Students can access the CBSE Sample Papers for Class 10 Science with Solutions and marking scheme Term 2 Set 1 will help students in understanding the difficulty level of the exam.
CBSE Sample Papers for Class 10 Science Term 2 Set 1 with Solutions
Time : 2 Hours
Max. Marks : 40
General Instructions :
- All questions are compulsory.
- The question paper has three sections and 15 questions. All questions are compulsory.
- Section-A has 7 questions of 2 marks each; Section-B has 6 questions of 3 marks each; and Section-C has 2 case based questions of 4 marks each.
- Internal choices have been provided in some questions. A student has to attempt only one of the alternatives in such questions.
Section – A
Question 1.
The table shows the electronic structures of four elements. (2)
| Element | Electronic Structure |
| P | 2,6 |
| Q | 2,8,1 |
| R | 2,8,7 |
| S | 2,8,8 |
(a) Identify which element(s) will form covalent bonds with carbon.
(b) “Carbon reacts with an element in the above table to form several compounds.” Give suitable reason.
Answer:
(a) P and R will form the covalent bond since a covalent bond is formed by the sharing of the electrons. The electronic configuration of P is 2, 6, and that of the R is 2, 8, 7. P and R are oxygen and chlorine atoms respectively. Chlorine and Oxygen both are non-metals having 7 and 6 electrons in their outer most orbit respectively. So the type of bond that they will form is covalent bond which is based on sharing of electrons. For example: Cl2O where each chlorine atom contributes one electron while the oxygen atom contributes two electrons thus each of them individually satisfying the criteria to have 8 electrons in the outermost orbit.
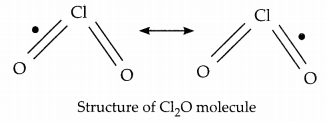
(b) Carbon will react with element P to form several compounds due to the tetravalency of carbon atom. Tetravalency is the state of an atom in which there are four electrons available with the atom for covalent chemical bonding. It also forms long chain of compounds due to the inherent property of the catenation. The property of self-linking of elements to form a long chain is known as catenation. Thus, due to the properties of catenation and tetravalency carbon will form several compounds.
Question 2.
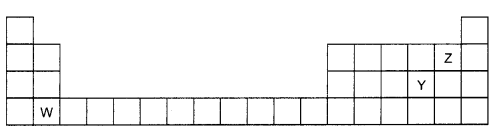
The diagram below shows part of the periodic table. (2)
(a) Which elements would react together to form covalent compounds?
(b) Between the two elements W and Z, which will have a bigger atomic radius? Why?
Answer:
(a) Y and Z will form the covalent compounds by the sharing of the electrons since Y belongs to group 6 and Z belongs to group 7, and they both are non-metals having 6 and 7 electrons in their outer most orbit respectively. So the type of bond that they will form is Covalent Bond which is based on sharing of electrons.
(b) Out of the two elements W and Z, W will have bigger atomic radius since as we go down a group the number of shells increases and valence electrons are present in higher shell and the distance of valence electrons from nucleus increases. Thus the atomic radii increases in a group from top to bottom while in a period, atomic radius generally decreases from left to right. In a period there is a gradual increase in the nuclear charge. Since valence electrons are added in the same shell, they are more and more strongly attracted towards nucleus. This gradually decreases atomic radii.
Question 3.
(a) Trace the path a male gamete takes to fertilise a female gamete after being released from the penis. (2)
(b) State the number of sets of chromosomes present in a zygote.
Answer:
(a) Male gamete (sperm) travels in the female reproductive tract after being released. The path which it takes to fertilise the female gamete (egg) after being released from the penis is:
Vagina → Uterus → Fallopian tube → Zygote
(b) Zygote has 2 sets of chromosomes. A zygote is formed from the fusion of a male gamete with a female gamete. Thus, it is diploid and contains two sets of chromosomes, one of which is paternal and the other is maternal i.e. from male and female parent respectively.
Question 4.
Rajesh observed a patch of greenish black powdery mass on a stale piece of bread. (2)
(a) Name the organism responsible for this and its specific mode of asexual reproduction.
(b) Name its vegetative and reproductive parts.
Answer:
(a) The greenish black powdery mass on a stale piece of bread is due to bread mould Rhizopus which reproduces by spore formation.
(b) Hyphae or thread like structures are the vegetative part and tiny blob like structures or sporangia are the reproductive parts of the bread mould Rhizopus.
Question 5.
Mustard was growing in two fields- A and B. While Field A produced brown coloured seeds, field B produced yellow coloured seeds. (2)
It was observed that in field A, the offsprings showed only the parental trait for consecutive generations,
whereas in field B, majority of the offsprings showed a variation in the progeny.
What are the probable reasons for these?
OR
In an asexually reproducing species, if a trait X exists in 5% of a population and trait Y exists in 70% of the same population, which of the two trait is likely to have arisen earlier? Give reason.
Answer:
In field A, the reason for parental trait in consecutive generations of the offsprings is self-pollination while in field B, variations occur because of recombination of genes as cross – pollination is taking place.
OR
Trait Y which exists in 70% (larger fraction) of the population, is likely to have arisen earlier because in asexual reproduction, identical copies of DNA are produced and variations do not occur. New traits come in the population due to sudden mutation and then are inherited. 70 % of the population with trait Y is likely to have been replicating that trait for a longer period than 5 % of population with trait X.
Question 6.
A simple motor is made in a school laboratory. A coil of wire is mounted on an axle between the poles of a horseshoe magnet, as illustrated. (2)
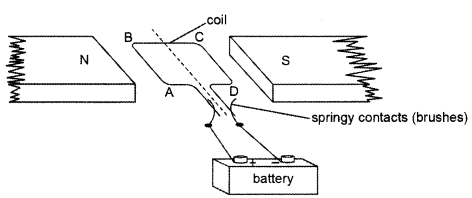
In the example above, coil ABCD is horizontal and the battery is connected as shown.
(a) For this position, state the direction of the force on the arm AB.
(b) Why does the current in the arm BC not contribute to the turning force on the coil?
OR
A circuit contains a battery, a variable resistor and a solenoid. The figure below shows the magnetic field pattern produced by the current in the solenoid.
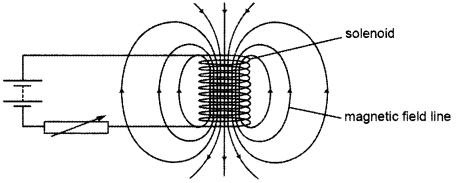
(a) State how the magnetic field pattern indicates regions where the magnetic field is stronger.
(b) What happens to the magnetic field when the current in the circuit is reversed?
Answer:
(a) The direction of the force on the arm AB will be downwards.
(b) The current in the arm BC not contribute to the turning force on the coil because BC is in the same direction as the direction of field lines. Force is minimum when the direction of current in the conductor is the same as that of the magnetic field. BC will not contribute as the force on this part of the coil will be cancelled by the force on DA.
OR
(a) Relative closeness of field lines indicates the strength of magnetic field. Since field lines are crowded around the ends of the solenoid, hence these are the regions of strongest magnetism.
(b) The direction of the field will also be reversed when the current in the circuit is reversed.
Question 7.
DDT was sprayed in a lake to regulate breeding of mosquitoes. How would it affect the trophic levels in the following food chain associated with a lake? Justify your answer. (2)
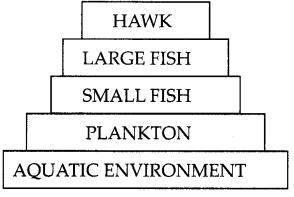
OR
In the following food chain, vertical arrows indicate the energy lost to the environment and horizontal arrows indicate energy transferred to the next trophic level. Which one of the three vertical arrows (A, C and E) and which one of the two horizontal arrows (B and D) will represent more energy transfer? Give reason for your answer.
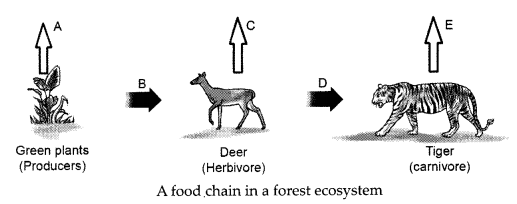
A food chain in a forest ecosystem
Answer:
DDT being a non- biodegradable pesticide will enter the food chain from the first trophic level i.e Plankton. Non – biodegradable pesticides accumulate progressively at each trophic level. This phenomenon is known as biological magnification. Hawk will have the highest level of pesticide.
OR
In accordance with 10% law of transfer of energy in a food chain only 10% of energy available at one trophic level is transferred to the next trophic level. A will represent more energy transfer as compared to C and E. B will represent more energy transfer as compared to D. When green plants are eaten by primary consumers, a great deal of energy is lost as heat to the environment, some amount goes into digestion and in doing work and the rest goes towards growth and reproduction. An average of 10% of the food eaten is made available for the next level of consumers. This loss of energy takes place at every trophic level.
Section – B
Question 8.
Choose an element from period 3 of modern periodic table that matches the description given below in each instance. Give reason for your choice. (3)
(a) It has a similar structure to diamond.
(b) It has same valency as Lithium.
(c) It has variable valency and is a member, of the Oxygen family (group 16).
Answer:
(a) Silicon has the similar structure to diamond because it has tetravalency and shows the property of catenation similar to the carbon atom also it has similar tetrahedral structure as that of the diamond.
(b) Sodium has the same valency as that of the lithium since it has only 1 valence electron like that of the lithium.
(c) Sulphur has variable valency and is a member of the Oxygen family (group 16) and it forms oxides such as SO2 and SO3.
Question 9.
(a) How many isomers are possible for the compound with the molecular formula C4H8? Draw the electron dot structure of branched chain isomer. (3)
(b) How will you prove that C4H8 and C4H10 are homologues?
OR
A carbon compound A’ having melting point 156K and boiling point 351K, with molecular formula C2H6O is soluble in water in all proportions.
(a) Identify ‘A’ and draw its electron dot structure.
(b) Give the molecular formulae of any two homologues of ‘A’.
Answer:
(a) Four isomers are possible for the compound with the molecular formula C4H8. The electron dot structure of branched chain isomer is:
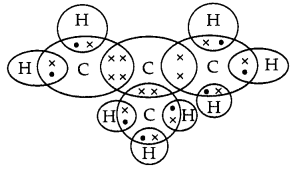
(b) A homologous series is a group of organic chemical compounds, usually listed in order of increasing size, that have a similar structure (and hence also have similar properties) and whose structures differ only by the number of CH2 units in the main carbon chain. C4H8 and C5H10 are homologues as they differ by CH2 unit and the difference in their molecular mass is of 14 u, also they have same general formula along with the same functional group.
OR
(a) A carbon compound having melting point 156K and boiling point 351K, with molecular formula C2H6O and is soluble in water in all proportions is Ethanol (C2H5OH).
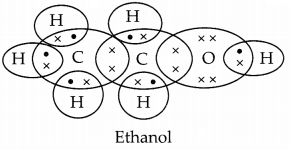
(b) The molecular formula of the two homologous of A [Ethanol (C2H5OH)] are methanol (CH3OH) and propanal (C3H7OH) or CH4O and C3H8O.
Question 10.
Two pea plants – one with round yellow seeds (RRYY) and another with wrinkled green (rryy) seeds produce F1 progeny that have round, yellow (RrYy) seeds. (3)
When F1 plants are self-pollinated, which new combination of characters is expected in F2 progeny? How many seeds with these new combinations of characters will be produced when a total 160 seeds are produced in F2 generation? Explain with reason.
Answer:
According to Mendel’s law of independent inheritance and law of Dihybrid inheritance, if two pairs of contrasting characteristics were cross-bred by self-pollination. In F1 generation, plants with dominant traits are produced which are round in shape and are yellow in colour. While in F generation, different combinations of shape and colour are obtained in the ratio 9:3:3:1.
| Trait | Ratio | Number of Seeds |
| Round Yellow | 9 | 9/16 x 160 = 90 |
| Round Green | 3 | 3/16 x 160 = 30 |
| Wrinkled Yellow | 3 | 3/16 x 160 = 30 |
| Wrinkled Green | 1 | 1/16 x 160 = 10 |
| Total | 16 | 160 |
Thus, the new combination of the characters obtained are:
Round green – 30 and Wrinkled yellow – 30.
Question 11.
(a) It would cost a man ₹ 3.50 to buy 1.0 kW h of electrical energy from the Main Electricity Board. His generator has a maximum power of 2.0 kW. The generator produces energy at this maximum power for 3 hours. Calculate how much it would cost to buy the same amount of energy from the Main Electricity Board.
(b) A student boils water in an electric kettle for 20 minutes. Using the same mains supply he wants to reduce the boiling time of water. To do so should he increase or decrease the length of the heating element? Justify your answer. [1 + 2 = 3]
Answer:
(a) E = P x T
So, E = 3 x 2 = 6 kWh
(Cost of buying electricity from the main electricity board = 6 x 3.50 = ₹ 21.0
(b) To reduce the boiling time using the same mains supply, the rate of heat production should be large. We know that P= V²/R. Since V is constant, R should be decreased. Since R is directly proportional to 1 so length should be decreased.
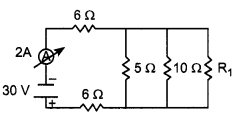
In the above circuit, if the current reading in the ammeter A is 2A, what would be the value of R1?
OR
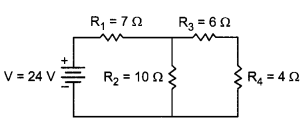
Calculate the total resistance of the circuit and find the total current in the circuit.
Answer:
5 Ohm, 10 Ohm and Rj are in parallel

R3 and R4 are in series, hence the equivalent resistance of those two = R5 = R3 + R4 = 10 Ω.
R5 and R2 are in parallel. Let R6 be the equivalent resistance for them. Hence R6 = \(\frac{\left(\mathrm{R}_{5} \times \mathrm{R}_{2}\right)}{\left(\mathrm{R}_{5}+\mathrm{R}_{2}\right)}=\frac{100}{20}\)Ω = 10 Ω.
Now R4 are R6 are in series and hence the final equivalent resistance of the entire circuit is R = R1 + R6 = 12 Ω.
By Ohm’s Law, we know that V = IR, hence I = V/R. Hence the current in the circuit is 24/12 = 2A.
Question 13.
Gas A, found in the upper layers of the atmosphere, is a deadly poison but is essential for all living beings. The amount of this gas started declining sharply in the 1980s. (3)
(a) Identify Gas A. How is it formed at higher levels of the atmosphere?
(b) Why is it essential for all living beings? State the cause for the depletion of this gas.
Answer:
(a) Gas A is Ozone (O3). Ozone at the higher levels of the atmosphere is a product of UV radiation acting on oxygen (O2) molecule. The higher energy UV radiations split apart some molecular oxygen (O2) into free oxygen (O) atoms. These atoms then combine with molecular oxygen to form ozone.
\(\mathrm{O}_{2} \stackrel{\mathrm{UV}}{\longrightarrow} \mathrm{O}+\mathrm{O}\)
O + O2 → O3
(b) Ozone is essential for the living beings because it shields the surface of the earth and protects the living organisms from ultraviolet (UV) radiation released by the sun. Chlorofluorocarbons (CFCs) which are used as refrigerants or in fire extinguishers lead to depletion of ozone layer.
Section – C
This section has 02 case-based questions (14 and 15). Each case is followed by o3 sub-questions (a, b and c). Parts a and b are compulsory. However, an internal choice has been provided in part c.
Question 14.
Sahil performed an experiment to study the inheritance pattern of genes. He crossed tall pea plants (TT) with short pea plants (tt) and obtained all tall plants in F4 generation. (4)
(a) What will be set of genes present in the F4 generation? (1)
(b) Give reason why only tall plants are observed in F4 progeny. (1)
(c) When F4 plants were self-pollinated, a total of 800 plants were produced. How many of these would be tall, medium height or short plants? Give the genotype of F2 generation. (2)
OR
When F4 plants were cross-pollinated with plants having tt genes, a total of 800 plants were produced. How many of these would be tall, medium height or short plants? Give the genotype of F2 generation.
Answer:
(a) Dominant trait is tallness which is represented by TT and recessive trait is dwarfness which is represented by tt. Thus, on crossing dominant trait with dwarf trait, Tt will be obtained. Hence, Tt will be the set of genes present in the F1 Generation.
(b) According to Mendel’s law of monohybrid inheritance and law of segregation, if single pair of contrasting characteristics were cross-bred by self-pollination, than, in F1 progeny, plants with dominant traits are produced. Thus only tall plants are observed in the F1 Progeny because T’ are called dominant traits and they expresses themselves.
(c) Out of 800 plants 600 plants will be tall and 200 plants will be short plants. Thus in F2 generation different traits are obtained in the ratio 3:1.
OR
In the cross between Tt x tt, 400 Tall (Tt) and 400 short (tt) plants will be produced. 1Tt : 1tt will be the genotype of the F2 generation.
Question 15.
Ansari sir was demonstrating an experiment in his class with the setup as shown in the figure below:
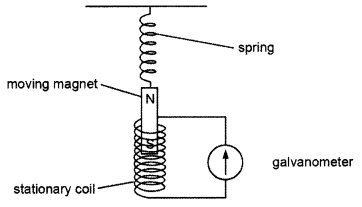
A magnet is attached to a spring. The magnet can go in and out of the stationary coil.
He lifted the magnet and released it to make it oscillate through the coil. Based on your understanding of the phenomenon, answer the following questions:
(a) What is the principle which Ansari Sir is trying to demonstrate?
(b) What will be observed when the magnet starts oscillating through the coil. Explain the reason behind this observation.
(c) Consider the situation where the magnet goes in and out of the coil. State two changes which could be made to increase the deflection in the galvanometer.
OR
Is there any difference in the observations in the galvanometer when the magnet swings in and then out of the stationary coil? Justify your answer.
Answer:
(a) Ansari sir is trying to demonstrate the principle of Electromagnetic induction.
(b) When the magnet starts oscillating through the coil there will be induced current in the coil due to relative motion between the magnet and the coil. Changing the magnetic field around the coil generates induced current.
(c) Two changes which could be made to increase the deflection in the galvanometer are by using a stronger magnet and using a coil with more number of turns.
OR
When the magnet moves into the coil, the ammeter shows a momentary deflection towards one side say left. When the magnet moves out of the coil, the ammeter shows a momentary deflection now towards right. This is due to changing magnetic field or flux associated with the coil as the magnet moves in and out.