These Class 7 Science Curiosity Worksheet and Class 7 Science Chapter 2 Exploring Substances Acidic Basic and Neutral Worksheet with Answers are excellent for board exam practice.
Class 7 Science Chapter 2 Exploring Substances Acidic Basic and Neutral Worksheet
Class 7 Exploring Substances Acidic Basic and Neutral Worksheet
Worksheet On Exploring Substances Acidic Basic and Neutral Class 7 – Exploring Substances Acidic Basic and Neutral Worksheet Class 7
A. Multiple-Choice Questions
Question 1.
Which of the following substances will turn blue litmus red?
(a) Baking soda
(b) Soap solution
(c) Lemon juice
(d) Salt solution
Question 2.
A student wants to test whether fruit juice contains acid. Which two indicators can confirm it?
(a) Litmus and turmeric
(b) Rose extract and blue litmus
(c) Red litmus and salt solution
(d) Turmeric and water
Question 3.
Hydrangea flowers turn blue in acidic soil. A gardener wants pink flowers. What should they add?
(a) Lemon juice
(b) Vinegar
(c) Lime
(d) Salt
![]()
Question 4.
This question consists of an Assertion (A) and a Reason (R). Read the Assertion and Reason and choose the appropriate answer.
Assertion (A): Onion is an olfactory indicator.
Reason (R): Its smell changes in acidic and basic media.
(a) Both A and R are true, and R is the correct explanation of A.
(b) Both A and R are true, but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false, but R is true.
Question 5.
This question consists of an Assertion (A) and a Reason (R). Read the Assertion and Reason and choose the appropriate answer.
Assertion (A): Turmeric paper can be used to identify acidic substances.
Reason (R): Turmeric turns red in basic substances.
(a) Both A and R are true, and R is the correct explanation of A.
(b) Both A and R are true, but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false, but R is true.
B. State True (T) or False (F).
1. Sugar solution is acidic.
2. Ant bites inject a basic liquid into the skin.
3. Soap solution feels slippery because it is acidic.
4. Lime water is a basic solution.
5. Litmus is obtained from lichens.
C. Fill in the blanks.
1. _____________ indicators change their odour in an acidic or basic medium.
2. _____________ is added to acidic soil to make it suitable for crops.
3. Substances that show no effect on litmus paper are called _____________
![]()
4. The basic substance used to treat ant bites is _____________
5. Vinegar contains the acid called _____________ acid.
D. Assign one word to the following terms.
1. A substance that changes colour in an acid or a base. _____________
2. A scientist known as the Father of Modern Indian Chemistry. _____________
3. Colour of hydrangea in acidic soil. _____________
4. Reaction between acid and base. _____________
5. The acid responsible for the sour taste in lemon. _____________
E. Match the Columns.
Question 1.
Match the statements given in Column I with the correct terms listed in Column II.
| Column I | Column II |
| (i) Does not change the colour of any indicator | (a) Tartaric acid |
| (ii) Natural indicator that changes its odour in basic solutions | (b) Adding lime/calcium oxide |
| (iii) Used in ancient and Ayurvedic medicine | (c) Neutral substance |
| (iv) Acidic compound responsible for the sour taste in tamarind | (d) Onion juice |
| (v) Remedy used by a farmer to fix acidic soil | (e) Turmeric |
Question 2.
Unjumble the words given in Column II and write them in the space provided with the help of the images given in Column I. Then match the images with the correct terms.

Wonder Why?
A. Read the paragraph and answer the following questions.
Litmus is a natural indicator derived from lichen that helps determine whether a substance is acidic, basic, or neutral. When tested with blue or red litmus paper, different household substances show distinct reactions. Acids like lemon juice turn blue litmus red, while bases like soap solution turn red litmus blue. Neutral substances, such as salt solution, do not change the colour of litmus paper. Acids usually taste sour, and bases feel slippery and are bitter in taste.
Question 1.
Name two substances that do not change the colour of either blue or red litmus paper.
____________________________________________________________
Question 2.
You have spilled a colourless liquid in the lab and want to know if it is acidic or basic. How would you test it using litmus paper?
____________________________________________________________
____________________________________________________________
Question 3.
Is it safe to identify all acids just by tasting them? Why/Why not?
____________________________________________________________
____________________________________________________________
![]()
Question 4.
Compare the reactions of vinegar and lime water with litmus paper. What do these reactions tell you about their nature?
____________________________________________________________
____________________________________________________________
B. Red rose indicator has been added to test tubes I and il, and the changes are shown in the image below. Observe the changes and answer the following questions.

Question 1.
Which of the test tubes contains lemon juice and which contains soap solution? Why?
____________________________________________________________
Question 2.
Name two substances that would show the same colour on adding red rose extract as in test tube II.
____________________________________________________________
Question 3.
What other natural substances, besides red rose petals, can be used as acid-base indicators? Give one example.
____________________________________________________________
C. Amisha loves solving mysteries. Today, she’s playing the role of a food scientist. Her mom, Usha, filled a basket with juicy and tangy fruits and vegetables. Amisha wants to identify the “tangy taste agents” – the natural acids hidden in each item.
Can you help Amisha match the correct acids to the fruits and vegetables below?

D. Complete the following table and find out the nature of the different substances. One example has been done for you.
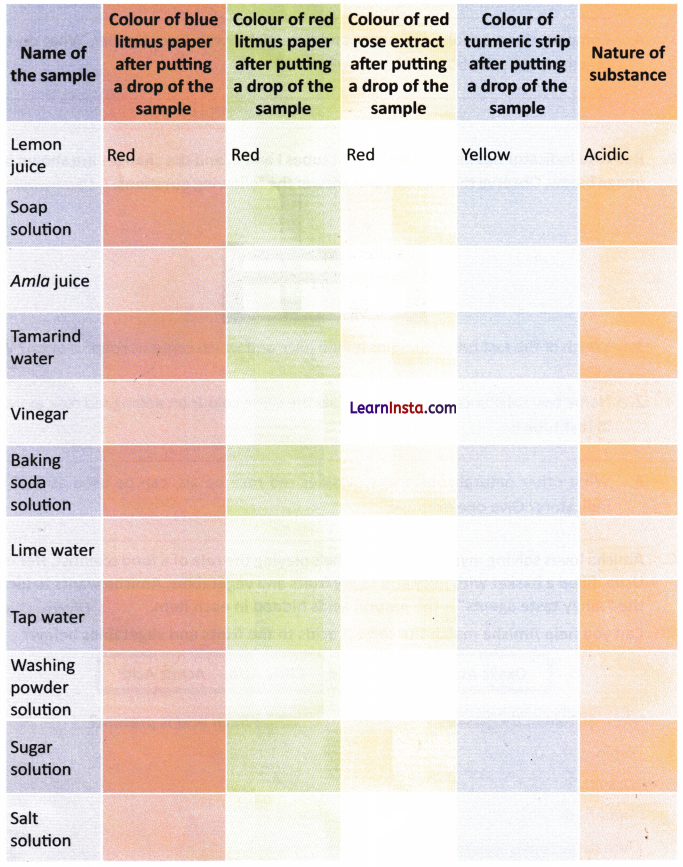
Learn By Doing
A. Take a few pieces of onion and crush them slightly to release their smell. Place the crushed onion on two small cotton strips or paper pieces. Smell both strips to observe the original odour. Now, add a few drops of lemon juice (acidic) to one strip and baking soda solution (basic) to the other. Gently smell both again. You will notice that the smell may disappear or weaken in one of them. Write down your observations and identify which substance caused a change in the smell.
____________________________________________________________
____________________________________________________________
____________________________________________________________
____________________________________________________________
____________________________________________________________
![]()
B. Take a drop of lemon juice in a test tube and mix it with about 20 drops of water. Add a drop of blue litmus solution — it turns red, showing the solution is acidic. Now, add lime water drop by drop while swirling the test tube. The red colour slowly changes back to blue, showing the acid has been neutralised by the base. Add another drop of lemon juice — the blue turns red again. Record your observations and find out how neutralisation reactions are useful in everyday life.
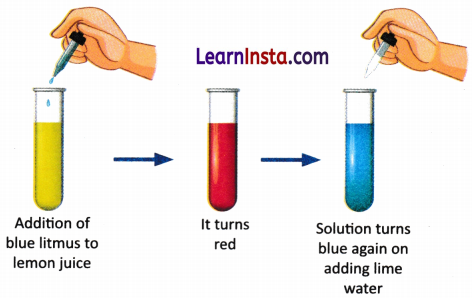
____________________________________________________________
____________________________________________________________
____________________________________________________________
____________________________________________________________
____________________________________________________________
Explore With Curiosity
Read the conversation between Ankit and Shubham and answer the following questions.

Question 1.
What could be the reason for the decline in fish population in the lake?
____________________________________________________________
____________________________________________________________
Question 2.
What should be done before releasing factory waste into the lake?
____________________________________________________________
____________________________________________________________
Question 3.
What can you conclude from Ankit and Shubham’s conversation?
____________________________________________________________
____________________________________________________________
B. Priya visits her cousin in Sikkim and sees Hydrangea plants with blue flowers. When she takes a cutting home to Delhi and plants it in her garden, the flowers turn pink. What could be the reason for the change in flower colour in Priya’s garden?

____________________________________________________________
____________________________________________________________
____________________________________________________________
____________________________________________________________
C. Rahul tested four substances with turmeric paper. All the strips remained yellow. His friend Riya said he must have made a mistake. Why did the turmeric paper not change colour in any test? What should Rahul do next to confirm if his turmeric paper is working properly?

____________________________________________________________
____________________________________________________________
____________________________________________________________
____________________________________________________________
D. Tanvi wants to surprise her mother by writing a secret message that appears only when a specific liquid is applied. She remembers the turmeric paper activity from class. Name one safe household substance she could use for writing, and explain your choice.
____________________________________________________________
____________________________________________________________
____________________________________________________________
![]()
E. Aarav was playing in the garden when an ant bit his younger sister. She started crying in pain. Aarav remembered what his teacher had taught in class and rushed to the kitchen. What do you think Aarav brought from the kitchen to help his sister, and why would it work?
____________________________________________________________
____________________________________________________________
____________________________________________________________
F. For a school science fair, Ishaan created a model showing how soil acidity affects plant growth. He wanted to demonstrate how to correct acidic soil using a common substance. What safe base could Ishaan add to the acidic soil in his mode to show improvement in plant growth?
____________________________________________________________
____________________________________________________________
____________________________________________________________
Word Puzzle
Complete the crossword with the help of the given clues.
ACROSS
2. The reaction between an acid and a base
6. It contains tartaric acid
7. A natural indicator from lichens
8. A natural yellow spice and indicator
DOWN
1. A plant whose flower colour changes with soil type
3. These indicators are made in laboratories
4. Indicators that change smell in acid or base
5. It is used to neutralise basic soil
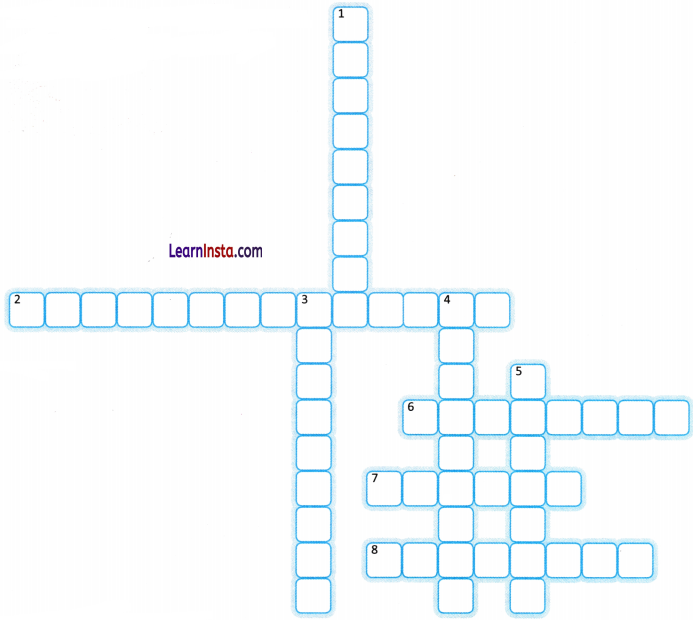
Activities
A. Create a simple design or pattern on chart paper using natural indicators like turmeric paste, beetroot juice, or red cabbage extract. After it dries, apply drops of common substances like lemon juice or vinegar (acidic), and soap water or baking soda solution (basic). Observe how the colours change in different areas and record your observations in your notebook.
![]()
B. Collect water samples from different sources, like rainwater, tap water, and pond water. Add a few drops of natural indicator, such as red cabbage juice or hibiscus extract, to each sample. Watch for any colour change. Use the result to determine whether the water is acidic, basic, or neutral. Discuss how the nature of water affects its use in drinking, cleaning, farming, or other activities.
C. Collect a sample of soil from your garden or neighborhood. Use natural indicators like red cabbage or beetroot juice to test the soil. Observe any colour change as you add the indicator and carefully note down your observations. Use the result to determine whether the soil is acidic, basic, or neutral.