Students can use Curiosity Class 8 Science Book Solutions Chapter 9 The Amazing World of Solutes, Solvents, and Solutions Class 8 Question and Answer as a quick reference guide.
Class 8 Science Chapter 9 The Amazing World of Solutes, Solvents, and Solutions Question Answer
Science Class 8 Chapter 9 Question Answer The Amazing World of Solutes, Solvents, and Solutions
The Amazing World of Solutes, Solvents, and Solutions Class 8 Question Answer (InText)
Question 1.
Probe and Ponder
- What do you think is happening in the picture (Refer NCERT page 134)?
- What happens when you add too much sugar to your tea and it stops dissolving? How can you solve this problem?
- Why do sugar and salt dissolve in water but not in oil? Why is water considered a good solvent?
- Why are water bottles usually tall and cylindrical in shape instead of spherical?
- Share your questions
…………………………………………………….. ?
Answer:
(Questions may vary)
- Why does sawdust float on water while sand sinks, even though both are solids?
- What do you think will happen if you keep adding sugar to water but do not stir it?
![]()
Question 2.
We know air is a mixture. Would a mixture of gases also be considered a solution? (Page 135)
Answer:
Yes, a mixture of gases like air is considered a solution. This is because the gases are evenly distributed and form a uniform mixture. In air, nitrogen is present in the largest amount and acts as the solvent, while other gases like oxygen, carbon dioxide, and argon are the solutes as they are present in smaller amount.
Question 3.
What will happen if we keep on adding more salt in a given amount of water? (Page 136)
Answer:
Salt dissolves in water to form a solution. If we keep adding more salt, a point comes when it stops dissolving and settles at the bottom. This means the solution has become saturated, it cannot dissolve any more salt at that temperature.
Question 4.
Do gases also dissolve in water? (Page 139)
Answer:
Yes, some gases dissolve in water. For example, oxygen dissolves in water in small amounts. It forms a uniform solution because the gas mixes evenly with the water.
![]()
Question 5.
I observed that in some non-uniform mixtures, such as sawdust in water, the sawdust floats, whereas in the mixture of sand and water, the sand sinks. I wonder why that happens? (Page 139)
Answer:
In a mixture of sawdust and water, the sawdust floats because it is lighter (less dense) than water. On the other hand, in a mixture of sand and water, the sand sinks because it is heavier (more dense) than water.
Question 6.
Why are measuring cylinders always designed narrow and tall instead of wider and short like a beaker? (Page 144)
Answer:
Measuring cylinders are tall and narrow so that they can measure liquid volumes more accurately. The narrow shape allows clearer markings and helps detect even small changes in volume easily.
Question 7.
I wonder how the level of a coloured liquid is measured? (Page 145)
Answer:
The level of a coloured liquid is measured by looking at the markings on a measuring cylinder. We should read the level at the top of the curved surface (called the meniscus) of the liquid, keeping our eyes at the same level as the liquid.
NCERT Class 8 Science Chapter 9 Question Answer The Amazing World of Solutes, Solvents, and Solutions (Exercise)
Keep the curiosity alive(Pages 149-151)
Question 1.
State whether the statements given below are True [T] or False [F]. Correct the false statement(s).
(i) Oxygen gas is more soluble in hot water rather than in cold water.
(ii) A mixture of sand and water is a solution.
(iii) The amount of space occupied by any object is called its mass.
(iv) An unsaturated solution has more solute dissolved than a saturated solution.
(v) The mixture of different gases in the atmosphere is also a solution.
Answer:
(i) False. Oxygen gas is more soluble in cold water than in hot water.
(ii) False. A mixture of sand and water is a non-uniform mixture, not a solution.
(iii) False. The amount of space occupied by an object is called its volume.
(iv) False. An unsaturated solution has less solute dissolved than a saturated solution.
(v) True
![]()
Question 2.
Fill in the blanks.
(i) The volume of a solid can be measured by the method of displacement, where the solid is ……….. in water and the………. in water level is measured.
(ii) The maximum amount of ……….. dissolved in ……….. at a particular temperature is called solubility at that temperature.
(iii) The density ……….. with increase in temperature.
(iv) The solution in which glucose has completely dissolved in water, and no more glucose can dissolve at a given temperature, is called a ……….. solution of glucose.
Answer:
(i) immersed, rise
(ii) solute, solvent
(iii) decreases
(iv) saturated
Question 3.
You pour oil into a glass containing some water. The oil floats on top. What does this tell you?
(i) Oil is denser than water
(ii) Water is denser than oil
(iii) Oil and water have the same density
(iv) Oil dissolves in water
Answer:
(ii) Water is denser than oil
Question 4.
A stone sculpture weighs 225 g and has a volume of 90 cm3. Calculate its density and predict whether it will float or sink in water.
Answer:
Mass of a stone sculpture =225 g
Volume of a stone sculpture =90 cm3
Density =\(\frac{\text { Mass }}{\text { Volume }}=\frac{225 \mathrm{~g}}{90 \mathrm{~cm}^3}=2.5 \mathrm{~g} / \mathrm{cm}^3\)
Since the density of water is 1g/cm3, and the density of stone sculpture is greater than the density of water, the stone will sink in water.
![]()
Question 5.
Which one of the following is the most appropriate statement, and why are the other statements not appropriate?
(i) A saturated solution can still dissolve more solute at a given temperature.
(ii) An unsaturated solution has dissolved the maximum amount of solute possible at a given temperature.
(iii) No more solute can be dissolved into the saturated solution at that temperature.
(iv) A saturated solution forms only at high temperatures.
Answer:
(iii) Most appropriate – No more solute can be dissolved into the saturated solution at that temperature.
(i) Not appropriate – A saturated solution cannot dissolve more solute at the same temperature.
(ii) Not appropriate – An unsaturated solution can still dissolve more solute.
(iv) Not appropriate – Saturated solutions can form at any temperature, not only at high temperatures.
Question 6.
You have a bottle with a volume of 2 litres. You pour 500 mL of water into it. How much more water can the bottle hold?
Answer:
Bottle capacity =2 litres =2000 mL
Water added =500mL
Remaining capacity =2000-500
=1500 mL or 1.5 litres
Hence, the bottle can hold 1500 mL or 1.5 litres more water.
Question 7.
An object has a mass of 400 g and a volume of 40 cm3. What is its density?
Answer:
Mass of an object =400 g
Volume of an object =40 cm3
Density = \(\frac{\text { Mass }}{\text { Volume }}=\frac{400 \mathrm{~g}}{40 \mathrm{~cm}^3}=10 \mathrm{~g} / \mathrm{cm}^3\)
Question 8.
Analyse Fig. 9.25a and 9.25b. Why does the unpeeled orange float, while the peeled one sinks? Explain.
Answer:
The unpeeled orange floats because its peel has air pockets, making it less dense than water. On the other hand, the peeled orange sinks because removing the peel increases its overall density, making it more dense than water.
Question 9.
Object A has a mass of 200 g and a volume of 40 cm3. Object B has a mass of 240 g and a volume of 60 cm3. Which object is denser?
Answer:
Compare densities of Object A and B
Mass of object A=200g
Volume of object A=40 cm3
Density =\(\frac{\text { Mass }}{\text { Volume }}=\frac{200 \mathrm{~g}}{40 \mathrm{~cm}^3}=5 \mathrm{~g} / \mathrm{cm}^3\)
Now,
Mass of object B=240 g
Volume of object B=60 cm3
Density =\(\frac{\text { Mass }}{\text { Volume }}=\frac{240 \mathrm{~g}}{60 \mathrm{~cm}^3}=4 \mathrm{~g} / \mathrm{cm}^3\)
Hence, object A is denser than object B.
![]()
Question 10.
Reema has a piece of modeling clay that weighs 120g. She first moulds it into a compact cube that has a volume of 60 cm3. Later, she flattens it into a thin sheet. Predict what will happen to its density.
Answer:
When Reema flattens the clay, its shape changes but its mass and volume remain the same, so density remains the same.
Question 11.
A block of iron has a mass of 600 g and a density of 7.9g/ cm3 What is its volume?
Answer:
Mass =600 g
Density =7.9 g/cm3
Volume = \(\frac{\text { Mass }}{\text { Density }}=\frac{600 \mathrm{~g}}{7.9 \mathrm{~g} / \mathrm{cm}^3} \approx 75.95 \mathrm{~cm}^3\)
Hence, the volume of the iron block is approximately 76 cm3
Question 12.
You are provided with an experimental setup as shown in Fig. 9.26a and 9.26b. On keeping the test tube (Fig 9.26b) in a beaker containing hot water ( ∼ 70°C), the water level in the glass tube rises. How does it affect the density?
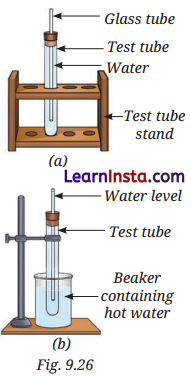
Answer:
When water is heated, its particles move apart, causing it to expand. The rising water level in the glass tube shows expansion. Since the mass remains the same and the volume increases, the density decreases. Hence, heating reduces the density of water due to expansion.
Class 8 The Amazing World of Solutes, Solvents, and Solutions Question Answer (Activities)
Activity 9.1: let us investigate (page 136)
Aim: To observe how much salt can dissolve in water and to understand the difference between saturated and unsaturated solutions.

Observation:
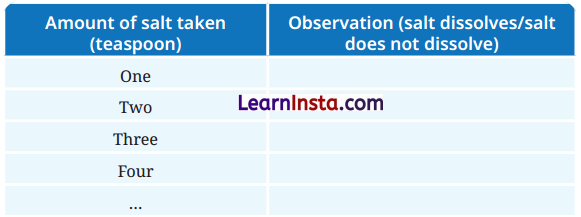
Answer:
| Amount of salt taken (teaspoon) | Observation (salt dissolves/salt does not dissolve) |
| One | Salt dissolves completely |
| Two | Salt dissolves completely |
| Three | Salt dissolves completely |
| Four | Salt dissolves completely |
| Five | Salt dissolves partially, some salt remains undissolved |
| Six | Salt does not dissolve, settles at the bottom |
Note:
The number of teaspoons may vary slightly depending on the size of the spoon, and the temperature of water.
- Initially, the salt dissolved completely in the water and formed a clear solution (up to around 4 teaspoons).
- When more salt was added, a point was reached where it stopped dissolving and began to settle at the bottom.
![]()
Conclusion: Water can dissolve only a limited amount of salt at a given temperature. When it dissolves all the added salt, the solution is unsaturated. When no more salt dissolves and some remains undissolved, it becomes a saturated solution. This activity also shows the difference between dilute and concentrated solutions based on the amount of solute dissolved.
Activity 9.2: Let Us Experiment (Demonstration Activity) (Pages 137-138)
Aim: To study how the solubility of a substance changes with temperature.

Observation: Water at 70°C dissolves more baking soda than water at 50°C. The amount of baking soda dissolved in water at 20°C is even lesser.
Conclusion:
- The solubility of baking soda in water increases with an increase in temperature.
- A solution that is saturated at a lower temperature can become unsaturated when the temperature is increased.
Activity 9.3: Let Us Measure (Page 142)
Aim: To measure the mass of an object using a digital weighing balance.

Observation: Do it yourself.
Conclusion: Mass of a substance can be measured by using a digital weighing balance.
![]()
Activity 9.4: Let Us Observe And Calculate (Pages 143-144)
Aim: To understand how to read the volume using a measuring cylinder and to calculate the smallest volume it can measure.
Observation and Conclusion:
- The maximum volume the measuring cylinder can measure is 100 mL
- The volume difference between two bigger marks (such as 10 mL and 20 mL ) is 10 mL
- There are 10 smaller divisions between two bigger marks.
- Each smaller division represents a volume of 1 mL
- Therefore, the smallest volume this measuring cylinder can measure accurately is 1 mL
Activity 9.5: Let Us Measure 50 Ml Of Water (Pages 144-145)
Aim: To accurately measure 50 mL of water using a measuring cylinder by reading the correct level of the meniscus.

Activity 9.5: Let Us Measure 50 Ml Of Water (Pages 144-145)
Aim: To accurately measure 50 mL of water using a measuring cylinder by reading the correct level of the meniscus.
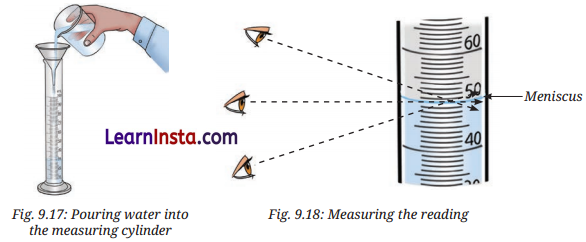
Observation: Do it yourself.
Conclusion:
- For measuring volume of water or colourless liquids, read the mark on the measuring cylinder that coincides with the bottom of the meniscus.
- For measuring volume of coloured liquids, read the mark on the measuring cylinder that coincides with the top of the meniscus.
![]()
Activity 9.6: Let Us Calculate (Pages 145)
Aim: To calculate the volume of cuboid-shaped objects using the formula:
Volume =length × width × height
Do it yourself.
Activity 9.7: let us measure (pages 146)
Aim: To measure the volume of irregular solids using the water displacement method.
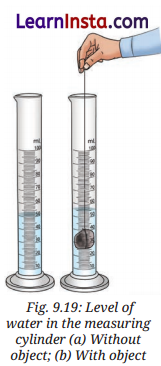
Observation:
| S. No. | Object | Initial volume of water in the measuring cylinder (mL) (A) | Final volume of water in the measuring cylinder (mL) (B) | Volume of water displaced in the measuring cylinder (mL) (B-A) | Volume of the object (cm3) |
| 1. | Stone | 50 mL | 55 mL | 5 mL | 5 cm3 |
| 2. | Metal key | 50 mL | 53 mL | 3 mL | 3 cm3 |
| 3. | Wooden block | 50 mL | 51 mL | 1 mL | 1 cm3 |
Conclusion: The volume of an irregular-shaped solid can be accurately measured using the water displacement method. Since 1 mL of water equals 1cm3, the volume displaced is equal to the object’s volume. Knowing the volume and mass of an object helps calculate its density.