NCERT Solutions for Class 12 Chemistry Chapter 15 provides answers to the questions provided in the textbook. The answers are accompanied by diagrammatic representations for better understanding. Also, the solutions are explained in such a language that the students find easy to understand.
NCERT Solutions are beneficial for the students appearing in UP board, MP board, CBSE, Gujarat board, etc. Also the students appearing for competitive exams such as NEET and JEE will find the NCERT Solutions for Class 12 Chemistry beneficial.
| Board | CBSE |
| Textbook | NCERT |
| Class | Class 12 |
| Subject | Chemistry |
| Chapter | Chapter 15 |
| Chapter Name | Polymers |
| Number of Questions Solved | 26 |
| Category | NCERT Solutions |
Class 12 Chemistry Chapter 15 Polymers NCERT Solutions
Polymers NCERT Solutions
Polymers are very large molecules having high molecular mass. These are also known as macromolecules. This chapter explains the mechanical properties and applications of polymers. The classification of polymers is also described in detail.
The chapter gives an overview of different polymerization reactions. The chapter explains that the polymers are the backbone of four major industries, plastic, elastomers, fibres and paints.
NCERT INTEXT QUESTIONS
Question 1.
What are polymers?
Answer:
The word polymer has a Greek origin where poly means many and mers or meros stands for unit or part. Thus, the polymer may be defined as a substance of a high molecular mass (103 – 107 u) formed by the combination of a large number of simple
molecules called monomers by chemical bonds. The process by which monomers are converted into polymers is called polymerization.
Question 2.
How are polymers classified on the basis of structure?
Answer:
On the basis of structure, polymers are classified as:
(i) Linear polymers in which the monomers are joined together to form long straight chains of polymer molecules. Forex: HDPE, PVC, nylons, etc.
(ii) Branched-chain polymers in which the monomers not only join in a linear fashion but also form branches of different lengths along the main chain. For ex: LDPE, glycogen, etc.
(iii) Cross-linked polymers in which the initially formed linear polymer chains join together to form a 3D network structure. For ex: bakelite, Urea-formaldehyde resin, etc.
Question 3.
Write the names of the monomers of the following polymers:
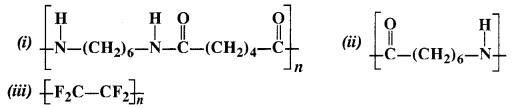
Answer:
(i) The polymer is Nylon 66. Its monomer units are hexamethylenediamine and adipic acid.
(ii) The polymer is Nylon 6. Its monomer units are of caproilactum.
(iii) The polymer is Teflon. Its monomer units are of tetrafluoroethylene.
Question 4.
Classify the following as addition and condensation polymers:
(i) Terylene
(ii) Bakelite
(iii) Polyvinyl chloride
(iv) Polythene
Answer:
(i) Condensation polymer
(ii) Condensation polymer
(iii) Addition polymer
(iv) Addition polymer
Question 5.
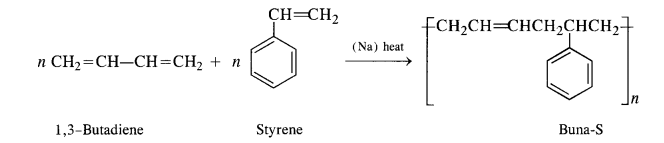
Explain the difference between Buna-N and Buna-S.
Answer:
Both Buna-N and Buna-S are synthetic rubber and are co-polymers in nature. They differ in their constituents.
Buna-N: Constituents are : buta-1, 3-diene and acrylonitrile.
Buna-S: Constituents are : buta-1, 3-diene, and styrene. They condense in the presence of Na.
Buna – S: It is a co—polymer of 1. 3 – butadiene and styrene and is prepared by the polymerisation of these components in the
ratio of 3 : 1 in the presence of sodium.
Buna-N (Nitrile rubber): h is a co-polymer of buta-1. 3-diene and acrylonitrile. It is formed as follows:
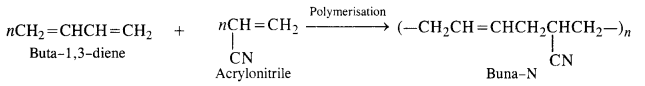
Question 6.
Arrange the following polymers in increasing order of their intermolecular forces :
(i) Nylon-66, Buna-S, Polythene
(ii) Nylon-6, Neoprene, Polyvinyl chloride
Answer:
We have studied the classification of polymers based upon intermolecular forces. These follow the order :
Elastomers < Plastics < Fibres.
The polymers listed may be arranged in increasing order of intermolecular forces as follows :
(i) Buna-S (Elastomer) < Polythene (Plastic) < Nylon-66 (Fibre)
(ii) Neoprene (Elastomer) < Polyvinyl chloride (Plastic) < Nylon-6 (Fibre).
NCERT Exercise
Question 1.
Explain the terms polymers and monomers.
Answer:
Polymers are a high molecular mass of macromolecules composed of repeating structural units derived from monomers. Polymers have a high molecular mass (103-107u). In a polymer, various monomer units are joined by strong covalent bonds. Polymers can be natural as well as synthetic. Polythene, rubber, and nylon 6,6 are examples of polymers.
Monomers are simple, reactive molecules that combine with each other in large numbers through covalent bonds to give rise to polymers. For example ethene, propene, styrene, vinyl chloride.
Question 2.
What are natural and synthetic polymers? Give two examples of each.
Answer:
Natural polymers:
Natural polymers are high molecular mass macromolecules and are found in nature mainly in plants and animals. For example, protein, nucleic acids, starch, cellulose, etc.
Synthetic polymer:
Synthetic polymers are man-made high molecular mass macromolecules. For example, plastics (Polyethene, P.V.C.), synthetic fibers (Polyesters, Nylon-6,6), synthetic rubber (Neoprene, Buna-S), etc.
Question 3.
Distinguish between the terms homopolymer and copolymer and give one example of each. (C.B.S.E. Outside Delhi 2008, Pb. Board 2008)
Answer:
A polymer in which the monomers are the same is called a homopolymer. For example, polystyrene is made from styrene only.
A polymer in which the monomers are different is known as a copolymer. For example, in Nylon-66 the monomers are adipic acid and hexamethylenediamine.
Question 4.
How do you explain the functionality of a monomer?
Answer:
The functionality of a monomer is the number of binding sites that is/are present in that monomer.
For example, the functionality of monomers such as ethene and propene is one and that of 1, 3-butadiene, and adipic acid is two.
Question 5.
Define the term polymerisation. (C.B.S.E. Delhi 2008)
Answer:
A very widely used polymer is polyethylene which is formed by the polymerisation of ethylene molecules (monomers) by heating under pressure in the presence of oxygen.
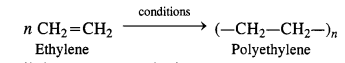
It may be noted that all the monomer units in a polymer may or may not be the same, In case, they are the same as in case of polyethylene, then the polymer is called homopolymer. On the other hand, if they happen to be different, then the polymer is known as copolymer or mixed polymer. For example, the monomer units in terylene are ethylene glycol and terephthalic acid. Similarly, hexamethylenediamine and adipic acid are the monomer units of nylon-66. Both are co-polymers or mixed polymers. We shall discuss these at a later stage in the present unit.
Question 6.
![]()
Answer:
It is a homopolymer in nature. It has only one type of monomer units i.e., NH2 – CHR – COOH. These area-amino acids.
Question 7:
In which classes, are the polymers classified on the basis of molecular forces?
Answer:
Polymers are classified into four classes on the basis of molecular forces. These are:
elastomers, fibres, thermoplastic polymers, and thermosetting polymers.
1. Elastomers: In these polymers, the intermolecular forces are the weakest. As a result, they can be readily stretched by applying small stress and regain their original shape when the stress is removed. The elasticity can be further increased by introducing some cross-links in the polymer chains. Natural rubber is the most popular example of elastomers. A few more examples are of: buna-S, buna-N and neoprene.
2. Fibres: Fibres represent a class of polymers which are thread-like and can be woven into fabrics in a number of ways. These are widely used for making clothes, nets, ropes, gauzes etc. Fibres possess high tensile strength because the chains possess strong intermolecular forces such as hydrogen bonding. These forces are also responsible for close packing of the chains. As a result, the fibres are crystalline in nature and have also sharp melting points. A few common polymers belonging to this class are nylon – 66, terylene and polyacrylonitrile etc.
3. Thermoplastics: These are linear polymers and have weak van der Waals forces acting in the various chains and are intermediate of the forces present in the elastomers and in the fibres. When heated, they melt and form a fluid which sets into a hard mass on cooling, Thus, they can be cast into different shapes by using suitable moulds. A few common examples are polyethylene and polystyrene polyvinyls etc. These can be used for making toys, buckets, telephone apparatus, television cabinets etc.
4. Thermosetting plastics: These are normally semifluid substances with low molecular masses. When heated, they become hard and infusible due to the cross-linking between the polymer chains. As a result, they also become three-dimensional in nature. They do not melt when heated. A few common thermosetting polymers are bakelite, melamine-formaldehyde, urea-formaldehyde and polyurethane etc.
Question 8:
How can you distinguish between addition and condensation polymerisation?
Answer:
In addition polymerization: The monomers are unsaturated in nature i.e., they have atleast one double or triple bond in their molecules. They represent the functionality of the monomers which combine with each other at these sites. The addition polymerisation is generally chain growth polymerisation in nature. For example, polythene, polystyrene, PVC, Teflon etc. are all formed as a result of additional polymerisation.
In condensation polymerization: The monomer units have specific functional groups present which represent their functionality. The monomers combine through these functional groups and the polymerisation is step growth polymerisation in nature. For example, terylene, nylon-6, nylon-66, bakelite etc. are all formed as a result of condensation polymerisation.
Question 9:
Explain the term co-polymerization and give two examples.
Answer:
Co-polymerisation is a process in which a mixture of more than one different monomeric units are allowed to polymerise. The copolymer thus formed contains multiple units of each monomer in the chain. The formation of buna-S copolymer in which the monomers are : buta-1, 3-diene, styrene and sodium is an example of copolymerisation.
Another Example of co-polymer: Buna-S is a co-polymer of 1 : 3 – butadiene and styrene in the presence of sodium metal.
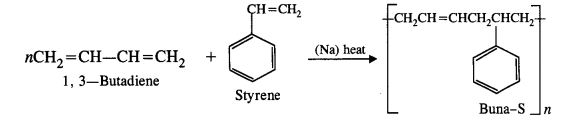
Thus, as a result of addition polymerisation, the double bonds change to single bonds. A large number of monomer units can be linked in this way.
The addition polymerisation is normally a chain reaction in which the chain is built up by the successive addition of the monomer units. It involves a number of intermediates such as free radicals, carbocations or carbanions. Let us illustrate the chain growth polymerisation based upon different mechanisms.
Question 10:
Write the free radical mechanism for the polymerisation of ethene.
Answer:
The polymerisation of ethene to polythene consists of heating or exposing to light a mixture of ethene with a small amount of benzoyl peroxide initiator The process starts with the addition of phenyl free radical formed by the peroxide to the ethene double bond thus regenerating a new and larger free radical. This step is called chain initiating step. As this radical reacts with another molecule of ethene, another bigger sized radical is formed. The repetition of this sequence with new and bigger radicals carries the reaction forward and the step is termed as chain propagating step. Ultimately; at some stage the product radical thus formed reacts with another radical to form the polymerised product. This step is called the chain terminating step. The sequence of steps may be depicted as follows :
Chain initiating steps:
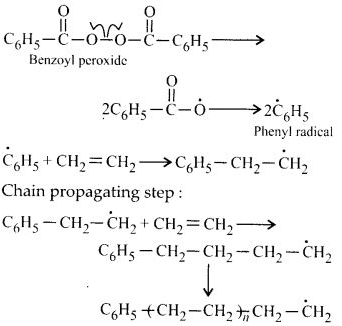
Chain terminating step:
For termination of the long-chain, these radicals can combine in different ways to form polythene. One mode of termination of the chain is shown as under:
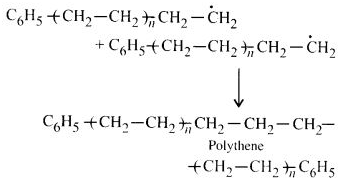
Question 11:
Define thermosetting and thermoplastic polymers with two examples of each.
Answer:
Thermoplastics are polymers which can be easily softened repeatedly on heating and hardened on cooling. Therefore, it can be used again and again. For example, polyethylene and polyvinyl chloride. Thermosetting polymers are those which undergo permanent change on heating. They become hard and infusible on heating and cannot be softened again. For example, Bakelite, and Melamine formaldehyde.
Question 12:
Write the monomers used for getting the following polymers :
- Polyvinyl chloride
- Teflon
- Bakelite
Answer:
- vinyl chloride
- tetrafluoroethylene
- phenol and formaldehyde.
Question 13:
Write the name and structure of one of the common initiators used in free radical addition polymerisation.
Answer:
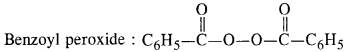
Question 14:
How does the presence of double bonds in rubber molecules influence their structure and reactivity?
Answer:
Chemically natural rubber is polyisoprene in which monomer units are of isoprene i.e., 2-methyl-i, 3-butadiene. It is in fact, 1, 4 polymer in which monomers are linked through CH2 groups located at 1.4 positions. The residual double bonds are located at C2 and C3 positions in the isoprene units. All the double bonds have cis configurations. Thus, natural rubber is cis polyisoprene.
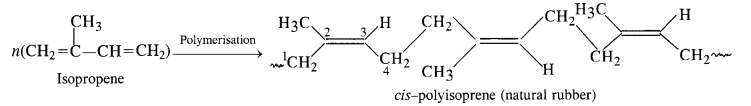
The high elasticity of natural rubber is due to the absence of polar groups and the cis-configurations about double bonds do I not allow the polymer chains to come closer. Therefore, only weak van der Waals’ forces are present. Since the chains are not linear, they can be stretched just like springs and exhibit elastic properties.
Question 15:
Discuss the main purpose of vulcanisation of rubber.
Answer:
The main purpose of vulcanization of rubber is to improve the following draw-back of natural rubber:
- At high temperature (T >335K) natural rubber becomes soft.
- At low temperature (T< 283K) natural rubber becomes brittle.
- Natural rubber is soluble in non-polar solvents.
- It is non-resistant to attack by oxidizing agents.
Question 16:
What are the monomers repeating units of Nylon 6 and Nylon 66?
Answer:
The monomeric repeating unit of nylon 6 is [NH – (CH2)5 – CO], which is derived from caprolactam.
The monomeric repeating unit of nylon 6, 6 is [NH – (CH2)6 – NH – CO – (CH2)4 – CO], which is derived from hexamethylene diamine and adipic acid.
Question 17:
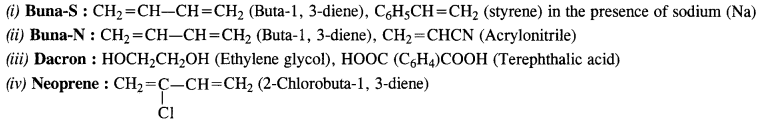
Write the names and structures of the monomers of the following polymers:
(i) Buna-S
(ii) Buna-N
(iii) Dacron
(iv) Neoprene (C.B.S.E. Delhi 2008)
Answer:
Question 18:
Identify the monomers in the following polymeric structures:
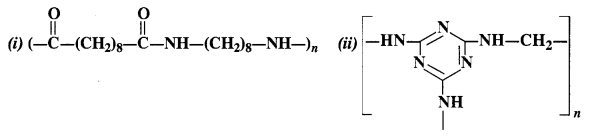
Answer:
(i) Decanedioic acid: HOOC(CH2)8COOH
Octamethylenediamine: H2N(CH2)8NH2
(ii)
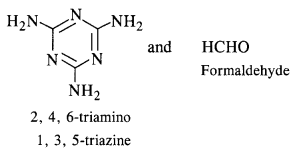
Question 19:
How is dacron obtained from ethylene glycol and terephthalic acid?
Answer:
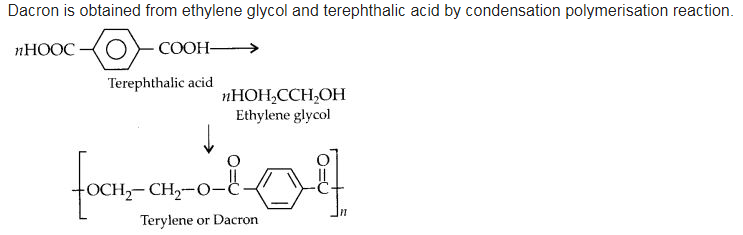
Question 20:
What is a biodegradable polymer? Give an example of an aliphatic biodegradable polyester.
Answer:
A polymer that can be decomposed by bacteria is called a biodegradable polymer. Poly – β- hydroxybutyrate – Co -β – hydroxy valerate (PHBV) is a biodegradable aliphatic polyester.
We hope the NCERT Solutions for Class 12 Chemistry Chapter 15 Polymers help you. If you have any query regarding NCERT Solutions for Class 12 Chemistry Chapter 15 Polymers, drop a comment below and we will get back to you at the earliest.
Class 12 Chemistry NCERT Solutions
- The Solid State Class 12 NCERT Solutions
- Solutions Class 12 NCERT Solutions
- Electrochemistry Class 12 NCERT Solutions
- Chemical Kinetics Class 12 NCERT Solutions
- Surface Chemistry Class 12 NCERT Solutions
- General Principles and Processes of Isolation of Elements Class 12 NCERT Solutions
- The p-Block Elements Class 12 NCERT Solutions
- d-and f-Block Elements Class 12 NCERT Solutions
- Coordination Compounds Class 12 NCERT Solutions
- Haloalkanes and Haloarenes Class 12 NCERT Solutions
- Alcohols, Phenols and Ethers Class 12 NCERT Solutions
- Aldehydes, Ketones and Carboxylic Acids Class 12 NCERT Solutions
- Amines Class 12 NCERT Solutions
- Biomolecules Class 12 NCERT Solutions
- Polymers Class 12 NCERT Solutions
- Chemistry in Every Day Life Class 12 NCERT Solutions