Students can access the CBSE Sample Papers for Class 10 Science with Solutions and marking scheme Set 2 will help students in understanding the difficulty level of the exam.
CBSE Sample Papers for Class 10 Science Set 2 with Solutions
Time Allowed: 3 hours
Maximum Marks: 80
General Instructions:
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 Objective type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 02 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 03 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 05 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based units of assessment of 04 marks each with sub-parts.
Section – A (20 Marks)
Select and write the most appropriate option out of the four options given for each of the questions 1 – 20. There is no negative mark for incorrect response.
Question 1.
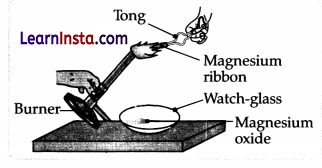
Which of the following is the correct observation of the reaction shown in the above setup?
(A) Brown powder of Magnesium oxide is formed.
(B) Colourless gas which turns lime water milky is evolved.
(C) Magnesium ribbon burns with brilliant white light.
(D) Reddish brown gas with a smell of burning Sulphur has evolved.
Answer:
(C) Magnesium ribbon burns with brilliant white light.
Explanation: When a piece of magnesium ribbon is ignited, light and heat are produced. Magnesium ribbon on burning in the air is an exothermic reaction
![]()
Question 2.
Methane gas released from wastewater treatment plants can be used as a source of fuel. Which chemical equation represents the combustion of methane to produce heat energy?
(A) CH4 + CO2 → 2O2 + 2H2O
(B) CH4 + 2O2 → CO2 + 2H2O
(C) 2O2 + 2H2O → CO2 + CH4
(D) CO2 + 2O2 → CH4 + 2H2O
Answer:
(B) CH4 + 2O2 → CO2 + 2H2O
Explanation: Reaction of methane is:
CH4 (Methane) + 2O2 (Oxygen) → CO2 (Carbon dioxide) + 2H2O (Water) + 890 KJ (Heat)
Question 3.
Acid present in tomato is:
(A) Methanoic acid
(B) Acetic acid
(C) Lactic acid
(D) Oxalic acid
Answer:
(D) Oxalic acid
Explanation: The acid present in tomatoes is oxalic acid.
Question 4.
Builders use Plaster of Paris to make the surface layer of the inner walls of a building. Which property of Plaster of Paris powder makes it a suitable building material?
(A) It is lightweight.
(B) It is white in colour.
(C) It is found readily in nature.
(D) It gets hard when mixed with water.
Answer:
(D) It gets hard when mixed with water.
Explanation: Plaster of Paris is a powder made of calcium sulphate hemihydrate. It forms a paste that hardens as it dries when mixed with water.
Question 5.
The electron dot structure of chlorine molecule is:
(A) 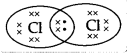
(B) 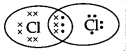
(C) 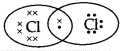
(D) ![]()
Answer:
(C) 
Explanation: The atomic number of chlorine is 17 so the electronic configuration of chlorine is 2,8,7. Since the outermost shell contains 7 valence electrons; hence, chlorine needs 1 more electron to complete its outermost shell. It combines with another chlorine atom by sharing 1 electron each to complete their outermost shells.
The number of shared electrons is 2. Hence, the correct presentation of a chlorine molecule is:

Question 6.
A part of a homologous series is shown below.
C3H4, C4H6, C5H8
Which of these compounds is a part of the series shown above?
(A) C2H2
(B) C2H4
(C) C8H6
(D) C6H14
Answer:
(A) C2H2
Explanation: The next homologous of the above series is C2H2. This is the homologous series of alkyne with general formula (CnH2n-2)
![]()
Question 7.
What is X in the reaction?
2AI + 3H2O → AI2O3 + X
(A) AI
(B) H2
(C) O3
(D) AIH3
Answer:
(B) H2
Explanation: X is hydrogen gas. The reaction is:
2AI(s) + 3H2O(g) → AI2O3(s) + 3H2(g)
Question 8.
Small hair-like structures line the upper part of the human respiratory tract. These structures trap the dust particles, germs and chemicals entering the human body during breathing. Smoking is likely to cause infections in the respiratory tract. Which statement best explains the fact?
(A) Smoking destroys the hair-like structures.
(B) Smoking causes excessive growth of the hair-like structures.
(C) Smoking stimulates the hair-like structures to release harmful chemicals.
(D) Smoking makes the hair-like structures wet and they fail to trap dust particles.
Answer:
(A) Smoking destroys the hair-like structures.
Explanation: Cilia are tiny hair-like projections that protect the airways by sweeping away mucus and dust particles and keeping the lungs clear. Smoking damages and eventually destroys these cilia.
Question 9.
The cerebellum in the brain controls the voluntary actions of the body. Which of these actions is controlled by the cerebellum?
(A) Beating of the heart
(B) Blinking of the eyes
(C) Watering of the mouth
(D) Jumping from a height
Answer:
(D) Jumping from a height
Explanation: The cerebellum controls voluntary movements such as walking, posture, balance coordination, eye movements and speech. This control results in smooth and balanced muscular activity.
Question 10.
A student while observing an embryo of a gram seed listed various parts of the embryo as listed below: Testa, Micropyle, Cotyledon, Tegmen, Plumule, Radicle.
On examining the list, the teacher commented that only three parts are correct. Select these three correct parts:
(A) Cotyledon, Testa, Plumule
(B) Cotyledon, Plumule, Radicle
(C) Cotyledon, Tegmen, Radicle
(D) Cotyledon, Micropyle, Plumule
Answer:
(B) Cotyledon, Plumule, Radicle
Explanation: When a plant produces a seed, it has 3 basic parts: plumule (the future shoot), radicle (the future root) and the cotyledons which may be 2 or 1 based on the type of plant being it dicot or monocot, respectively.
Question 11.
A cross between pea plant with white flowers (vv) and pea plant with violet flowers (VV) resulted in F2 progeny in which the ratio of violet (VV) and white (vv) flowers will be:
(A) 1:1
(B) 2:1
(C) 3:1
(D) 1:3
Answer:
(A) 1:1
Explanation: A cross between pea plants with white flowers (vv) and pea plants with violet flowers (VV) resulted in F, progeny of violet flowers (Vv). On self pollinating the F, progeny, the ratio of violet (VV) and white (vv) flowers in F2 is 1:1.
Question 12.
A student wants to obtain an erect image of an object using a concave mirror of 10 cm focal length. What will be the distance of the object from mirror?
(A) Less than 10 cm
(B) 10 cm
(C) Between 10 cm and 20 cm
(D) More than 20 cm
Answer:
(A) Less than 10 cm
Explanation: Object distance should be less than the focal length for the formation of an erect image. Hence, the range of distance of an object from the mirror should be less than 10 cm, i.e., from 0 to 10 cm in the front of the mirror from the pole. The nature of image so formed will be virtual and erect.
![]()
Question 13.
The phenomena of light involved in the formation of a rainbow are:
(A) Refraction, dispersion and scattering.
(B) Refraction, reflection and dispersion.
(C) Refraction, dispersion and internal reflection.
(D) Reflection, dispersion and total internal reflection.
Answer:
(C) Refraction, dispersion and internal reflection.
Explanation: The formation of a rainbow involves all three phenomena: Refraction, dispersion and total internal reflection. The sunlight shines on a water droplet. As the light passes into the raindrop, the light bends or refracts. Due to slow down of light, white light gets dispersed into seven colours. Then total internal reflection happens on the other end of the drop.
Question 14.
Choose the option giving correct matching the items given in Column I & Column II.
| Column I | Column II |
| (a) Physical environment | (i) Ozone depletion |
| (b) Exposure to UV radiation | (ii) Bacteria and fungi |
| (c) Chlorofluoro carbon compounds | (iii) Abiotic components |
| (d) Decomposers | (iv) Skin cancer |
| (a) | (b) | (c) | (d) | |
| (A) | (iii) | (i) | (iv) | (ii) |
| (B) | (iii) | (iv) | (i) | (ii) |
| (C) | (iii) | (iv) | (ii) | (i) |
| (D) | (iii) | (i) | (ii) | (iv) |
Answer:
(B) (iii) (iv) (i) (ii)
Explanation: The correct matching is:
| Column I | Column II |
| (a) Physical environment | (iii) Abiotic components |
| (b) Exposure to UV radiation | (iv) Skin cancer |
| (c) Chlorofluoro carbon compounds | (i) Ozone depletion |
| (d) Decomposers | (ii) Bacteria and fungi |
Question 15.
“X” in the given food chain is :
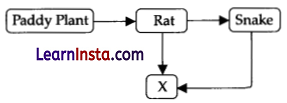
(A) Primary consumer
(B) Secondary consumer
(C) Tertiary consumer
(D) Producer
Answer:
(C) Tertiary consumer
Explanation: ‘X’ is represented by tertiary consumers. Tertiary consumers (X) feed on secondary consumer (snake), which feed on primary consumers (rat), which in turn feed on producers (paddy plant).
Question 16.
Which of these statements is incorrect about a balanced ecosystem?
(A) It is made up of interconnected food chains.
(B) It involves interdependence among living organisms and the environment.
(C) Animals are dependent on plants but plants are not dependent on animals.
(D) It involves communities made up of different populations of organisms.
Answer:
(C) Animals are dependent on plants but plants are not dependent on animals.
Explanation: The components of an ecosystem depend on each other to maintain the ecological balance. Plants are dependent on animals for carbon dioxide and other processes like pollination and dispersal of seeds.
![]()
Question Nos. 17 to 20 consist of two statements- Assertion (A) and Reason (R). Answer these questions by selecting the appropriate option given below:
(A) Both A and R are true, and R is the correct explanation of A.
(B) Both A and R are true, and R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Question 17.
Assertion (A): Reaction of quicklime with water is an exothermic reaction.
Reason (R): Quicklime reacts vigorously with water releasing a large amount of heat.
Answer:
(A) Both A and R are true, and R is the correct explanation of A.
Explanation: Exothermic reactions are those reactions in which heat is released along with the formation of products. Quicklime reacts vigorously with water releasing a large amount of heat along with the formation of calcium hydroxide.
Therefore, it is an exothermic reaction.
CaO (s) + H2O (l) → Ca(OH)2 (aq) + Heat
Question 18.
Assertion (A): Characteristics of parental plants can be preserved through vegetative reproduction.
Reason (R): Asexual reproduction involves only mitosis.
Answer:
(A) Both A and R are true, and R is the correct explanation of A.
Explanation: Vegetative reproduction is a type of asexual reproduction. Asexual reproduction involves a single individual, which gives rise to new individuals that are genetically identical to. their parents. It is because, when organisms reproduce asexually, only mitotic divisions are involved and.
Question 19.
Assertion (A): A current carrying straight conductor experiences a force when placed perpendicular to the direction of a magnetic field.
Reason (R): The net charge on a current carrying conductor is always zero.
Answer:
(B) Both A and R are true, and R is not the correct explanation of A.
Explanation: A current carrying conductor experiences force when placed perpendicular to the direction of magnetic field. The current carrying conductor has only electrons flowing though it which experience force, not the protons which are stationary. Hence, the net charge on it is zero. Hence, the statements are correct but reason is not the correct explanation for assertion.
Question 20.
Assertion (A): An ecosystem consists of biotic components and abiotic components.
Reason (R): Biotic and abiotic components play important roles for the sustenance of life and work independently in all ecosystems.
Answer:
(C) A is true but R is false.
Explanation: An ecosystem is a unit of biosphere in which biotic and abiotic components interact with each other. Biotic components depend on abiotic factors for survival.
Section – B (12 Marks)
(Q. no. 21 to 26 are very short answer questions.)
Question 21.
Trupti mixes an aqueous solution of sodium sulphate (Na2SO4) and an aqueous solution of copper chloride (CuCl2). Will this lead to a double displacement reaction? Justify your answer.
Answer:
Yes, this will lead to a double displacement reaction. A double displacement reaction is a type of chemical reaction in which the reactant ions exchange places to form new products. Sodium ions in the sodium sulphate bond with the chlorine ions in copper chloride and the copper ion bonds with the sulphate ion to form sodium chloride and copper sulphate.
Na2SO4 + CuCl2 → 2NaCl + CuSO4
Question 22.
Two green plants are kept separately in oxygen free containers, one in the dark and the other in sunlight- It was observed that plant kept in the dark could not survive longer. Give a reason for this observation.
Answer:
Plants release oxygen during photosynthesis, which can be utilized by the plant for respiration. The plant kept in the dark was not able to perform photosynthesis to generate some oxygen for respiration.
The carbon dioxide released by the plant after respiration was utilized by the plants to photosynthesize food, hence, the plant was able to survive.
Question 23.
Give two reasons why bile juice is considered to be an important secretion of the liver in the process of digestion?
Answer:
(i) Bile juice has bile salts such as bilirubin and biliverdin. These break down large fat globules into smaller globules so that the pancreatic enzymes can easily act on them. This process is known as emulsification of fats.
(ii) Bile juice also makes the medium alkaline and activates lipase.
OR
What is the other name of ’tissue fluid’? Write its two functions.
Answer:
Another name for tissue fluid is interstitial fluid.
Functions:
(i) Tissue fluid acts as a middleman. It hands over food materials and oxygen from the blood to the tissue cells and receives C02, nitrogenous wastes, hormones and other synthetic substances from the tissue cells and discharges them into blood.
(ii) It also keeps the tissue cells moist.
![]()
Question 24.
A light ray enters from medium A to medium B as shown in the figure.
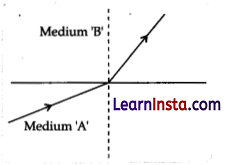
(a) Which one of the two media is denser w.r.t. other medium? Justify your answer.
Answer:
As it is clear from the figure, when the light ray travelled from medium A to medium B, then it bends towards the normal which means that medium B is optically denser than medium A.
(b) If the speed of light in medium A is VA and B is VB, what is the refractive index of B with respect to A?
Answer:
Refractive index of medium A

Question 25.
Priya has a copper wire and an aluminium wire of the same length. Can the electrical resistance of the two wires be the same? Justify your answer.
Answer:
Yes, the electrical resistance of the two wires can be the same.
If the area of cross-section of the two wires is different/ if the thickness of two wires is different.
OR
(a) Name the poles P, Q, R and S of the magnets in the following figures ‘a’ and ‘b’:

Answer:
In figure (a)- P is the north pole and Q is the south pole.
In figure (b)- R is the north pole and S is the south pole.
(b) State the inference drawn about the direction of the magnetic field lines on the basis of these diagrams.
Answer:
In the given diagrams, the direction of the magnetic field shows that outside the magnet, the magnetic field lines emerge from the North Pole of a magnet and merge at the South Pole of the magnet
Question 26.
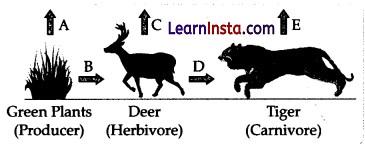
In the following food chain, vertical arrows indicate the energy lost to the environment and horizontal arrows indicate energy transferred to the next trophic level. Which one of the three vertical arrows (A, C and E) and which one of the two horizontal arrows (B and D) will represent more energy transfer? Give a reason for your answer.
Answer:
- Arrow A will represent more energy transfer as compared to C and E.
- Arrow B will represent more energy transfer as compared to D.
- When green plants are eaten by primary consumers, a great amount of energy is lost as heat to the environment. Some amount goes into digestion and in doing work and the rest goes towards growth and reproduction. An average of 10% of the food eaten is made available for the next level of consumers. This loss of energy takes place at every trophic level.
- Alternatively, In accordance with the 10% law of transfer of energy in a food chain, only 10% of energy available at one trophic level is transferred to the next trophic level.
![]()
Section – C (21 Marks)
(Q.no. 27 to 33 are short answer questions.)
Question 27.
(a) Suggest a safe procedure of diluting a strong concentrated add.
(b) Name the salt formed when sulphuric add is added to sodium hydroxide and write its pH.
(c) Dry HCL gas does not change the colour of dry blue litmus paper. Why?
Answer:
(a) The acid is always diluted by adding the acid to water slowly and with constant stirring. In addition to it, it is advised to wear gloves and
goggles and maintain safe distance.
(b) The salt formed when sulphuric acid is added to sodium hydroxide is sodium sulphate.
The balanced equation is:
2NaOH + H2SO4 → Na2SO4 + 2H2O
Its pH is approximately equal to 7.
(c) The colour of litmus paper changes only in the presence of ions like hydrogen (H+) or hydronium (H2O+) ions.
HCl can produce these ions only in the form of aqueous solution.
Hence, dry HCl gas does not change the colour of dry litmus paper.
Question 28.
A reddish brown metal used in electrical wires when powdered and heated strongly turns black. When hydrogen gas is passed over this black substance, it regains its original colour. Based on this information answer the following questions:
(a) Name the metal and the black substance formed.
(b) Write balanced chemical equations for the two reactions involved in the above information.
Answer:
(a) The brown colour metal is copper (Cu) and black coloured substance is copper oxide (CuO).
(b) (i) When Cu metal is heated in an open china dish then Cu metal gets oxidised.
Cu + O2 → 2CuO
(ii) When hydrogen gas is passed over the black substance then CuO gets reduced.
CuO + H2 → Cu + H2O
OR
A metal ‘M’ on reacting with dilute acid liberates a gas ‘G’. The same metal also liberates gas ‘G’ when reacts with a base.
(a) Write the name of gas ‘G’.
(b) How will you test the presence of this gas?
(c) Write chemical equations for the reactions of the metal with (1) an acid and (2) a base.
Answer:
(a) Hydrogen gas
(b) On bringing a burning match stick near the mouth of the test tube, a pop sound is heard.
(c) Reaction with acid:
Zn(s) + 2HCl(aq) → ZnCl2 + H2(g)
Reaction with base:
2NaOH(aq) + Zn(s) → Na2ZnO2(aq) + H2(g)
Question 29.
Name the gland and the hormone secreted by it in scary situations in human beings. List any two responses shown by our body when this hormone is secreted into the blood.
Answer:
Hormone: Adrenaline
Functions:
(i) Increases the heart rate.
(ii) Increases blood pressure.
(iii) Expands the air passages of the lungs.
(iv) Enlarges the pupil in the eye.
Question 30.
What are chromosomes? Explain how the stability of the DNA of the species is ensured in sexually reproducing organisms.
Answer:
Chromosomes are thread like structures which are made up of proteins and DNA. DNA contains the information of traits which are passed from parents to offspring from one generation to another. In sexually reproducing organisms, the stability of DNA is ensured by the DNA copying mechanism. The DNA replication process is very efficient and error free which in turn maintains the stability of the DNA. The chromosome number is also maintained during reproduction by the process of meiosis during gamete formation.
![]()
Question 31.
The diagram given shows an object O and its image I. Without actually drawing the ray diagram state the following:
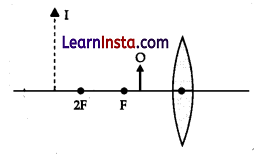
(a) Type of lens (Converging/Diverging)
Answer:
Converging lens
(b) Name two optical instruments where such an image is obtained.
Answer:
Microscope and Telescope
(c) List three characteristics of the image formed if this lens is replaced by a concave mirror of focal length F and an object is placed at a distance F/2 in front of the mirror.
Answer:
Characteristics of the image formed by concave mirror are:
(i) Virtual image
(ii) Magnified image
(iii) Image behind the mirror
Question 32.
(a) List the factors on which the resistance of a uniform cylindrical conductor of a given material depends.
Answer:
Factors on which the resistance of a conductor depends:
- Length of conductor Resistance is directly proportional to the length of the conductor. This means resistance increases with an increase in the length of the conductor. This is why long wires create more resistance to the electric current.
- Area of cross-section: Resistance is inversely proportional to the area of cross-section of the conductor. This means
resistance decreases with an increase in the area of cross-section of the conductor and vice versa. This is why, a thick copper wire creates less resistance to electric current. - Temperature: Resistance is directly proportional to the temperature.
- Nature of material: Resistance depends on the nature of material. Some materials like silver are good conductor of electricity while some like plastic are bad conductor of electricity.
(b) The resistance of a wire of 0.01 cm radius is 10 Ω. If the resistivity of the wire is 50 × 10-8 Ω m, find the length of this wire.
Answer:
Given, Radius, r = 0.01 cm = 0.0001 m
Resistance, R = 10 Ω
Resistivity, ρ = 50 × 10-8 Ω m
\(\begin{aligned}
R & =\rho \frac{L}{A} \\
L & =\frac{R A}{\rho} \\
& =\frac{10 \times 314 \times(0.0001)^2}{50 \times 10^{-8}}
\end{aligned}\)
= 0.628 m (Area, A = πr²)
Hence, the length of the wire is 0.628 m.
Question 33.
(a) Why is an alternating current (A.C.) considered to be advantageous over direct current (D.C.) for the long distance transmission of electric power?
Answer:
Alternating current can be transmitted over long distances at very high voltages so as to ensure that current value is very low and as a result very low amount of energy is lost during transmission. When it reaches near the household, the voltage can be stepped down to restore the required value of current whereas the voltage of direct current cannot be stepped up and down.
(b) How is the type of current used in household supply different from the one given by a battery of dry cells?
Answer:
The type of current used in domestic household supply is alternating while a battery of dry cells supplies direct current.
(c) How does an electric fuse prevent the electric circuit and the appliances from a possible damage due to short circuiting or overloading?
Answer:
Electric fuse is made of wires of very low melting point. If there is a current larger than the specified value, the temperature of a fuse wire increases and it melts to break the electric circuit and stop the flow of unduly high electric current. This saves appliances from possible damage.
![]()
Section – D (15 Marks)
(Q.no. 34 to 36 are long answer questions.)
Question 34.
(a) A compound “A” with a molecular formula of C2H4O2 reacts with a base to give salt and water. Identify ‘A’, state its nature and the name of the functional group it possesses. Write the chemical equation for the reaction involved.
Answer:
Compound A – Acetic add/ CH3COOH
Nature: Its nature is acidic.
Name of the functional group: COOH
Chemical equation:
CH3COOH(aq) + NaOH(aq) → CH3COONa(aq) + H2O(l)
(b) When the above-stated compound A reacts with another compound ‘B’ having molecular formula C2H6O in the presence of an add, a sweet-smelling compound ‘C’ is formed.
(i) Identify ‘B’ and ‘C’.
Answer:
B- Ethanol (C2H5OH)
C- Ethyl acetate
(ii) State the role of add in this reaction.
Answer:
Role of acid: It is a catalyst which will speed up the process and esterification will proceed speedily and complete on time.
(iii) Write the chemical equation for the reaction involved.
Answer:
Chemical equation:
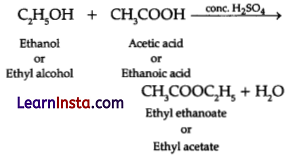
OR
(a) Match the following pH values 1, 7, 10, and 13 to the solutions given below:
(i) Milk of magnesia
(ii) Gastric juices
(iii) Brine
(iv) Aqueous sodium hydroxide.
Answer:
(i) Milk of magnesia: 10
(ii) Gastric juices: 1
(iii) Brine: 7
(iv) Aqueous sodium hydroxide: 13
![]()
(b) Amit and Rita decided to bake a cake and added baking soda to the cake batter. Explain with a balanced reaction, the role of the baking soda. Mention any other use of baking soda.
Answer:
Baking soda undergoes thermal decomposition to form Na2CO3 C02 and H2O. CO2 makes the cake fluffy & soft.
\({NaHCO}_3 \stackrel{\text { heat }}{\longrightarrow} {Na}_2 {CO}_3+{CO}_2+{H}_2 {O}\)
Uses:
Used in fire extinguishers/antacid to neutralize excess add in the stomach /to neutralize the effect of add in insect sting.
Question 35.
Give a reason for the following:
(a) During reproduction, inheritance of different proteins will lead to altered body designs.
Answer:
During reproduction, the information for the inheritance of characteristics is passed on from the parents to the offspring in the form of DNA. DNA in the cell nucleus carries the genetic information. Hence DNA copying is the basic event in reproduction. If the information dining the DNA copying is changed, different proteins will be made. Different proteins will eventually lead to altered body designs.
(b) Fertilisation cannot take place in flowers if pollination does not occur.
Answer:
The process of fertilization is the fusion of both male and female gametes. If pollination does not occur it means that the male gamete is not available, hence fertilisation cannot take place.
(c) All multicellular organisms cannot give rise to new individuals through fragmentation or regeneration.
Answer:
Regeneration and fragmentation are only possible when the entire body part is made up of similar kinds of cells. All multicellular organisms cannot give rise to new individuals through fragmentation or regeneration because the complex multicellular organisms have organ system level organisation. The tissues in these organisms are highly differentiated to perform specialised functions and cannot regenerate a new individual.
(d) Vegetative propagation is practised for growing only some types of plants.
Answer:
Vegetative propagation is practised for growing some types of plants because of the following advantages:
- It is used to grow a plant in which viable seeds are not formed or very few seeds are produced such as orange, banana, pineapple.
- It helps to introduce plants in new areas where the seed germination fails to produce mature plant due to change in environmental factors and the soil.
- It is more rapid, easier and cheaper method.
- By this method a good quality of a race or variety can be preserved.
(e) The parents and off-springs of organisms reproducing sexually have the same number of chromosomes.
Answer:
Gametes of sexually-reproducing animals have half the number of chromosomes as that of the parents. During fertilisation, when two gametes i.e. male and female gametes fuse, the offspring produced will have the same amount of DNA or the same number of chromosomes as that of the parent.
OR
(a) Why is the use of iodised salt advisable? Name the disease caused due to deficiency of iodine in our diet and state its one symptom.
Answer:
- Iodine is essential for the functioning of thyroid/formation of thyroxine hormone.
- Disease is Goitre.
- Swollen neck
(b) How do nerve impulses travel in the body? Explain.
Answer:
Impulse travels from dendrite to cell body, then along the axon to its end. At the end, some chemicals are released which fill the gap of synapse, and start a similar electrical impulse to another neuron and the impulse further travel in the body.
Detailed(b) Answer:
The information received at the end of the dendritic tip, sets off a chemical reaction that creates an electrical impulse. This impulse travels from the dendrite to the cell body and then along the axon to it send. Here, the electrical impulse sets off the release of some chemicals which cross synapse, inducing a similar impulse in a dendrite of the next neuron.
![]()
Question 36.
(a) Define the following terms in the context of spherical mirrors:
(i) Pole
(ii) Centre of curvature
(iii) Principal axis
(iv) Principal focus
Answer:
(i) Pole: Centre of the reflecting surface of the mirror.
(ii) Centre of curvature: The centre of the hollow sphere of which the reflecting surface is a part.
(iii) Principal axis: Straight line passing through the pole and the centre of curvature of a spherical mirror.
(iv) Principal focus: Incident rays parallel to the principal axis, after reflection, either converge or appear to diverge from a fixed point on the principal axis called the principal focus of the spherical mirror.
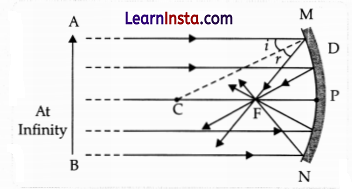
(b) Draw ray diagrams to show the principal focus of a:
(i) Concave mirror
Answer:
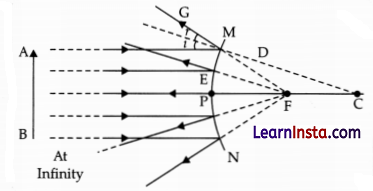
(ii) Convex mirror
Answer:
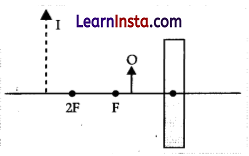
(c) Consider the given diagram in which Mis a mirror and P is an objed and Q is its magnified image formed by the mirror.
State the type of the mirror M and one Characteristic property of the image Q.
Answer:
Concave mirror
Image formed is virtual
(a) u =- 60 cm, f = -30 cm, v = ?
\(\begin{aligned}
\frac{1}{f} & =\frac{1}{v}-\frac{1}{u} \\
\frac{1}{v} & =\frac{1}{f}+\frac{1}{u} \\
\frac{1}{v} & =\frac{1}{(-30 \mathrm{~cm})}+\frac{1}{(-60 \mathrm{~cm})}=\frac{-3}{60} \\
∴ \quad v & =-20 \mathrm{~cm} \\
m & =\frac{v}{u}=\frac{-20 \mathrm{~cm}}{-60 \mathrm{~cm}}=\frac{1}{3}
\end{aligned}\)
Distance of the image will be 20 cm in front of lens.
(b) Nature: Virtual
Position: 20 cm from the lens on the same side as the object.
Size: Diminished
Erect/Inverted: Erect
(c) 
Section – E (12 Marks)
(Q.no. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts.)
(Internal choice is provided in one of these sub-parts.)
Question 37.
Read the given passage and answer the questions that follow:
The pictures show the bonds formed by the sharing of electrons by the atoms of four different molecules.

(a) Out of these molecules- Hydrogen, carbon, nitrogen and oxygen, which has the strongest bond between its atoms? What is this type of bond between atoms known as?
Answer:
Carbon can gain four electrons and form C4- anion or lose four electrons to form the C4+ cation. It forms four covalent bonds by sharing its valence electrons with other atoms.
This type of bond between atoms is known as covalent bond covalent, carbon compounds are those where there is a carbon-carbon bond.
![]()
(b) Which of these statements is true about carbon compounds? Write ‘Yes’ or ‘No’ for the correct response.
| Is this true about carbon compounds? | Yes or No |
| (i) They are good conductors of electricity? | |
| (ii) They exist in either saturated or unsaturated form. | |
| (iii) They have lower boiling points than ionic compounds |
Answer:
| Is this true about carbon compounds? | Yes or No |
| (i) They are good conductors of electricity? | No |
| (ii) They exist in either saturated or unsaturated form. | Yes |
| (iii) They have lower boiling points than ionic compounds | Yes |
OR
(b) Which element exhibits the property of catenation to the maximum extent and why?
Answer:
Carbon, due to strong C-C bond.
Question 38.
The most obvious outcome of the reproductive process is the generation of individuals of similar design, but in ‘ sexual reproduction they may not be exactly alike. The resemblances as well as differences are marked. The rules of heredity determine the process by which traits and characteristics are reliably inherited. Many experiments have been done to study the rules of inheritance
(a) Why an offspring of a human being is not a true copy of his parents in sexual reproduction?
Answer:
An offspring is not a true copy of his parents in sexual reproduction because of variations due to the recombination of genetic material from two different parents.
(b) While performing experiments on inheritance in plants, what is the difference between F1 and F2 generation?
Answer:
All the F1 offspring are genetically identical and exhibit the dominant trait.
F2 offspring can exhibit a range of dominant to recessive traits.
(c) Why do we say that variations are useful for the survival of a species over time?
Answer:
Variations help the organism to become more adapted to the changing environmental conditions. This helps the organisms to overcome the adverse conditions.
OR
(c) Study Mendel’s cross between two plants with a pair of contrasting characters.
RRYY (Round Yellow) × rryy (Wrinkled Green)
He observed 4 types of combinations in F2 generation. Which of these were new combinations? Why do new features which are not present in the parents, appear in F2 generation?
Answer:
Round Green and Wrinkled Yellow are the new combinations in F2 generation.
New features which are not present in parents appear in F2 generation due to the segregation of different combinations of alleles of different characters independently during gamete formation and fertilisation.
Question 39.
Study the circuit shown in which three identical bulbs B1, B2 and B3 are connected in parallel with a battery of 4.5 V and answer the following questions.
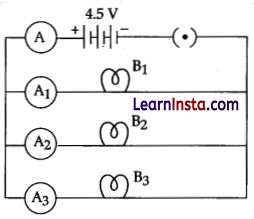
(a) What will happen to the glow of the other two bulbs if the bulb B3 gets fused?
(b) If the wattage of each bulb is 1.5 W how much readings will the ammeter A show when all the three bulbs glow simultaneously.
(c) Find the total resistance of the circuit.
Answer:
(a) Other bulbs will glow with the same brightness.
(b) When the bulbs are in parallel, wattage will be added (4.5 W) and the ammeter reading would be,
I = P/V
= 4.5W / 4.5V
= 1.0 Ampere
(c) Since, the ammeter reading is 1.0 ampere, the resistance of the combination is 4.5 V/ 1.0 A = 4.5 Ω
![]()
OR
How many resistors of 88 Ω are connected in parallel to carry 10 A current on a 220 V line?
Answer:
Let n number of resistances is connected in
parallel.
Equivalent resistance, 1/ Rp = n/88
Rp = 88/n …. (i)
Since, V = IR …. (ii)
Now from equations (i) and (ii):
n = I × 88/220
= 10 × 88/220
Or, n = 88/22 = 4 resistors.