Experts have designed these Class 7 Science Notes Chapter 4 The World of Metals and Non metals Class 7 Notes for effective learning.
Class 7 Science Chapter 4 The World of Metals and Non metals Notes
Class 7 The World of Metals and Non metals Notes
Class 7 Science Chapter 4 Notes – The World of Metals and Non metals Notes Class 7
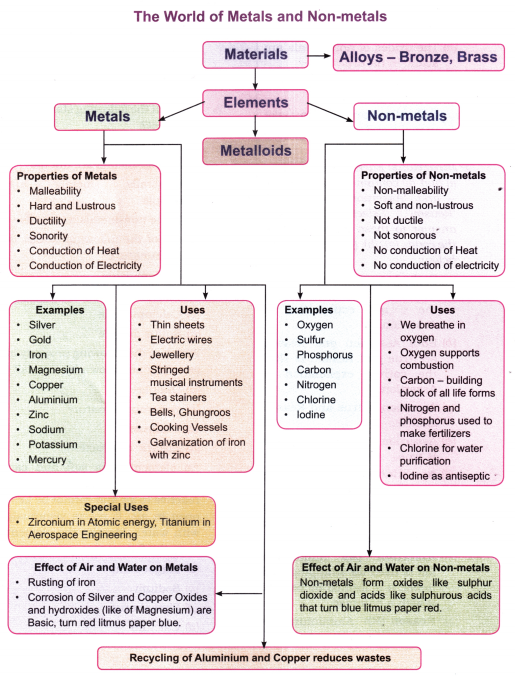
Properties Of Materials
Malleability
- Most metals e.g. copper, aluminium and iron are malleable. They can be beaten or hammered into very thin sheets. This property of metals is called malleability.
- Aluminium foil used for wrapping food and silver leaf used in sweets are familiar examples that show the extreme malleability of these metals.
- A piece of coal or a lump of sulfur breaks into powder on hammering. Both substances are non-metals; they are brittle and cannot be beaten into sheets.
- A block of wood is neither malleable nor brittle.
Hard & Lustrous:

- Objects made from copper, aluminium and iron are lustrous in appearance and are hard.
- Metals possess a metallic lustre (shine).
- Most of the metals are hard.
- Some metals like sodium and potassium are very soft and can be easily cut with a knife. Mercury is the only metal which is found in the liquid state at room temperature.
- Coal, Sulfur are non-lustrous and not as hard as metals. Wood is also not lustrous and hard.
Ductility
- Metals are also ductile, i.e. they can be drawn out into fine wires.
- This property of materials by which these can be drawn into wires is called ductility.
- Wires of copper and aluminium are widely used in electrical fittings because of their ductility (and good electrical conductivity).
- Jewellery (bangles, earrings, necklaces, etc.) is made from fine metal wires, usually of gold or silver.
- Stringed musical instruments such as the veena, sitar, violin and guitar all have metal strings.
- A tea-strainer is woven from metal wire.
- Gold and silver are the most ductile metals; one gram of gold can be drawn into a wire about two kilometers long.
- Coal and Sulfur cannot be drawn into wires and are therefore non-ductile. They are non-metals.
- Ropes made of steel (an alloy of iron and the non-metal carbon) are used in suspension bridges and cranes for lifting heavy loads.
![]()
Sonority
- When struck, a metal spoon or a metal coin produces a pleasant ringing sound. The ability of metals to produce such a sound is called sonority; metals are therefore said to be sonorous.
- The ringing of the school bell and the sound of ghungroos are examples of sonority.
- Coal does not produce ringing sound as it is a non-metal.
- A block of wood is neither a metal nor a non-metal; it produces a dull sound.
Conduction Of Heat
Metals are good conductors of heat; that is why most cooking vessels are made of metal. There is transfer of heat from one point to another of a material. This is called conduction and materials that transfer heat are called conductors.

- Wood is a poor conductor of heat.
- Handles of metal vessels used for cooking are made with wood or other materials which do not conduct heat.
- Silver is the best conductor of heat and lead is the poorest conductor of heat among the metals.
- Copper and aluminium are both excellent conductors of heat and are very suitable for making cooking utensils.
Conduction Of Electricity
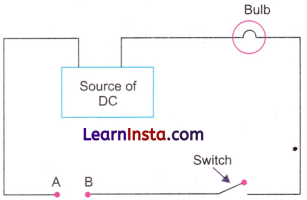
- Materials that allow electricity to flow through them easily are called good conductors of electricity.
- Materials that do not allow electricity to flow through them are called poor conductors of electricity or the insulators.
- Metals are good conductors of electricity. They have electric conductivity. They offer little resistance to the flow of electric current.
- Materials like rubber, glass, porcelain are poor conductors (insulators).
- You have seen in activities in Chapter 3, that the objects made of aluminium, copper and iron connected in a circuit make the lamp glow. These are good conductors of electricity.
- Silver and copper are the best electric conductors but copper and aluminium are used in electrical wiring because they are less costly and are more abundant.
- Sulfur, coal, wood, stone, eraser and nylon rope when used in a circuit, could not make the lamp to glow and are poor conductors of electricity.
Effect Of Air And Water On Metals (Iron) — Rusting
- When iron objects are exposed to moist air (air + water vapour) they gradually develop a reddish- brown coating called rust.
- The chemical process that produces rust is known as rusting.
- Rusting weakens iron structures and is a serious problem in construction.
- Silver left in the open air becomes coated with a black layer of silver sulphide.
- Green coating appears on copper objects when kept in open atmosphere.
- The slow deterioration of metal surfaces due to the action of air, water or other substances is broadly termed as corrosion.
- Rusting of iron can be prevented by
- painting,
- oiling,
- greasing, or
- applying a protective layer of zinc on iron (galvanisation).
- The Iron Pillar of Delhi was made 1600 years ago. It is 8 meters high and weighs more than 6000 kilograms. It has been made by unique skillful metal technology in India. It resists rusting despite so many years of rainfall, winds and extreme weather conditions.
![]()
Effect Of Air & Water On Other Metals & Their Oxides
- Almost all metals burn in oxygen. For example, a strip of magnesium burns with a dazzling white flame and forms magnesium oxide, a white powder.
- The oxide of magnesium turns moist red litmus paper blue, showing the basic character of metal oxides.
- Most metal oxides are insoluble in water.
- Sodium is so reactive that it is stored under kerosene; it reacts violently with oxygen and water, giving off heat and sometimes flames.
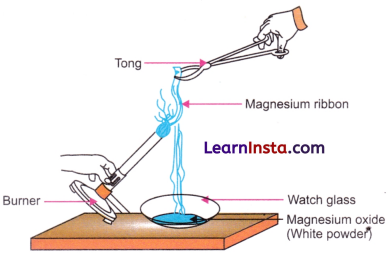
Fig. Burning Magnesium ribbon
Substances That Behave Differently From Metals
- Sulfur is anon-metal. When sulfur burns in oxygen it forms sulfur dioxide gas, which dissolves in water to give sulfurous acid.
- Phosphorus also behaves differently; it is stored under water because it catches fire on exposure to air.
- Such substances are usually soft and dull in appearance. They are neither malleable nor ductile. They are non-sonorous and poor conductors of heat and electricity.
- The oxides of non-metals are generally acidic. Aqueous solutions of these oxides turn blue litmus red.
Elements
- An element is a substance that cannot be broken down into simpler substances. All elements are classified into metals and non-metals.
- Presently 118 elements are known.
The World Of Metals And Non-Metals
- Elements are basic building blocks of all matter. Some are naturally occurring while others are artificially made in the laboratory.
- Materials like glass, wood, plastic, rubber and paper are not elements (metals or non-metals).
Metals are used in our daily life because of their distinct properties. Non-metals are also important to us.
Uses Of Non-Metals In Our Daily Life
- We breathe in oxygen. We cannot survive without oxygen. It also supports combustion.
- Carbon (another non-metal) is the building block of all life forms.
It is a key component of proteins, fats, and carbohydrates, which are necessary for growth and energy. - Nitrogen is essential nutrient for the growth of plants. It is used in the manufacture of fertilizers and chemicals.
- Chlorine is commonly used in water purification.
- Iodine solution is applied on wounds as an antiseptic.
Science And Society
- Numerous metals and alloys (mixtures of metals with other elements) are used to make utensils, tools and machinery.
- Certain special metals are vital for
- atomic energy (e.gvzirconium),
- aerospace engineering (e.g. titanium).
- Recycling of common metals such as iron and aluminium helps reduce waste and conserves resources for future generations.
→ Element: A pure substance which cannot be broken down into simpler substances.
→ Malleability: The property of metals that they can be beaten or hammered into very thin sheet is called malleability.
→ Metals: The elements possessing characteristic properties such as lustre, malleability, ductility, thermal and electrical conductivity, high tensile strength, etc.
→ Non-metals: The elements which are brittle, non-lustrous and bad conductors of heat and electricity.
→ Ductility: Property by which metals can be drawn out into fine wires.
→ Sonority: When struck, a metal spoon or a metal coin produces a pleasant ringing sound. The ability of metals to produce such a sound is called sonority.
![]()
→ Conduction: Property of materials to allow transfer of heat or flow of electric current.
→ Rust: When iron objects are exposed to moist air (air + water vapour) they gradually develop a reddish-brown coating called rust.