Experts have designed these Class 8 Science Notes Chapter 9 The Amazing World of Solutes, Solvents, and Solutions Class 8 Notes for effective learning.
Class 8 Science Chapter 9 The Amazing World of Solutes, Solvents, and Solutions Notes
Class 8 The Amazing World of Solutes, Solvents, and Solutions Notes
Class 8 Science Chapter 9 Notes – The Amazing World of Solutes, Solvents, and Solutions Notes Class 8
→ Uniform Mixture: A mixture in which the components are evenly distributed throughout and are not visibly separate. For example, Oral Rehydration Solution (ORS), used to treat dehydration, it is a mixture of salt, sugar and water.
→ Non-uniform Mixture: A mixture in which the components are not evenly distributed and can be seen with the naked eye or with a magnifying device. For example, a mixture of chalk powder or sand, or sawdust in water.
![]()
→ Solution: It is a uniform mixture of two or more substances.
- When a solid is mixed with a liquid to form a solution, the solid component is called the solute, and the liquid component is called the solvent. The solute dissolves in the solvent to form a solution.
Solute + Solvent → Solution - In a solution of two liquids, the substance present in smaller amount is called the solute, while the one in larger amount is called the solvent.
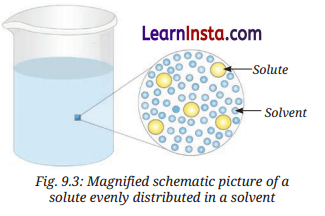
- In a gaseous solution, the gas present in smaller amount is called the solute, while the one in larger amount is called the solvent. For example, air is a gaseous solution. Since nitrogen is present in the largest amount in the air, it is considered as the solvent, while oxygen, argon, carbon dioxide, and other gases are considered as solutes.
- The Chashni (sugar syrup) of the Indian sweet Gulab jamun is made of a large amount of sugar (solid) dissolved in a small amount of water (liquid). In this sugar syrup, water is still considered the solvent, and sugar is the solute.
→ Unsaturated Solution: A solution in which more solute can be dissolved at a given temperature.
→ Saturated Solution: A solution in which the maximum amount of solute has been dissolved, and no more of it can be dissolved at that temperature.
![]()
→ Concentration: The amount of solute present in a fixed quantity of solution (or solvent).
- Depending upon the amount of solute present in a fixed quantity of solution, it can be called a dilute solution (less amount of solute) or a concentrated solution (more amount of solute).
→ Solubility: The maximum amount of solute that dissolves in a fixed quantity of the solvent at a particular temperature.
- Solubility of solids in liquids usually increases with an increase in temperature. A saturated solution at a particular temperature becomes unsaturated solution if the temperature is increased.
- Solubility of gases in liquids usually decreases with an increase in temperature. Gases like oxygen dissolve in water, but only in small amount. This small amount of dissolved oxygen supports all aquatic life. The mixture is uniform because the gases dissolve evenly in water. As temperature increases, gas solubility decreases. Cold water holds more oxygen, which benefits aquatic organisms, while warm water holds less.
→ Solvents in Traditional Indian Medicine: Water is mainly used as a solvent to prepare medicines in Ayurveda, Siddha, and other traditional systems of medicine in India. Drug formulations are also prepared using hydro-alcoholic extracts of herbs. Substances like oil, ghee, and milk are also used as solvents for their therapeutic benefits.
![]()
→ Asima Chatterjee-A Pioneer Scientist: Asima Chatterjee was a renowned scientist who developed anti-epileptic and anti-malarial drugs. She used solvents and solutions to extract useful compounds from medicinal plants.
- She was the second Indian woman to earn a Doctorate of Science, after Janaki Ammal.
- She was the first woman to receive the Shanti Swarup Bhatnagar Award, in the field of chemical science and was honoured with the Padma Bhushan.
→ Floating and Sinking: Objects that float in a liquid are generally lighter, while those that sink are heavier.
→ Density: It is defined as the mass present in a unit volume of a substance.
Mathematically, Density =\(\frac{\text { Mass }}{\text { Volume }}\)
- The density of a substance is independent of its shape or size.
- Density depends on temperature and pressure.
→ Units of Density:
- SI unit: kg/ m3 (kilogram per cubic metre)
- Common units for liquids and solids: g/ mL or g/cm3
→ Conversion factor for density:
1kg/m3 =1000g/m3
= 1000g/1000L = 1g/L
= 1/1000 mL=1g/1000cm3
→ Relative Density: Relative density of any substance with respect to water
![]()
= \(\frac{\text { Density of that substance }}{\text { Density of water at that temperature }}\)
For example, A 1-litre packet of oil weighing 910 grams shows that oil is less dense than water (which weighs 1000 grams per litre). So, its relative density is less than 1, and it floats on water.
→ Mass and Weight: The ‘mass’ and ‘weight’ have different meanings in Science.
- Mass is the quantity of matter present in any object. Its units are gram (g) and kilogram (kg). The instrument used to measure the mass of an object is known as a balance.
- Weight is the force by which the Earth attracts an object or a substance towards itself. It is measured in newtons (N).
- Most balances (except two-pan balances) measure weight, but their scales are marked in mass units like grams or kilograms. This is because they are calibrated to give mass readings under normal gravity.
- A two-pan balance, however, works differently. It compares the object with known standard masses, so it directly measures mass, not weight.

→ Volume: It is the space occupied by an object.
- The SI unit of volume is cubic metres (m)3. It is the volume of a cube whose each side is one metre in length.
- Volume of smaller objects is expressed in a decimetre cube (dm3) or centimetre cube (cm3). One centimetre cube is also written as one cc.
- Volume of liquids is expressed in litres (L).
1L = 1 dm3, 1 mL=1 cm3
→ Measuring Cylinder: An apparatus used to measure the volume of liquids. It is a narrow, transparent cylindrical container with one side open and the other side closed. There are markings on the transparent body of the cylinder that indicate the volume of liquid in the measuring cylinder.
![]()
- Measuring cylinders are available in different sizes such as 5 mL, 10mL, 25 mL, 50 mL, etc.
- The smallest volume that a measuring cylinder can measure depends on its capacity.
→ Meniscus: It is the curved surface formed by a liquid inside the measuring cylinder.

- For measuring volume of water or colourless liquids, read the mark on the measuring cylinder that coincides with the bottom of the meniscus.
- For measuring volume of coloured liquids, read the mark on the measuring cylinder that coincides with the top of the meniscus.
→ Measuring Volume of Solids:
- For regular shapes (cuboid):
Volume = length (l) × width (w) × height (h) - For irregular shapes: Use water displacement method. Subtract the initial water level from the final level after submerging the object to find its volume. The values of volume are obtained in units of mL , which can be written in the equivalent unit cm3 for solids.
![]()
→ Density of Different Layers of Earth: The Earth consists of several layers: Crust, upper mantle, lower mantle, outer core, and inner core.
- The outermost layer (crust) is the lightest.
- Density of the different layers increases as we go deeper due to rising pressure and temperature, which make the materials heavier and more compact.
→ Traditional Boats in Ancient Times: Before big ships were invented, people used bamboo and wooden logs to make simple boats and rafts.Bamboo was used because it is light and floats easily on water. People tied bamboo poles or used strong tree logs to cross rivers, fish, and trade. Even today, such traditional boats are still used in some areas for transport and tourism.
→ Effect of Temperature on Density: Generally, the density of a substance decreases with an increase in temperature.
- As temperature increases, the particles of a substance (solid, liquid, or gas) tend to move away and spread. This causes an increase in volume but no change in mass. Thus, density decreases.
- Hot air moves up as it is less dense than the cool air around it. The hot air balloon works on the same principle.
→ Effect of Pressure on Density: Pressure affects density differently depending on the state of matter.
- For gases, density increases with an increase in pressure.
- For liquids, pressure has a small effect because they are nearly incompressible.
- For solids, pressure has very little effect than liquids because the particles in solids are very close to each other.
→ Ice Floats on Water: Density of water is highest at 4°C. It means water is heaviest at 4°C. When it freezes at 0°C, its particles arrange themselves in a way that takes up more space (expansion). This makes ice less dense than water, so it floats. The floating ice forms a layer on the water, keeping the water below warmer, which helps aquatic animals survive in cold weather.
![]()
→ An egg sinks in tap water because it is denser than the water. Adding salt increases the water’s density, making the egg float. This shows how density affects floating and sinking.
→ Our Scientific Heritage: In Ningel village (Thoubal district, Manipur), salt is made using traditional methods. Women collect salty water from wells and boil it in metal pans over firewood kilns. After evaporation, salt crystals are shaped into round cakes using banana leaves and simple tools. These cakes are wrapped in a cloth called phanek and are believed to have medicinal value. This tradition is a part of Ningel’s rich culture and heritage.