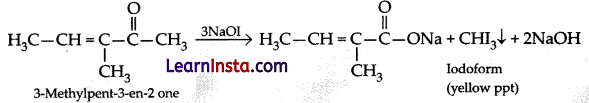
Methyl ketones are oxidised by sodium hypohaliate to sodium salts of carboxylic acid and the methyl group is converted into haloform. This oxidation does not affect carbon-carbon double bond.
Question 5.
Which of the following is not correct?
(a) In haloarenes, the electron pairs on halogen atom are in conjugation with of the ring.
(b) The carbon-magnesium bond is covalent and non-polar in nature .
(c) During SN1 reaction, the carbocation formed in the slow step being sp2 hybridised is planar.
(d) Out of CH2 = CH—Cl and C6H5CH2Cl, C6H5CH2Cl is more reactive towards SN1 reaction
Answer:
(b) The carbon-magnesium bond is covalent and non-polar in nature.
Question 6.
Match the properties with the elements of 3d series:
| (i) lowest enthalpy of atomisation | (P) Sc |
| (ii) shows maximum number of oxidation states | (q) Mn |
| (iii) transition metal that does not form coloured compounds | (r) Zn |
| (s)Ti |
(a) (i) (r), (ii) (q), (iii) (p)
(b) (i) (r), (ii) (s), (iii) (p)
(c) (i) (p), (h) (q), (hi) (r)
(d) (i) (s), (ii) (r), (iii) (p)
Answer:
(a) (i) (r), (ii) (q), (iii) (p)
Explanation: Zn has no unpaired electrons in 3d or 4s orbitals, so enthalpy of atomisation is low.
Mn = 3d5 4s2 , shows +2, +3, +4, +5, +6 and +7 oxidation state, maximum number of oxidation states in 3d series.
Sc has common oxidation state +3 it has no electron in 3d subshell (3d0). Hence, it does not form coloured compounds.
Question 7.
Which of the following statement is true?
(a) molecularity of reaction can be zero or a fraction.
(b) molecularity has no meaning for complex reactions.
(c) molecularity of a reaction is an experimental quantity.
(d) reactions with the molecularity three are very rare but are fast.
Answer:
(b) molecularity has no meaning for complex reactions.
Explanation: The molecularity ofa reaction is defined as the number of reacting molecules thatcollide simultaneously to bring about a chemical reaction. The molecularity of a reaction is a theoretical concept. The molecularity of a reaction can neither be zero nor fractional.
Question 8.
In which of the following solvents, the C4H8NH3+X– is soluble:
(a) ether
(b) acetone
(c) water
(d) bromine water
Answer:
(c) water
Explanation: Due to the presence of the nitrogen atom, which can form hydrogen bonds with water molecules amines like the C4H8NH3+ have a polar nature and soluble in water.
Question 9.
Which of the following observation is shown by 2-phenyl ethanol with Lucas reagent?
(a) Turbidity will be observed within five minutes
(b) No turbidity will be observed
(c) Turbidity will be observed immediately
(d) Turbidity will be observed at room temperature but will disappear after five minutes.
Answer:
(b) No turbidity will be observed
Explanation: The chemical method used for distinguishing between primary, secondary and tertiary alcohol is known as the Lucas test. When the Lucas reagent is added to primary alcohol, no turbidity is observed. In addition to the Lucas reagent in the solution containing secondary alcohol, an oily layer is formed in three to five minutes and turbidity is observed in the solution. Tertiary alcohols on reaction with the Lucas reagent quickly form an oily layer and turbidity is also observed immediately in the solution.
Since 2-phenyl ethanol is a primary alcohol, therefore, no turbidity will observed with Lucas reagent.
![]()
Question 10.
If the initial concentration of substance A is 1.5 M and after 120 seconds the concentration of substance A is 0.75 M, the rate constant for the reaction if it follows zero-order kinetics is:
(a) 0.00625 mol L-1s-1
(b) 0.00625 s-1
(c) 0.00578 mol L-1s-1
(d) 0.00578 s-1
Answer:
(a) 0.00625 mol L-1s-1
Explanation: For zero order react k = \(\frac{\left[\mathrm{R}_0\right]-[\mathrm{R}]}{t}=\frac{1.5-0.75}{120}\)
= \(\frac{0.75}{120}=0.00625 \mathrm{~mol} \mathrm{~L}^{-1} \mathrm{~s}^{-1}\)
Question 11.
Anisole undergoes bromination with bromine in ethanoic acid even in the absence of iron(III) bromide catalyst:
(a) Due to the activation of benzene ring by the methoxy group.
(b) Due to the deactivation of benzene ring by the methoxy group.
(c) Due to the increase in electron density at ortho- and para-positions
(d) Due to the formation of stable carbocation.
Answer:
(a) Due to the activation of benzene ring by the methoxy group.
Explanation: Anisole undergo bromination with bromine in ethanoic acid (acetic acid) even in the absence of iron(HI) bromide catalyst due to activation of benzene ring by the methoxy group, to from mixture of ortho and para-bromo anisole.
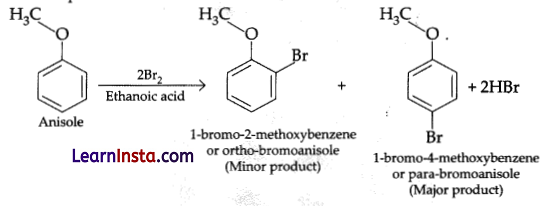
Question 12.
The trend of which property is represented by the following graph?
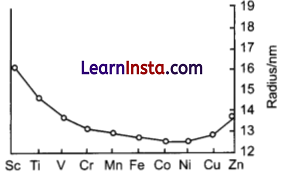
(a) ionization enthalpy
(b) atomic radii
(c) enthalpy of atomization
(d) melting point
Answer:
(b) atomic radii
Explanation: The atomic radips increases down the group. The atomic radii of second (4d) series is larger than those of the first transition series. But the atomic radii of the second (4d) and third (5d) transition series are almost the same due to lanthanoid contraction.
Question 13.
Given below are two statements labelled as Assertion (A) and Reason (R):
Assertion (A): Alcohols react both as nucleophiles and electrophiles.
Reason (R): The bond between C-O is broken when alcohols react as nucleophiles. Select the most appropriate answer from the options given below:
(a) Both A and R are true and R is the correct explanation of A
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Answer:
(c) A is true but R is false.
Explanation:
Alcohols as nucleophile: The bond between O-H is broken when alcohol react as nucleophiles.
Alcohols as electrophile: The bond between C-O is broken when alcohol reacts as electrophiles.
Question 14.
Given below are two statements labelled as Assertion (A) and Reason (R):
Assertion (A): Strong oxidising agents oxidise toluene and its derivatives to benzoic acids.
Reason (R): It is possible to stop the oxidation of toluene at the aldehyde stage with suitable reagents.
Select the most appropriate answer from the options given below:
(a) Both A and R are true and R is the correct explanation of A
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Answer:
(b) Both A and R are true but R is not the correct explanation of A.
Explanation: By oxidation of methyl bezene strong oxidising agents oxidise toluene and its derivatives to benzoic acids. However, it is possible to stop the oxidation at the aldehyde stage with suitable reagents that convert the methyl group to an intermediate that is difficult to oxidise further.
![]()
Question 15.
Given below are two statements labelled as Assertion (A) and Reason (R):
Assertion: Enzymes are very specific for a particular reaction and for a particular substrate.
Reason: Enzymes are biocatalysts.
Select the most appropriate answer from the options given below:
(a) Both A and R are true and R is the correct explanation of A
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Answer:
(b) Both A and R are true but R is not the correct explanation of A.
Explanation: Enzymes are biocatalysts and are very specific for a particular reaction and for a particular substrate. They are generally named after the compound or class of compounds upon which they work for example, the enzyme that catalyses hydrolysis of maltose into glucose is named as maltase.
Question 16.
Given below are two statements labelled as Assertion (A) and Reason (R):
Assertion (A): During electrolysis of aqueous copper sulphate solution using copper electrodes hydrogen gas is released at the cathode.
Reason (R): The electrode potential of Cu2+/Cu is greater than that of H+/ H2
Select the most appropriate answer from the options given below:
(a) Both A and R are true and R is the correct explanation of A
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Answer:
(d) A is false but R is true.
Section – B (10 Marks)
This section contains 5 questions with internal choice in one question. The following questions are very short answer type and carry 2 marks each.
Question 17.
(a) Radioactive decay follows first-order kinetics. The initial amount of two radioactive elements X and Y is 1 gm each. What will be the ratio of X and Y after two days if their half-lives are 12 hours and 16 hours respectively?
(b) The hypothetical reaction P + Q → R is half order w.r.t. ‘P’ and zero order w.r.t. ‘Q’. What is the unit of rate constant for this reaction?
Answer:
(a) The rate constant k for a first order reaction is given by k = \(\frac{0.693}{t_{1 / 2}}\)
Where t1/2 = half life of element
For X t1/2 = 12 hours
∴ Kx = \(\frac{0.693}{12}=0.5775 h^{-1}\)
Similarly,
For Y t1/2 = 16 hours
So, Ky = \(\frac{0.693}{16}=0.04331 h^{-1}\)
Given
Initial amount of X and Y = 1 gm
For X 2 days = 48 hours means 4 half lives therefore amount of X lift = 1/16 of initial value
For Y half life of Y = 16 hours
2 days = 48 hours means 3 half given amount of Y left = 1/8 of initial value
Ratio of X : Y = \(\frac{1}{16}: \frac{1}{8}\) = 1 : 2
(b) Rate = k[P]1/2 [Q]0, here k = Rate constant
So, Unit of k = \(\begin{aligned}
& \frac{\text { Unit of Rate }}{\text { Unit of }[\mathrm{P}]^{1 / 2}}=\frac{\text { Mole } / \mathrm{L} / \mathrm{sec}}{[\text { Mole }]^{1 / 2}[\mathrm{~L}]^{1 / 2}} \\
& =\text { Mole }^{1 / 2} \mathrm{~L}^{-1 / 2} \mathrm{sec}^{-1} .
\end{aligned}\)
Question 18.
A 5% solution of Na2SO4.10H2O (MW = 322) is isotonic with 2% solution of non- electrolytic, non-volatile substance X. Find out the molecular weight of X.
Answer:
Both given solutions are Isotonic
∴ Osmotic pressure of both solutions are equal.
Or, π1 = π2
Since, the solution of Na2SO4.10H2O is electrolytic, it dissociates into ions and will have higher osmotic pressure than the non-electrolytic solutions X.
Therefore, we need to find the molecular weight of X that will give an osmotic pressure equal to the solution of Na2SO4.10H2O.
Given: % weight of Na2SO4.10H2O = 5
% weight of X = 2
π = iCRT
Where π = Osmotic pressure
C = Concentration of solution
R = Gas constant
T = Temperature is Kelvin
i = van’t Hoff factor
Since, X is a non-electrolytic substance its van’t Hoff factor =1.
We can write the following equation: iC1RT = C2RT
\(\begin{aligned}
\frac{3 \times 5}{322} & =\frac{2}{M} \\
M & =\frac{2 \times 322}{3 \times 5}
\end{aligned}\)
M = 42. 9 gm
![]()
Question 19.
(a) Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
(b) Explain why the electrophilic substitution reactions in haloarenes occur slowly and require more drastic conditions as compared to those in benzene.
Answer:
(a) m-dichlorobenzene < o-dichlorobenzene < p-dichlorobenzene.
Para-isomers are high boiling and melting point as compared to their ortho- and meta-isomers. It is
due to symmetry of para-isomers that fits in crystal lattice better as compared to ortho- and meta-isomers.
(b) The resonating structures of haloarenes as shown:
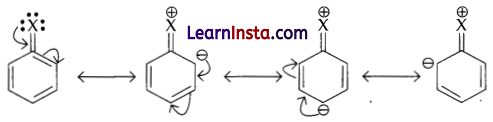
Due to resonance, the electron density increases more at ortho- and para-positions that at meta¬ positions. Further, the halogen atom because of its. I effect has some tendency to withdraw electrons from the benzene ring. As a result, the ring gets somewhat deactivated as compared to benzene and hence the electrophilic substitution reaction in holoarenes occur slowly and require more drastic condition as compared to those in benzene.
Question 20.
(a) Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
(b) Write the structure of the product formed when acetone reacts with 2, 4 DNP reagent.
Answer:
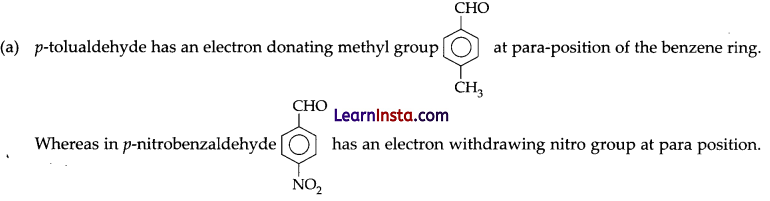
Thus, p-tolualdehyde is less reactive and p-nitrobenzaldehyde is more reactive towards nucleophilic addition reactions.
(b) When acetone react with 2,4-DNP reagent it gives 2,4-DNP, derivative.
The chemical reaction is given as below:
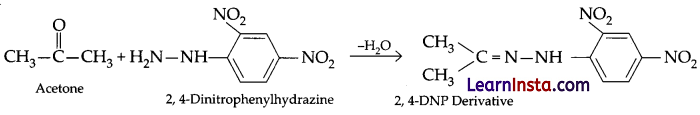
OR
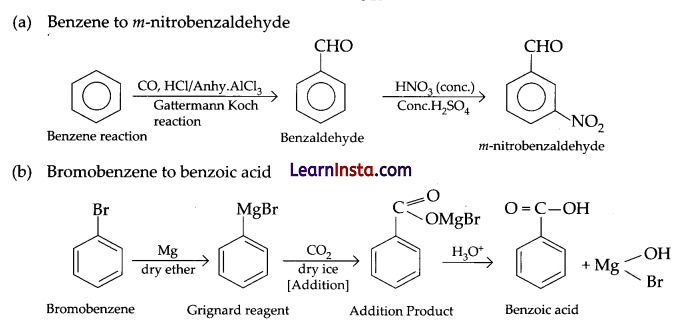
Convert the following:
(a) Benzene to m-nitrobenzaldehyde
(b) Bromobenzene to benzoic acid
Answer:
Question 21.
(a) DNA fingerprinting is used to determine paternity of an individual. Which property of DNA helps in the procedure?
(b) What structural change will occur when a native protein is subjected to change in pH?
Answer:
(a) A sequence of bases on DNA is unique for a person and information regarding this DNA is called DNA fingerprinting. It is same for every cell and cannot be altered by any known treatment. This unique property helps to determine paternity of an individual.
(b) When a native protein is subjected to change in pH, the hydrogen bond (present in structure) are disturbed. Due to this globules unfold and helix get uncoiled and protein loses its biological activity. This is called denaturation of protein. During denaturation secondary and tertiary structures are destroyed but primary structure remains intact.
Section – C (21 Marks)
This section contains 7 questions with internal choice in one question. The following questions are short answer type and carry 3 marks each.
Question 22.
(a) Write the formula for the following coordination compound Bis(ethane-1, 2-diamine) dihydroxidochromium(III) chloride.
(b) Does ionization isomer for the following compound exist? Justify your answer. Hg[Co(SCN)4].
(c) Is the central metal atom in coordination complexes a Lewis acid or a Lewis base? Explain.
Answer:
(a) [Cr(en)2(OH)2]Cl or [Cr(H2NCH2CH2NH2)2(OH)2]Cl
Bis (ethane-1,2-diamine) dihydroxidochromium(III) chloride
(b) No, Ionization isomer for the Hg [Co(SCN)4] does not exist because ionisation isomerism arises when counter ion in a complex salt is itself a potential ligand and can displace a ligand which can then compound a counter ion but in above compound no exchange is possible.
(c) In a co-ordination complex, central metal atom is electron pair acceptor so it is a lewis acid.
Example: In [NiCl2(CH2O)4] and [COCl(NH3)5]2+ Ni2+ and CO3+ are central atom/ion and these are also referred to as Lewis acids.
![]()
Question 23.
(a) Can we construct an electrochemical cell with two half-cells composed of ZnSO4 solution and zinc electrodes? Explain your answer.
(b) Calculate the ∧om for Cl– ion from the data given below:
∧om MgCl2 = 258.6 S cm2 mol-1 and ∧om Mg2+ = 106 S cm2 mol-1
(c) The cell constant of a conductivity cell is 0.146 cm-1. What is the conductivity of 0.01 M solution of an electrolyte at 298 K, if the resistance of the cell is 1000 ohm?
Answer:
(a) Yes, we can construct an electrochemical cell with two half cells composed of ZnSO4 solution and zinc electrodes this is possible if the concentration of ZnSO4 in the two half cell is different, the electrode potential will be different making the cell possible.
(b) We know from Kohlrausch law that: ∧om (MgCl2) = ∧om (Mg2+) + 2 ∧om (Cl–) …(i)
Given in question
∧om (MgCl2) = 258.65 S cm2 mol-1
∧om = (Mg2+) = 106 S cm2 mol-1
Put these values in equation (i),
258.6 = 106 + 2 ∧om (Cl–)
2 ∧om (Cl–) = 258.6 – 106
∧om (Cl–) = \( \frac{152.6}{2}\) = 76.3 S cm2 mol-1
(c) Given, Cell constant (G*) = 0.146 cm-1
Molarity of solution (M) = 0.01 m
Resistance (R) = 1000Ω
According to formula
G*=k × R
k = \(\frac{\mathrm{G}^*}{\mathrm{R}}=\frac{0.146}{1000}=1.46 \times 10^{-4} \mathrm{~cm}^{-1}\)
Question 24.
Write the name of the reaction, structure and IUPAC name of the product formed when (any 2):
(a) phenol reacts with CHCl3 in the presence of NaOH followed by hydrolysis.
(b) CH3CH2CH(CH3)CH(CH3)ONa reacts with C2H5Br
(c) CH3CH2CN reacts with stannous chloride in the presence of hydrochloric acid followed by hydrolysis
Answer:
(a) Reimer-Tiemann reaction: On treating phenol with chloroform in the presence of sodium hydroxide a – CHO group is introduced at ortho-position of benzene ring. This reaction is known as Reimer-Tiemann reaction.
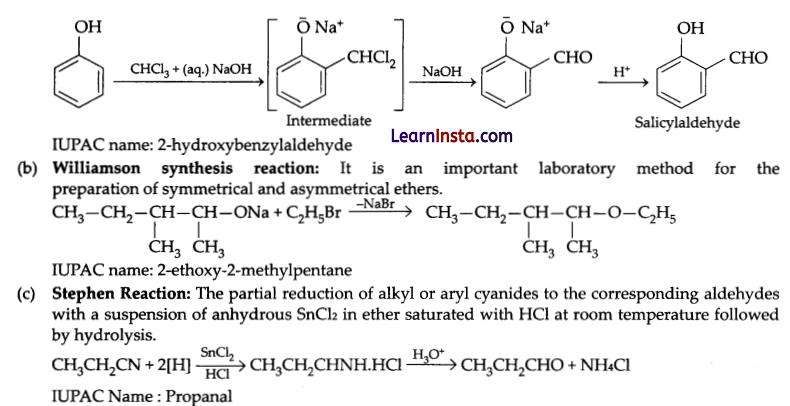
![]()
Question 25.
You are given four organic compounds “A”, “B”, “C” and “D”. The compounds “A”, “B” and “C” form an orange-red precipitate with 2, 4 DNP reagent. Compounds “A” and “B” reduce Tollen’s reagent while compounds “C” and “D” do not. Both “B” and “C” give a yellow precipitate when heated with iodine in the presence of NaOH. Compound “D” gives brisk effervescence with sodium bicarbonate solution. Identify “A”, “B”, “C” and “D” given the number of carbon atoms in three of these carbon compounds is three while one has two carbon atoms. Give an explanation for your answer.
Answer:
Compound A, B and C forms 2, 4-DNP derivatives. Therefore, they are either aldehyde or ketone. Compound A and B reduce Tollen’s reagent therefore, they are aldehyde. Compound C and D do not reduce Tollen’s reagent so they must ketone. Compound B and C give iodoform test therefore, they should be an aldehyde or methyl ketone. Compound C is methyl ketone. Compound D give decarboxylation reaction, so it must be a carboxylic acid.
Chemical equations are:
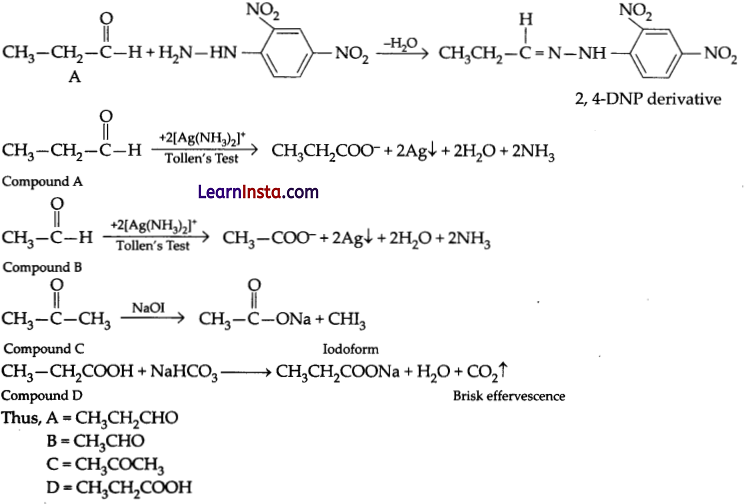
Question 26.
When sucrose is hydrolysed the optical rotation values are measured using a polarimeter and are given in the following table:
| S. No | Time(hours) | Specific Rotation |
| 1 | 0 | +66.5 |
| 2 | ∞ | -39.9° |
(a) Account for the two specific rotation values.
(b) What is the specific name given to sucrose based on the above observation .
(c) One of the products formed during the hydrolysis of sucrose is a glucose, that reacts with hydroxylamine to give compound A. Identify compound A.
Answer:
(a) The reactant sucrose is dextrorotatory but after hydrolysis it gives dextrorotatory glucose and laevoroatatory fructose. Since the laevorotation of fructose (-92.4°) is more than dextrorotation of glucose (+52.5°), the mixture is laevorotatory. Thus hydrolysis of sucrose brings about a change in the sign of rotation, from dextro(+) to laevo(-).
(b) The product name is invert sugar. The hydrolysis of sucrose is brings about a change in the sign of rotation, so product named as invert sugar.
(c) The name of compound is glucose oxime. The reaction is:
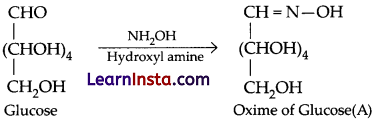
Question 27.
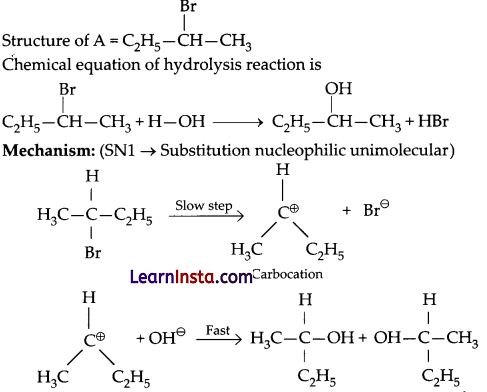
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (±) C4H9OH. Give the structure of A and write the mechanism of the reaction.
Answer:
Question 28.
The rate constants of a reaction at 200K and 500K are 0.02 s-1 and 0.20 s-1 respectively. Calculate the value of En. (Given 2.303R = 19.15 JK-1mol-1)
Answer:
From Arrhenius equation
We know that
\(\log \left(\frac{\mathrm{K}_2}{\mathrm{~K}_1}\right)=\frac{\mathrm{E}_a}{2.303 \mathrm{R}}\left[\frac{\mathrm{T}_2-\mathrm{T}_1}{\mathrm{~T}_1 \mathrm{~T}_2}\right]\) …(i)
Given: K1 = 0.02s-1
K2 = 0.20s-1
T1 = 200K
T2 = 500K
2.303R = 19.15 JK-1 mol-1
Put these values in equation (i)

Thus, the activation energy (Ea) for the reaction will be 6383J mol-1.
Section – D (8 Marks)
The following question are case-based questions. Each question has an internal choice and carries 4(1 + 1 + 2) marks each. Read the passage carefully and answer the questions that follow:
Question 29.
Crystal field splitting by various ligands
Metal complexes show different colours due to d-d transitions. The complex absorbs light of specific wavelength to promote the electron from t2g to eg’ level. The colour of the complex is due to the transmitted light, which is complementary of the colour absorbed. The wave number of light absorbed by different complexes of Cr ion are given below:
| Complex | Wave-number of light absorbed (cm-1) | Energy of light absorbed (kJ/mol) |
| [CrA6]3- | 13640 | 163 |
| [CrB6]3+ | 17830 | 213 |
| [CrC6]3+ | 21680 | 259 |
| [CrD6]3- | 26280 | 314 |
Answer the following questions:
(a) Out of the ligands “A”, “B”, “C” and “D”, which ligand causes maximum crystal field splitting? Why?
OR
Which of the two, “A” or “D” will be a weak field ligand? Why?
(b) Which of the complexes will be violet in colour? [CrA6]3- or [CrB6]3+ and why?
(Given: If 560- 570 nm of light is absorbed, the colour of the complex observed is violet.)
(c) If the ligands attached to Cr3+ ion in the complexes given in the table above are water, cyanide ion, chloride ion, and ammonia (not in this order).
Identify the ligand, write the formula and IUPAC name of the following: (i) [CrA6]3- (ii) [CrC6]3+
Answer:
(a) Ligand ‘D’ causes maximum crystal field splitting because energy is directly proportional to the wave number, so maximum energy of light is required for an electron to jump from t2g to eg’ in case of [CrA6]3-
OR
A is weak field ligand because splitting caused is least in this case as the energy required for an
electron to jump from t2g to eg’ is minimum.
(b) The complex [CrB6]3- will be violet in colour because wavelength of light absorbed by this complex is \( \frac{1}{17830}\) = 0.000056085 or 560 nm and for the complex [CrA6]3- its value is \( \frac{1}{13640}\) = 733 nm
(c) (i) [CrCl6]3- = Hexachloridochromate(III) ion.
(ii) [Cr(NH3)6]3+ = Hexaamminechromium(III) ion.
A = Cl–, B = H2O, C = NH3, D = CN–
![]()
Question 30.
The lead-acid battery represents the oldest rechargeable battery technology. Lead acid batteries can be found in a wide variety of applications including small-scale power storage such as UPS systems, ignition power sources for automobiles, along with large, grid-scale power systems. The spongy lead act as the anode and lead dioxide as the cathode. Aqueous sulphuric acid is used as an electrolyte. The half-reactions during discharging of lead storage cells are:
Anode: Pb(s) + SO2-4 (aq) → PbSO4(s) + 2e–
Cathode: PbO2 (s) + 4H+ (aq) + SO2-4 (aq) + 2e– → PbSO4 (s) + 2H2O
There is no safe way of disposal and these batteries end – up in landfills. Lead and sulphuric acid are extremely hazardous and pollute soil, water as well as air. Irrespective of the environmental challenges it poses, lead-acid batteries have remained an important source of energy.
Designing green and sustainable battery systems as alternatives to conventional means remains relevant. Fuel cells are seen as the future source of energy. Hydrogen is considered a green fuel. Problem with fuel cells at present is the storage of hydrogen. Currently, ammonia and methanol are being used as a source of hydrogen for fuel cell. These are obtained industrially, so add to the environmental issues.
If the problem of storage of hydrogen is overcome, is it still a “green fuel?” Despite being the most abundant element in the Universe, hydrogen does not exist on its own so needs to be extracted from the water using electrolysis or separated from carbon fossil fuels. Both of these processes require a significant amount of energy which is currently more than that gained from the hydrogen itself. In addition, this extraction typically requires the use of fossil fuels. More research is being conducted in this field to solve these problems. Despite the problem of no good means to extract Hydrogen, it is a uniquely abundant and renewable source of energy, perfect for our future zero-carbon needs.
Answer the following questions:
(a) How many coulombs have been transferred from anode to cathode in order to consume one mole of sulphuric acid during the discharging of lead storage cell?
(b) How much work can be extracted by using lead storage cell if each cell delivers about 2.0 V of voltage? (1 F = 96500 C)
(c) Do you agree with the statement- “Hydrogen is a green fuel.” Give your comments for and against this statement and justify your views.
OR
Imagine you are a member of an agency funding scientific research. Which of the following projects will you fund and why?
(i) safe recycling of lead batteries
(ii) extraction of hydrogen
Answer:
(a) 2 mole or 2F have been transferred from anode to cathode to consume 2 mol of H2SO4 therefore one mol H2SO4 required IF (one faraday) of electricity or 96500 coulombs.
(b) We know that Wmax = -nFE°
N = 2
Given: F = 96500 coulomb
E° = 2.0V
Wmax = -2 × 96500 × 2.0
= 386000J
386000 J work can be extracted using lead storage cell when the cell is in use.
(c) Yes, “Hydrogen is a green fuel”. Both are means yes and no should be accepted as correct answer, depending upon what explanation is provided.
Yes: Hydrogen is a fuel that on combustion gives water as a by product. There are no carbon emissions and no pollution caused.
No: However, at present the means to obtain hydrogen are electrolysis of water which use electricity obtained from fossil fuel and increase carbon emission.
“Hydrogen is no doubt a green fuel but the process of extraction is not green as of today.”
OR
(i) Safe recyclic of lead batteries: Lead batteries are currently the most important and widely used batteries These are rechargeable. The problem is waste management. Which needs research and awareness. Currently, these are being thrown into landfills and there is no safe method of disposal. The plastic in this battery is mostly polypropylene and it can be recycled. The lead in the batteries is sold to companies that make new batteries.
(ii) Fuel cell is a clean source of energy. Hydrogen undergoes combustion to produce water. The need is green fuel and hydrogen as a clean fuel. The current problem is oblaining hydrogen. Research that goes into this area will help solve the problem of pollution and will be a subtainable solution.
Section – E (15 Marks)
The following questions are long answer type and carry 5 marks each. All questions have an internal choice.
Question 31:
Attempt any five of the following:
(a) Which of the following ions will have a magnetic moment value of 1.73 BM
Sc3+, Ti3+, Ti2+, Cu2+, Zn2+.
(b) In order to protect iron from corrosion, which one will you prefer as a sacrificial electrode, Ni or Zn? Why? (Given standard electrode potentials of Ni, Fe and Zn are- 0.25 V, -0.44 V and -0.76 V respectively.)
(c) The second ionization enthalpies of chromium and manganese are 1592 and 1509 kJ/mol respectively. Explain the lower value of Mn.
(d) Give two similarities in the properties of Sc and Zn.
(e) What is actinoid contraction? What causes actinoid contraction?
(f) What is the oxidation state of chromium in chromate ion and dichromate ion?
(g) Write the ionic equation for reaction of KI with acidified KMnO4.
Answer:
(a) Both Ti3+ and Cu2+ will have magnetic moment value of 1.73BM. Because both ions have single unpaired electron. By the electronic configuration of Ti3+ → 3d1
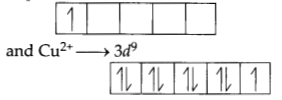
All ions which have single unpaired electron have magnetic moment 1.73 BM.
(b) Zn, because it has a more negative electrode potential so will corrode itself in place of ions.
(c) Electronic configuration of:
Mn+ → 3d54s1 and for Cr+ → 3d5
Therefore, ionisation enthalpy of Mn+ is lower than Cr+
(d) Sc and Zn both form colourless compound and are diamagnetic in nature.
(e) Actinoid Contraction: The decrease in the atomic and ionic radii with increase in atomic number of actinoids due to poor shielding effect of 5f electron.
(f) In both chromate and dichromate ion the oxidation state of Cr is + 6.
(g) The reaction of KI with acidified KMn04 can be expressed by following chemical ionic equation. 10I– + 2MnO4– + 16H+ → 2Mn2+ + 8H2O + 5I2
![]()
Question 32.
(a) What is the effect of temperature on the solubility of glucose in water?
(b) Ibrahim collected a lOmL each of fresh water and ocean water. He observed that one sample labeled ‘P’ froze at 0 °C while the other ‘Q’ at – 1.3°C. Ibrahim forgot which of the two, ‘F or ‘Q’ was ocean water. Help him identify which container contains ocean water, giving rationalization for your answer.
(c) Calculate van’t Hoff factor for an aqueous solution of K3[Fe(CN)6] if the degree of dissociation
(a) is 0.852. What will be boiling point of this solution if its concentration is 1 molal? (Kb = 0.52 K kg/mol)
OR
(a) What type of deviation from Rault’s law is expected when phenol and aniline are mixed with each other? What change in the net volume of the mixture is expected? Graphically represent the deviation.
(b) The vapour pressure of pure water at a certain temperature is 23.80 mm Hg. If 1 mole of a non-volatile non-electrolytic solute is dissolved in 100g water. Calculate the resultant vapour pressure of the solution.
Answer:
(a) Addition of glucose to water is an endothermic reaction. According to Le Chatelier’s principle, on increase in temperature, solubility will increase.
(b) Q is Ocean water, due to the presence of salts it freezes at lower temperature because of depression is freezing point.
(c) K3[Fe(CN)6] gives 4 ions in aqueous solution → K3[Fe(CN)6] → 3K+ + [Fe(CN)6]–
i = 1 + (n – 1)α
i = 1 + (4 – 1)α
Here, α = degree of dissociation = 0.852
i =1 + (4- 1) 0.852
i = 3.556
ΔTb = iKbm = 3.556 × 0.52 × 1 = 1.85
Tb = 101.85°C
OR
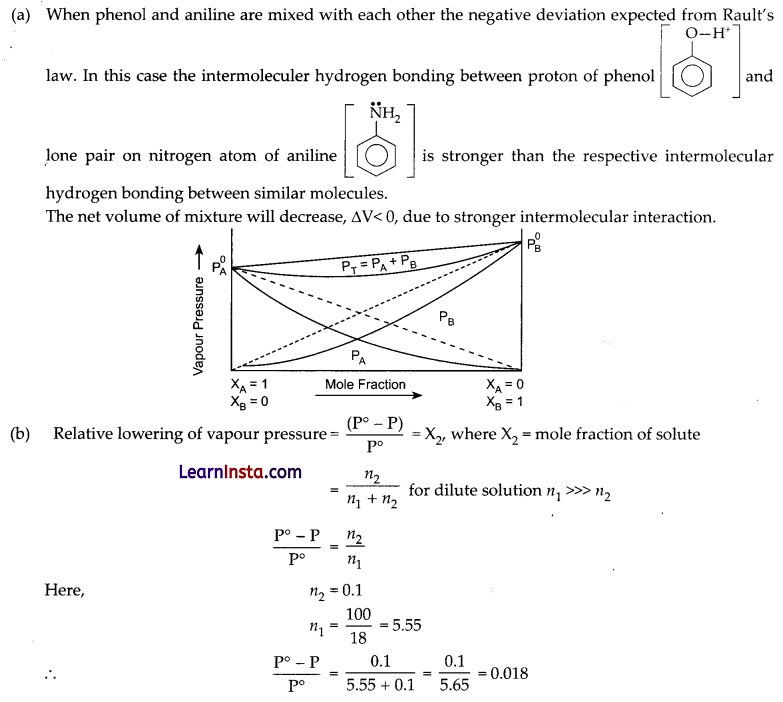
Relative lowering of vapour pressure = \(\frac{(23.80-P)}{23.80}\) = 0.018
23.80 – P = 23.80 × 0.018
23.80 – P = 0.428
P = 23.80 – 0.428
P = 23.37 mm Hg
Question 33.
An organic compound with molecular formula C7H7NO2 exists in three isomeric forms, the isomer ‘A’ has the highest melting point of the three. ‘A’ on reduction gives compound ‘B’ with molecular formula C7H9N. ‘B’ on treatment with NaNO2/HCl at 0-5 °C to form compound ‘C’. On treating C with H3PO2, it gets converted to D with formula C7H8, which on further reaction with CrO2Cl2 followed by hydrolysis forms ‘E’ C7H6O. Write the structure of compounds A to E . Write the chemical equations involved.
OR
(a) Account for the following:
(i) N-ethylbenzenesulphonyl amide is soluble in alkali .
(ii) Reduction of nitrobenzene using Fe and HCl is preferred over Sn and HCl.
(b) Arrange the following in:
(i) decreasing order of pKb values C6H5NH2, C6H5NHCH3, C6H5CH2NH2, CH3NH2, NH3
(ii) increasing order of solubility in water C2H5CI, C2H5NH2, C2H5OH
(iii) decreasing boiling point CH3COOH, C2H5OH, CH3NH2, CH3OCH3
Answer:
Given, molecular formula of organic compound = C7H7NO2
Three isomeric forms may be
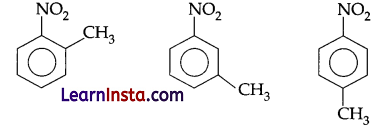
Compound A’ has highest melting point so it must be p-methylnitrobenzene. (p-isomeric form) on reduction nitrobenzene convert into corresponding amine. So ‘B’ must be methylbenzenamine. On treating with NaNO2/HCl, all amine gives diazonium chloride so compound C must be p-methyl benzenediazonium chloride which react with H2PO2 and gives Toluene which react with CrO2Cl2 followed by hydrolysis and gives benzaldehyde (E).
Whole chemical reaction given by following equation.
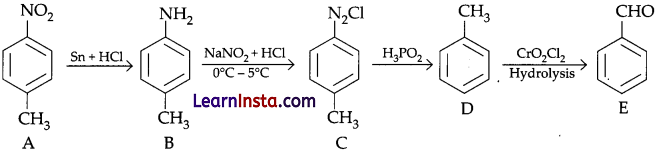
So, compound A = p-methylnitrobenzene
B = p-methylbenzenamine
C = p-methylbenzenediazonium chloride
D = Toluene (methyl benzene)
E = Benzaldehyde
OR
(a) Chemical
(i) Formula of N-ethylbenzenesulphonyl amide is
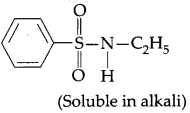
Here, the hydrogen attached to nitrogen is strongly acidic due to the presence of strong electron with drawing sulphonyl group. Hence it is soluble in alkali.
(ii) Reduction of nitrobenzene using Fe and HC1 is preferred over Sn and HCl because FeCl2 formed gets hydrolysed to release hydrochloric acid during the reaction. Thus, only a small amount of HC1 is required to initiate the reaction
(b) (i) C6H5NH2 > C6H5NHCH3 > NH3 > C6H5CH2NH2 > CH3NH2
pKb value of aniline C6H5NH2 is quite high because in aniline the – NH2 group is attached directly to the benzene ring. It results the unshared electron pair of ‘N’ to be in conjugation with benzene ring and thus it less available for protonation.
(ii) C2H5CI < C2H5NH2 < C2H5OH
Increasing order of extent of H-bonding in water, increases the solubility in water.
(iii) CH3COOH > C2H5OH > CH3NH2 > CH3OCH3
Due to intermolecular H-bonding carboxylic acids are higher boiling liquids than alcohols, amines, ether and carbonyl compounds.