Students can access the CBSE Sample Papers for Class 11 Chemistry with Solutions and marking scheme Set 4 will help students in understanding the difficulty level of the exam.
CBSE Sample Papers for Class 11 Chemistry Set 4 with Solutions
Time Allowed : 3 hours
Maximum Marks: 70
General Instructions:
- There are 33 questions in this question paper with internal choice.
- Section – A consists of 16 multiple -choice questions carrying 1 mark each.
- Section – B consists of 5 short answer questions carrying 2 marks each.
- Section – C consists of 7 short answer questions carrying 3 marks each.
- Section – D consists of 2 case – based questions carrying 4 marks each.
- Section – E consists of 3 long answer questions carrying 5 marks each.
- All questions are compulsory.
- Use of log tables and calculators is not allowed.
Section-A
The following questions are multiple-choice questions with one correct answer. Each question carries 1 mark. There is no internal choice in this section.
Question 1.
Which of the following will not show deflection from the path on passing through an electric field?
(A) Proton
(B) Cathode rays
(C) Electron
(D) Neutron
Answer:
(D) Neutron
Explanation:
Neutron will not show deflection from the path on passing through an electric field because it is neutral.
Question 2.
An atom having atomic mass number 15 has 7 neutrons. The atomic number of the atom will be:
(A) 6
(B) 10
(C) 5
(D) 8
Answer:
(D) 8
Explanation:
Mass number = 15 Number of neutrons = 7
Mass number = No. of protons + No. of neutrons
15 = No. of protons + 7
No. of protons = 15 – 7 = 8
Atomic number = No. of protons = 8
Question 3.
The element with atomic number 57 belongs to:
(A) s-block
(B) p-block
(C) d-block
(D) f-block
Answer:
(C) d-block
Question 4.
Which of the elements whose atomic numbers are given below, cannot be accommodated in the present set up of the long form of the periodic table?
(A) 107
(B) 118
(C) 126
(D) 102
Answer:
(C) 126
Explanation:
The element with atomic number 126 cannot be accommodated in the long form of the periodic table.
Question 5.
The elements with atomic numbers 35,53 and 85 are all:
(A) noble gases
(B) halogens
(C) heavy metals
(D) light metals
Answer:
(B) halogens
Explanation:
The elements with atomic numbers 35,53,85 are all halogens as they have 7 electrons in their outermost shells.
Z = 35 Bromine – electronic configuration is 1s22s2 2p63s23p63d104s24p5
Z = 53 Iodine – electronic configuration is 1S22S2 2p63s23p64s23d104p65s24D25p5
Z= 85 Astantine – electronic configuration is [Xe] 4f14 5d10 6S2 6p5
Question 6.
Which of the following statement is not correct from the view point of molecular orbital theory?
(A) Be2 is not a stable molecule.
(B) He2 is not stable but He2+ is expected to exist.
(C) Bond strength of N2 is maximum amongst the homonuclear diatomic molecules belonging to the second period.
(D) The order of energies of molecular orbitals in N2 molecule is
σ2s < σ*2S < σ2pz <(π2px = π2py) < (π*2px = π*2py) < σ*2pz
Answer:
(B) He2 is not stable but He2+ is expected to exist.
Explanation:
(i) Be2 = σ1S2 σ*1S2 σ2S2 σ*2S2
Bond order = \(\frac{4-4}{2}\) = 0
So, being unstable it does not exist.
(ii) He2 = σ1S2 σ*1S2
Bond order = \(\frac{2-2}{2}\) = 0
So, being unstable it does not exist.
He2+ = σ1S2 σ*1S1
Bond order = \(\frac{2-1}{2}\) = 0.5
So, being stable it is expected to exist.
(iii) N2< = σ1S2< σ*1S2 σ2S2 σ*2S2 π2px2 = π2py2 σ2pz2
Bond order = \(\frac{10-4}{2}\) = 3
Thus, dinitrogen molecule has triple bond while no molecule of second period has more than double bond. Thus, its bond strength is maximum amongst the homonuclear diatomic molecules belonging to second period.
(iv) The correct order of N2 molecule is σ1S < σ*1S < σ2S < σ*2S < (π2px2 = π2py2) < σ2pz < π*2px = π2py < σ*2pz.
Question 7.
Which of the following is intensive property?
(A) Molarity
(B) Temperature
(C) Density
(D) All of these
Answer:
(D) All of these
Explanation:
An intensive property, is a physical property of a system that does not depend on the system size or the amount of material in the system.
Question 8.
In adiabatic process:
(A) q > 0
(B) q -1
(C) q = 0
(D) q < 0
Answer:
(C) q = 0
Explanation:
Adiabatic process is the change occurring within a system as a result of transfer of energy to or from the system in the form of work only, i.e., no heat is transferred
Question 9.
Which of the following options will be correct for the stage of half completion of the reactions: A ⇌ B
(A) ΔGθ = 0
(B) ΔGθ > 0
(C) ΔGθ < 0
(D) ΔGθ = -RT ln2
Answer:
(A) ΔGθ = 0
Explanation:
ΔG° = -RT ln K At the stage of half completion of reaction, [A] = [B]. ; K = 1
Thus, ΔG° = 0
Question 10.
Which of the following arrangements represent increasing oxidation number of the central atom?
(A) CrO2–, C1O–, CrO42-^, MnO4–
(B) ClO3–“, CrO42-, MnO4–,CrO2–
(C) CrO2–, C1O3–, MnO4–, CrO42-”
(D) CrO42-, MnO4–, CrO2–“, CIO3–,
Answer:
(A) CrO2–, C1O–, CrO42-^, MnO4–
Explanation:
Oxidation number of central atom in the given arrangement:
![]()
Question 11.
In the reaction, when H2 combines with O2 to form water, there is an electron transfer from:
(A) O2 to H2O
(B) H to O
(C) O to H
(D) No electron transfer
Answer:
(B) H to O
Explanation:
The H atom goes from a neutral (zero) state in H2 to a positive state in H2O.
Question 12.
Using the standard electrode potential, find out the pair between which redox reaction is not feasible.
Eθ values : Fe3+/Fe2+ = +0.77; I2I– = +0.54:
Cu2+/Cu = +0.34: Ag+/Ag = +0.80 V
(A) Fe3+ and I–
(B) Ag+ and Cu
(C) Fe3+ and Cu
(D) Ag and Fe3+
Answer:
(D) Ag and Fe3+
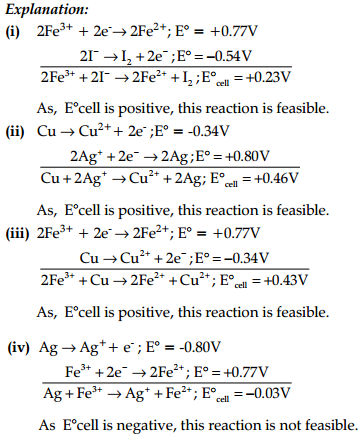
Question 13.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Combustion of all organic compounds is an exothermic reaction.
Reason (R): The enthalpies of all elements in their standard state are zero.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(B) Both A and R are true but R is not the correct explanation of A.
Explanation: Combustion reactions breaks the bonds of organic compound molecules, and the resulting water and carbon dioxide bonds always release more energy than was used to break them originally. That s why burning organic compounds produces energy and is exothermic.
Question 14.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): A solution containing a mixture of acetic acid and sodium acetate maintains a constant value of pH on addition of small amounts of acid or alkali.
Reason (R): A solution containing a mixture of acetic acid and sodium acetate acts as a buffer solution around pH 4.75. ‘
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A
Question 15.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): For any chemical reaction at a particular temperature, the equilibrium constant is fixed and is a characteristic property.
Reason (R): Equilibrium constant is independent of temperature.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A
Explanation:
As the concentration of reactants or products does not affect equilibrium constant at a particular temperature. Also, equilibrium constant varies with temperature.
Question 16.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): In the reaction between potassium permanganate and potassium iodide, permanganate ions act as oxidising agent.
Reason (R): Oxidation state of manganese changes from +2 to +7 during the reaction.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(C) A is true but R is false.
Explanation:
In the reaction mention in assertion, the oxidation number changes from +7 to +4.
Section-B
This section contains 5 questions with internal choice in one question. The following questions are very short answer type and carry 2 marks each.
Question 17.
Explain why:
(A) The three electrons present in 2p sub-shell of nitrogen remain unpaired.
(B) Cr has configuration 3ds4s^ and not 3d44s2.
Answer:
(A) The three electrons present in 2p sub-shell of nitrogen remain unpaired because 2p sub¬shell contains three orbitals 2px, 2py and 2pz of same energy. So, according to Hund’s rule of maximum multiplicity, pairing of electrons in these orbitals belonging to the same sub-shell does not take place until each orbital belonging to that sub-shell is singly occupied.
(B) Cr has configuration 3d54s1 and not 3d44s2 because in configuration 3d54s1, 3d sub-shell is half-filled which is more stable configuration as compared to partially filled configuration.
Question 18.
Explain why PC15 is trigonal bipyramidal where as IF5 is square pyramidal?
Answer:
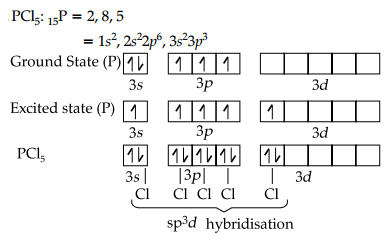
In PCl5, the central atom contains five valence electrons. All these electrons are used to form bond with five Cl-atoms. So, there is no lone pair present in PCl5. It possesses sp3d hybridisation. So, its shape in trigonal bipyramidal. Its structure is shown as below:
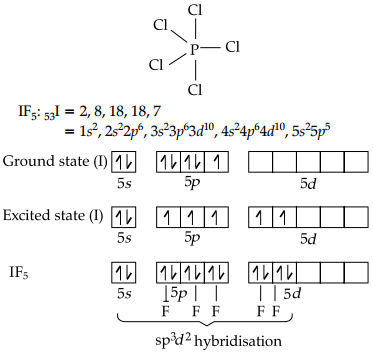
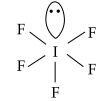
In IF5, the central atom contains 7 valence electrons. Out of which, 5 electrons are used to form bonds with five fluorine atoms. The remaining two electrons are present as one lone pair. It possesses sp3d2 hybridisation. But, due to lp-bp repulsion, shape of IF5 is distorted i.e., square pyramidal. Its structure is shown as below:
Question 19.
(A) The enthalpy of atomization of CH4 is 1665 kj mol-1. What is the bond enthalpy of C-H bond?
(B) If enthalpy of fusion and enthalpy of vaporisation of sodium metal are 2.6 kj mol-1 and 98.2 kj mol-1 respectively, calculate the enthalpy of sublimation of sodium.
Answer:
(A) ∆aH° of CH4 = 1665 kJ mol-1
Number of covalent bonds (C – H) in CH4 = 4
∴ Bond enthalpy of C-H bond = \(\frac{1665 \mathrm{~kJ} \mathrm{~mol}^{-1}}{4}\)
= 416.25 KJ mol-1
(B) ∆fusHθ = 2.6 kJ mol-1
∆vapHθ = 98.2 kJ mol-1
∴ ∆subHθ = ∆fusHθ + ∆vapHθ
= 2.6 + 98.2
= 100.8 KJ mol-1
Question 20.
(A) What type of organic compounds cannot be Kjeldahlised?
(B) How will you separate a mixture of o-nitrophenol and p-nitrophenol ?
OR
For the following bond cleavages, use curved-arrows to show the electron flow and classify each as homolysis or heterolysis. Identify reactive intermediate produced as free radical, carbocation and carbanion.
![]()
Answer:
(A) The compounds with a nitrogen atom inside the ring as well as those in which nitrogen is directly connected to either oxygen or another nitrogen atom like -NO2 group (nitro) and —N = N— group (azo) cannot be Kjeldahlised.
(B) It can be separated by steam distillation. o-nitrophenol being volatile distills over along with water while p-nitrophenol being non¬volatile remains in the flask.
Or
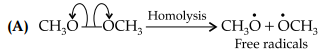
It is an example of homolytic cleavage as one of the shared pair in a covalent bond goes with the bonded atom. The reaction intermediate formed is a free radical.
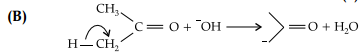
It is an example of heterolytic cleavage as the bond breaks in such a manner that the shared pair of electrons remains with the carbon of propanone. The reaction intermediate formed is carbanion.
Question 21.
(A) Write hydrocarbon radicals that can be formed as intermediates during monochlorination of 2-methylpropane?
(B) Which of them is more stable? Give reasons.
Answer:
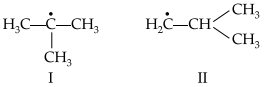
(B) Radical I is tertiary whereas radical II is primary. Radical I is more stable due to hyperconjugation
Section-C
This section contains 7 questions with internal choice in one question. The following questions are short answer type and carry 3 marks each. (No internal choice given in Board SQP)
Question 22.
Convert the following into basic units:
(A) 28.7 pm,
(B) 15.15 pm,
(C) 25365 mg.
Answer:
∴ 1 pm = 10-12m
∴ 28.7 pm = 28.7 × 10-12 m or 2.87 × 10-11 m
(B) 1 pm = 10-12 m
∴ 15.15 pm = 15.15 × 10-12 m or 1.515 × 10-11 m
(C) ∴ 1 mg = 106 kg
∴ 2.5365 × 10-2 mg = 25365 × 10-6 kg
= 2.5365 × 10-2 kg
Question 23.
Give reason for following:
(A) Halogens act as good oxidising agents.
(B) A cation is always smaller than its parent atom.
(C) Na and Mg+ have same number of electrons but removal of electron from Mg+ requires more energy.
Answer:
(A) Halogens act as good oxidising agents on account of their high value of reduction potential, they have more tendency to attract electrons.
(B) It is due to increase in effective nuclear charge due to decrease in number of electrons but protons remains the same.
(C) The effective nuclear charge increases across a period. Both Na and Mg+ has same numbers of electrons in outermost shell, but Mg+ has more effective nuclear charge than Na. As the effective nuclear charge increases, the hold of the nucleus on the outermost electrons will also increase. Thus, it will be very difficult to remove the electrons from outermost shell. That’s why, it is difficult to remove electron from Mg+ as compared to Na.
Question 24.
(A) What is enthalpy change at constant volume? Explain.
(B) Calculate the enthalpy of transition for carbon from the following:
Cdiamond + O2 → COdiamond(g); ∆H = – 943 kcal
Camorphous + O2 → CO2(g); ∆H = – 97-6 kcal
Also calculate the heat required to change 1 g of Cdiamond to Camorphous
Answer:
(A) When reaction is carried out in a closed vessel
so that volume remains constant, i.e., ∆V = 0, then qp = qv = ∆V or ∆H = ∆U. This is known as enthalpy change at constant volume.
(B) Heat of transition of Cdiamond to Camorphous
Cdiamond + O2 → CO2(g); ∆H = -94.3 Kcal …..(1)
Camorphous + O2 → CO2(g); ∆H = -97.6 Kcal …..(2)
Subtract equation (2) from (1)
Cdiamond – Camorphous = -94.3 – (-97.6)
= 3.3 Kcal
This heat represents the transformation of 1 mole
Cdiamond to Camorphous
Therefore, heat of transformation for 1 g of
Cdiamond to 1 g Camorphous = 3.3 kcal/12
= 0.275 Kcal.
Question 25.
What is the difference between distillation, distillation under reduced pressure and steam distillation?
Answer:
Distillation:
It is used for the purification of compounds that are associated with non-volatile impurities or those liquids, which do not decompose on boiling. Mixture of petrol and kerosene is separated by this method.
Distillation under reduced pressure:
This method is used to purify a liquid that tends to decompose on boiling. Under the conditions of reduced pressure, the liquid will boil at a low temperature than its boiling point and will, therefore, not decompose. Glycerol is purified by this method. It boils with decomposition at a temperature of 593 K. At a reduced pressure, it boils at 453 K without decomposition.
Steam distillation:
It is used to purify an organic compound, which is steam volatile and immiscible in water. On passing steam, the compound gets heated up and the steam gets condensed to water. After sometime, the mixture of water and liquid starts to boil and passes through the condenser. This condensed mixture of water and liquid is then separated by using a separating funnel. A mixture of water and aniline is separated by steam distillation.
Question 26.
What is the relationship between the members of following pairs of structures? Are they structural or geometrical isomers or resonance contributors?
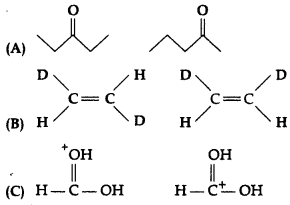
Answer:
(A) The given compounds have the same molecular formula but they differ in the position of the functional group, so they are structural isomers.
(B) The given compounds having the same molecular formula, the same constitution, and the sequence of covalent bonds, but with different relative position of their atoms in space, so they are called geometrical isomers. (1)
(C) The given compounds are resonance contributors or canonical structures because they differ in the position of electrons but not atoms.
Question 27.
Explain the method of estimation of phosphorus element by Carius Method.
Answer:
Estimation of Phosphorus:
A known mass of an organic compound is heated with fuming nitric acid whereupon phosphorus present in the compound is oxidised to phosphoric acid. It is precipitated as ammonium phosphophosphomolybdate, (NH4)3PO4. 12MoO3, by adding ammonia and ammonium molybdate. Alternatively, phosphoric acid may be precipitated as MgNH4PO4 by adding magnesia mixture which on ignition yields Mg2P2O7.
Let the mass of organic compound taken = m g and mass of ammonium phosphomolybdate = ml g Molar mass of (NH4)3PO4.12MO3 = 1877 g Percentage of phosphorus = (31 × ml × 100) / (1877 × m)%
If phosphorus is estimated as Mg2P2O7,
Percentage of phosphorus = 62 × ml × 100/222 × m%
Question 28.
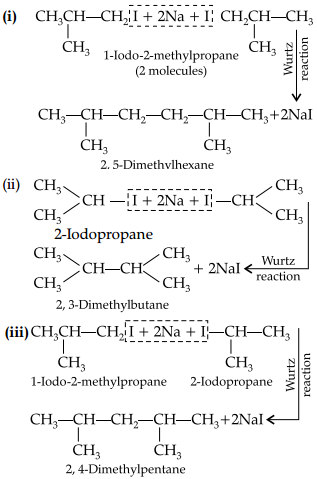
Write the structures and names of products obtained in the reactions of sodium with a mixture of l-iodo-2-
methylpropane and 2-iodopropane.
Answer:
Section-D
The following questions are case-based questions. Each question has an internal choice and carries 4( 1 + 1 + 2 ) marks each.
Question 29.
Read the passage given below and answer the following questions:
The number of molecules of a certain component in a mixture divided by the total number of moles in the given mixture is represented by the mole fraction. It’s a means of expressing a solution’s concentration. The total mole fraction of all the components is always one. The mole fraction is a fraction of molecules, and because different molecules have varying masses, it differs from the mass fraction.
(A) What is the formula for the calculation of a mole fraction of component a, in a mixture of component a and b?
OR
Why molality is preferred over molarity of a solution?
(B) What is the mole fraction of methanol in a mixture of 5.8 g of methanol and 9.2 g of ethanol?
(C) Prove that the sum of all the mole fractions present in a mixture is one.
Answer:
(A) Xa = number of moles of component ‘a’/total number of component ‘a’ + ‘b’
Xa = na/na + nb
(a and b are subscript…refer formula given on page 16 under mole fraction of component heading
OR
Because molality of a solution does not change with temperature, while molarity of a solution changes with temperature.
(B) molar mass of CH3OH = 12 + 3 + 16 + 1 = 29
molar mass of C2H5OH = 24 + 5 + 16 + 1 = 46
number of moles of CH3OH = 5.8/29 =0.2
moles number of moles of C2H5OH = 9.2/46 =0.2
X CH3OH = moles of CH3OH/total moles CH3OH + C2H5OH
X CH3OH = 0.2/0.2 + 0.2
= 0.2/0.4
= 0.5
(C) Let us take the above question for justification mole fraction of CH3OH = 0.5
mole fraction of C2H5OH = moles of C2H5OH / total moles of CH3OH and C2H5OH = 0.2/0.2 + 0.2 mole fraction ethanol = 0.5
Total mole fraction of CH3OH and C2H5OH = 0.5 + 0.5 = 1
Hence,proved
Question 30.
Read the passage given below and answer the following questions:
When a molecule cannot be canonically represented by a single structure but can be described by two or more canonical structures, the actual structure is said to be a resonance hybrid of these structures. These characteristics include the position of the nuclei, bonding and non-bonding pairs of electrons, and the number of these pairs. A double-headed arrow is used to symbolise it. The molecules of 03, C03~2, and C02 are a few instances of resonance structures. Because the energy of the resonance hybrid is lower than the energy of any one of the single canonical forms, resonance stabilises the molecule.
(A) What is resonance energy?
(B) What is the role of resonance?
(C) (i) Why all resonating structures must have same number of valence electrons?
(ii) What happens when the two resonance structures are not equivalent?
OR
Give the resonating structures of carbon dioxide.
Answer:
(A) It is the difference between the actual bond energy of the molecule and that of the most stable resonating structures (having least energy). OR Resonance energy = Actual bond energy – Energy of the most stable resonating structure.
(B) Resonance stabilizes the molecule because the
energy of the resonance hybrid is less than the energy of any single canonical structures. Resonance also averages the bond characteristics as a whole.
(C) (i) The number of valence electrons in each resonating structure must be the same as the creation or destruction of electrons is impossible. The number of electrons in the new structures must match that of the original.
(ii) When two resonance structures are not equal, the structure with the greatest number of atoms with no formal charge is the one that most closely resembles the real molecule.
OR
The resonating structures of carbon dioxide (CO2) are as follows:
![]()
Section-E
The following questions are long answer type and carry 5 marks each. All questions have an internal choice.
Question 31.
Attempt any five questions:
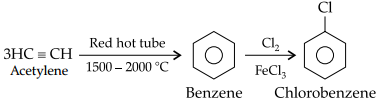
(A) How will you convert:
Acetylene to chlorobenzene
(B) How will you demonstrate that double bonds of benzene are somewhat different from that of olefins?
(C) What is benzenoid and non-benzenoid?
(D) How many isomers are possible for monosubstituted and disubstituted benzene?
(E) What is the use of BHC (benzene hexachloride)?
(F) Convert Benzene to Toulene.
(G) Write IUPAC names of the products obtained by the ozonolysis of Pent-2-ene
Answer:
(B) The double bonds of olefins decolorise bromine water and discharge the pink colour of Bayer’s reagent while those of benzene does not.
(C) Aromatic compounds containing benzene ring are known as benzenoids and those not containing a benzene ring are known as non- benzenoids.
(D) There is one mono substituted benzene and three di substituted benzene, i.e., ortho, para and meta.
(E) It is used as an insecticide under the name of Gammaxene or lindane.
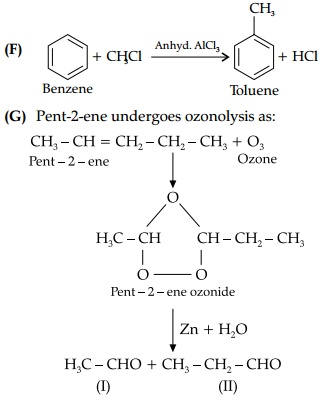
The IUPAC name of product (I) is ethanal and product (II) is propanal.
Question 32.
(A) Calculate the total number of electrons present in one mole of methane.
(B) Find:
(i) the total number and
(ii) the total mass of neutrons in 7 mg of 14C.
(Assume that mass of a neutron = 1.675 × 10-27 kg)
(C) Find:
(i) the total number and
(ii) the total mass of protons in 34 mg of NH3 at STP
(iii) Will the answer change if the temperature and pressure are changed?
OR
(A) The work function for caesium atom is 1.9 eV Calculate (i) the threshold wavelength and (ii) the threshold frequency of the radiation. If the caesium element is irradiated with a wavelength 500 nm, calculate the kinetic energy and the velocity of the ejected photoelectron.
(B) Lifetimes of the molecules in the excited states are often measured by using pulsed radiation source of duration nearly in the nano second range. If the radiation source has the duration of 2 ns and the number of photons emitted during the pulse source is 2.5 × 10-15, calculate the energy of the source.
Answer:
(A) (i) Number of molecules in 1 mole of CH4 = 6.022 × 1023
Number of electrons in one molecule of CH4 = 6 + 4 = 10
∴ Total number of electrons in 6.022 × 1023 molecules of CH4
= 6.022 × 1023 × 10 = 6.022 × 1024 electrons
(ii) Gram atomic mass of carbon (C – 14) = 14 g
= 14 × 103 mg
14 × 103 mg of carbon (C – 14) have
= 6.022 × 103 atoms
∴ 7 mg of carbon (C – 14) have
= \(\frac{6.022 \times 10^{23}}{14 \times 10^3}\) × 7
= 3.011 × 1020 atoms
Number of neutrons present in one atom of carbon (C – 14) = 14 – 6 = 8
∴ Total number of neutrons present in
3.011 × 1020 atoms of carbon (C-14)
= 3.011 × 1020 × 8
= 2.408 × 1021 neutrons
(B) ∴ Mass of one neutron = 1.675 × 10-27 kg
∴ Total mass of 2.408 × 1021 neutrons
= 1.675 × 10-27 × 2.408 × 1021
= 4.033 × 10-6 kg
(C) (i) Number of protons present in one molecule
of NH3 = 7 + 3 = 10
∴ Total number of protons present in 12.044 × 1020 molecules of NH3
= 12.044 × 1020 × 10 = 1.2044 × 1022 protons
(ii) Gram molecular mass of ammonia (NH3)
= 14 + 3 = 17g = 17 × 103 mg
∴ 17 × 103 mg of NH3 have
= 6.022 × 1023 molecules
∴ 34 mg of NH3 have = \(\frac{6.022 \times 10^{23} \times 34}{17 \times 10^3}\)
= 12.044 × 1020molecules
∵ Mass of one proton = 1.67 × 10-27Kg
Mass of 1.2044 × 1022 proton
= 1.67 × 10-27 × 1.2044 × 1022
= 2.01 × 10-5kg
(iii) No, the answer will not change upon changing the temperature and pressure because only the number of protons and mass of protons are involved.
OR
(A) Given, Work function (W0) = 1.9 eV
= 1.9 × 1.602 × 10-19J
(i) Threshold wavelength (λ0) = \(\frac{\mathrm{C}}{v}\)
= \(\frac{3 \times 10^8 \mathrm{~m} \mathrm{~s}^{-1}}{4.59 \times 10^{14} \mathrm{~s}^{-1}}\)
= 0.6536 × 10-6m
= 653.6 × 10-9m
= 653.6 nm
≅ 654 nm
(ii) Threshold frequency (v) = \(\frac{\mathrm{W}_0}{h}\)
= \(\frac{1.9 \times 1.602 \times 10^{-19} \mathrm{~J}}{6.626 \times 10^{-34} \mathrm{Js}}\)
= 0.459 × 1015 s-1
= 4.59 × 1014 s-1
Kinetic energy of photo electron = h(v – V0)
∵ λ = 500 nm = 500 × 10-9 m
∴ v = \(\frac{C}{\lambda}=\frac{3.0 \times 10^8 \mathrm{~ms}^{-1}}{500 \times 10^{-9} \mathrm{~m}}\)
= 6.0 × 1014 s-1
K.E. = 6.626 × 10-34 Js (6.0 × 1014 s-1 – 4.59 × 1014 s-1
= 9.34 × 10-20 J
∵ K.E. = \(\frac{1}{2}\) mv2
Mass of electron (m) = 9.1 × 10-31 kg
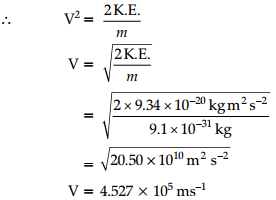
(B) Duration of the radiation source = 2 ns
∴ Period = 2 × 10-9s
Frequency (v) = \(\frac{1}{\text { Period }}=\frac{1}{2 \times 10^{-9}} s^{-1}\)
= 0.5 × 109 s-1
No. of photons emitted = 2.5 × 1015
∴ Energy of the source = nhv
= 2.5 × 1015 × 6.626 × 10-34 Js × 0.5 × 109 s-1
= 8.28 × 10-10J
Question 33.
The pH of milk, black coffee, tomato juice, lemon juice and egg white are 6.8, 5.0, 4.2, 2.2 and 7.8 respectively. Calculate corresponding hydrogen ion concentration in each.
OR
(A) The ionisation constant of nitrous acid is 4.5 × 10-4 Calculate
(i) the pH of 0.04 M sodium nitrite solution
(ii) The degree of hydrolysis
(B) Calculate the pH of the resultant mixtures:
(i) 10 mL of 0.2 M Ca(OH)2 + 25 mL of 0.1 M HC1
(ii) 10 mL of 0.01 M H2 SO4 + 10 mL of 0.01 M Ca(OH)2
(iii) 10 mL of 0.1 M H2 SO4 + 10 mL of 0.1 M KOH
Answer:
For milk
pH = 6.8
∵ pH = – log[H+]
6.8 = – log [H+]
or log [H+] = – 6.8 = \(\overline{7} .20\)
[H+] = antilog \(\overline{7} .20\)
= 1.585 × 10-7 M …….(1)
For black coffee –
pH = 5.0
∵ pH = – log [H+]
5.0 = -log [H+]
or log [H+] = – 5.0
[H+] = 10-5 M
For tomato juice –
pH = 4.2
∵ pH = – log [H+]
4.2 = – log [H+]
or log [H+] = – 4.2 = \(\overline{5} .80\)
[H+] = antilog \(\overline{5} .80\)
= 6.310 × 10-5
For lemon juice –
pH = 2.2
∵ pH = – log [H+]
2.2 = – log [H+]
or log [H+] = – 2.2 = \(\overline{3} .80\)
[H+] = antilog 3.80
= 6.310 × 10-3 M
For egg white –
pH = 7.8
∵ pH = – log [H+]
7.8 = – log [H+]
or log [H+] = – 7.8 = \(\overline{8} .20\)
[H+] = antilog 8.20
= 1.585 × 10-8 M
OR
(A) (i) Sodium nitrite is a salt of weak acid and strong base. Hence,
Kh = Kw /Ka
= 10-10/(4.5 × 10-4)
= 2.22 × 10-11

(B) (i) Base, Ca(OH)2 = Acid, HCl
M1V1 = M2V2 = 0.4M
Give,M1 = [OH–] = 2 × 0.2 = 0.4 M
v1 = 10 ml, M2 = 0.1 M
v2 = 25 ml
M1V1 = M2V2
0.4 × 10 = 0.1 × 25
M1V1 > M2V2 hence, solution is basic.
[OH–] = \(\frac{\mathrm{M}_1 \mathrm{~V}_1-\mathrm{M}_2 \mathrm{~V}_2}{\mathrm{~V}_1+\mathrm{V}_2}\) = \(\frac{4-2.5}{10+25}\)
= 0.043 M
pOH = – log [4.3 × 10-2]
= 2-0.6335 = 1.3665
pH = 14 – pOH = 14 – 1.3665
= 12.6335 = 12.63
(ii) Similarly, base, Ca(OH)2 = Acid, H2SO4
M1V1 = M2V2
(2 × 0.01) × 10 = (2 × 0.01) × 10
Because M1V1 = M2V2 hence solution is neutral.
∴ pH = 7
(iii) Acid, H2SO4 = Base, KOH
M1V1 = M2V2
(2 × 0.1)10 = 0.1 × 10
Because M1V1 > M2V2 , hence solution is acidic.
[H+] = \(\frac{M_1 V_1-M_2 V_2}{V_1+V_2}\)
\(\frac{(0.2 \times 10)-(0.1 \times 10)}{10+10}\) = \(\frac{2-1}{20}\) = 0.05 M
pH = – log [0.05] = – log[5.0 × 10-2]
pH = 2-0.6990 = 1.301 = 1.30
pH = 1.30