Students can access the CBSE Sample Papers for Class 10 Science with Solutions and marking scheme Term 2 Set 6 will help students in understanding the difficulty level of the exam.
CBSE Sample Papers for Class 10 Science Term 2 Set 6 with Solutions
Section – A
Question 1.
What is a homologous series? Explain with an example. (2)
Answer:
A homologous series is a series of carbon compounds that have different numbers of carbon atoms but contain the same functional group. There is a difference of -CH2 unit between each successive member and mass differ by 14 u. For example, methane, ethane, propane, butane, etc., are all part of the alkane homologous series. The general formula of this series is CnH2n+2.
Question 2.
What limits the number of trophic levels in a food chain?
OR
Suggest any four activities in daily life which are eco-friendly? (2)
Answer:
The flow of energy in each trophic level follows 10% rule i.e., only 10% of the energy is available to the next higher trophic level hence, the amount of energy goes on decreasing at each trophic level which limits the number of trophic levels in a food chain.
OR
The four activities in daily life which are eco-friendly are:
- Carrying paper bags instead of polythene bags for shopping.
- Use of compost and bio-fertilizers, bio-pesticides instead of chemical fertilizers and pesticides.
- Segregating biodegradable and non-biodegradable substances and putting them in separate dustbins.
Rain water harvesting.
Question 3.
An element belongs to third period and group 16 of modern periodic table. (2)
(a) Determine the number of valence electrons and valency of the element.
(b) Molecular formula of the compound when X reacts with hydrogen and write its electron dot structure.
(c) Name the element X and state whether it is metallic or non-metallic.
Answer:
(a) Valency electron = 6
Valency = 2
(b) H2S
(c) Sulphur, Non-metal.
Question 4.
Out of 60 W and 40 W lamps, which one has a higher electrical resistance when in use. (2)
OR
Write the formula for current ‘I’ flowing through a conductor if ‘n’ electrons flow through the crosssection of a conductor in time ‘t’.
Answer:
Power (P) = \(\frac { V² }{ R }\)
From the above formula, P is inversely proportional to R (resistance) as voltage remaining the same. Hence, 40 W lamp has high resistance.
OR
If ‘n’ electrons flow through the cross-section of a conductor in time T, the total charge ‘Q’ passing through the conductor is:
(e is the charge on an electron = 1.6 x 10-19 C)
The current T in the conductor is:
I = \(\frac { Q }{ t }\) = \(\frac { ne }{ t }\)
Question 5.
List, two differences between acquired traits and inherited traits by giving an example of each. (2)
Answer:
| Acquired traits | Inherited traits |
| 1. These are somatic variations and not bring any change in DNA. | 1. These are genetic variations and bring about changes in the DNA. |
| 2. These traits develop throughout the life time of an individual. Example, learning of dance and music. | 2. These traits are transferred by or (inherited) by the parents to the offsping. Example, Eye colour, hair colour. |
Question 6.
Study the below diagram and label the parts A, B, C and D. (2)
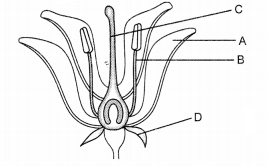
Answer:
A – Petals; B – Stamens; C – Pistil; D – Sepals.
Question 7.
Why is variation beneficial to the species but not necessary for the individual? (2)
OR
Distinguish between pollination and fertilisation. Mention the site and the product of fertilisation in a flower.
Answer:
Due to recombination and crossing over in meiosis process during formation of gametes as well as during sexual reproduction the mixing up of male with female gametes produce some variations in the offsprings. These variations are necessary for survival of a particular species to the changing environment. If there would be no variations then there will be less chance of a particular species to get adapted to the changed environment and with course of time that particular species may extinct. Variations also lead to evolution of the species. Thus, it is said that variation is beneficial to the species but not necessary for the individual.
OR
1. The transfer of pollen grains from anther of a stamen to the stigma of a carpel is called pollination whereas fertilisation is the process when the male gamete present in pollen grain joins the female gamete present in ovule.
2. Pollination is an external mechanism whereas fertilisation is an internal mechanism which takes place inside the flower. Site of fertilisation in flower is ovary. Product of fertilisation in flower is Zygote.
Section – B
Question 8.
The given box diagram represents the ratio of females to males or the sex ratio in our country for 10 decades (1901 to 2001). Answer the following questions in the light of your knowledge of sex determination and the data presented in the box diagram. (3)
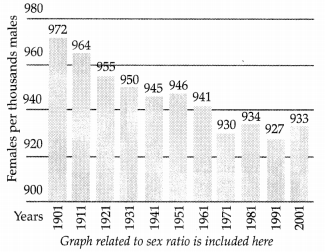
(a) What does the bar diagram show?
(b) As per scientific knowledge regarding sex determination, what should be the sex ratio or the male to female ratio at a given point of time?
(c) Assign one reason to the trend showing deviation from the expected sex ratio.
(d) Suggest a way by which such as trend can be stopped.
OR
Write the functions of each of the following parts in a human female reproductive system.
(a) Ovary
(b) Uterus
(c) Fallopian tube
Describe the structure and functions of placenta.
Answer:
(a) Bar diagram shows the proportion of females in the population over a decade.
(b) 1 : 1
(c) Female foeticide
(d) Banning sex test of unborn baby; increasing awarenesss and education.
OR
(a) Ovary: It produces female gametes and female sex hormones like estrogen, progesterone.
(b) Uterus: It supports and nourishes the developing foetus. It expands according to the growth of the baby.
(c) Fallopian tube: They are the site of fertilisation and carry eggs from ovary to uterus.
Placenta connects baby with mother’s body. It is a disc like structure embedded in uterine wall. It is a special tissue formed by the interlocking of two sets of villi, one set given by wall of uterus and other set from embryo. It is the site of exchange of materials between the blood of mother and baby. It provides nutrients, oxygen from mother to foetus and removes excretory substances, carbon dioxide from foetus.
Question 9.
Rhea noted the readings of her home’s electricity meter on Monday at 9 a.m. and again on Tuesday at 9 a.m. (as shown in figure given below). (3)
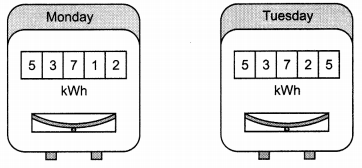
(a) What was the meter reading on Monday?
(b) What was the meter reading on Tuesday?
(c) How many units of electrical energy have been used?
Answer:
(a) Meter reading on Monday = 53712
(b) Meter reading on Tuesday = 53725
Number of units of electrical energy used
= 53725 – 53712
= 13 units
Question 10.
(a) Given any two examples of each: (3)
(i) Organisms occupying the first trophic level.
(ii) Biodegradable wastes of humans.
(iii) Ecosystem
(iv) Abiotic factors of an ecosystem
(b) Complete the below diagram by filling spaces marked as A, B, C and D:
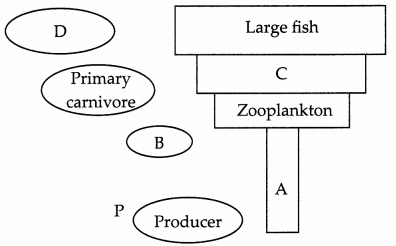
Answer:
(a) (i) Grass, green plants
(ii) Kitchen waste like peels of vegetables, fruits, left over foods and old newspaper.
(iii) Natural ecosystem includes forest, pond and artificial ecosystem which include garden, parks, crop fields.
(iv) Physical factors like temperature, sunlight and edaphic factors like soil.
(b) A – Phytoplankton
B – Herbivores
C – Small fish
D – Secondary carnivores
Question 11.
Based on the structures given below answer the following questions:
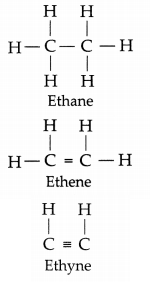
(a) Give the name of the third member of the homologous series of alkene.
(b) Write the number of covalent bonds in the molecule of butane.
(c) Write the number of covalent bonds in the molecule of propane.
Answer:
(a) Butene (C4H8)
(b) Thirteen covalent bonds
(c) 10 covalent bonds
(d)
Question 12.
Mendeleev arranged 63 elements known at that time in the periodic table. According to Mendeleev “the properties of the elements are a periodic function of their atomic masses.” The table consists of eight vertical columns called ‘groups’ and horizontal rows called ‘periods’. Merits of Mendeleev’s Periodic Table: At some places the order of atomic weight was changed in order to justify the chemical and physical nature. Mendeleev left some gap for new elements which were not discovered at that time. One of the strengths of Mendeleev’s periodic table was that, when inert gases were discovered they could be placed in a new group without disturbing the existing order. Its main characteristics are that the elements are arranged in vertical rows called groups and horizontal rows called periods.
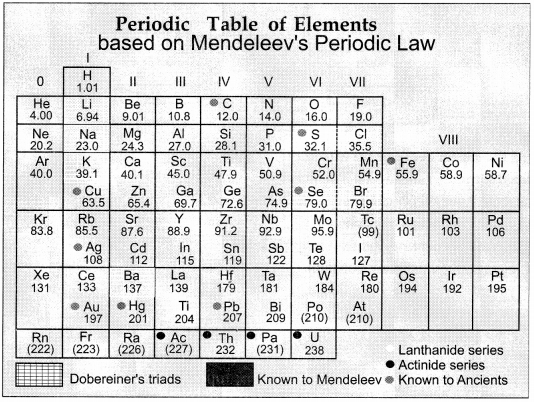
(a) What is the basis of arrangement of elements in Mandeleev’s periodic table?
(b) Why were there some gaps in Mendeleev’s periodic table?
(c) State whether the statement is true or false.
Mendeleev’s periodic table consists of eight vertical columns called ‘groups’ and horizontal rows called ‘periods’.
OR
Answer the following questions:
(a) The modern periodic table has been evolved through the early attempts of Dobereiner, Newland and Mendeleev. List one advantage and one limitation of all three attempts.
(b) Name the scientist who first of all showed that atomic number of an element is a more fundamental property than its atomic mass.
(c) State Modern Periodic law. (3)
Answer:
(a) In Mendeleev’s periodic table, elements are arranged according to atomic masses.
(b) There were some gaps in Mendeleev’s periodic table for unknown elements.
(c) Mendeleev’s periodic table was divided into groups and columns. Therefore, the given statement is true.
(d) According to Dobereiner’s triad, the atomic mass of middle element is the average of other two elements. Therefore, (7 + 39) = 46/2 = Y = 23.
OR
1. Dobereiner:
→ Advantage: Group three elements having similar properties they were called as the Dobereiner triads. He arranged them in order of increasing atomic mass and the mass of middle element was equal to the average of the other two elements.
→ Limitation: He was able to identify only 4 traids but he failed in his attempt to group nitrogen, phosphorus and arsenic because atomic mass of phosphorus is not the average of other two.
2. Newland:
→ Advantages: He arranged the elements in horizontal rows in order of increasing atomic mass. The property of every 8th element was similar to the 1st element.
→ Limitation: His arrangement was only applicable to lighter elements.
3. Mendeleev:
→ Advantage: He arranged the elements in order of their increasing atomic mass and he corrected the mass of beryllium from 4.5 to 13.5 u.
→ Limitation: Position of hydrogen and isotopes were not justified in his periodic table.
(b) Henry Moseley showed that atomic number of an element is a more fundamental property than its atomic mass.
(c) Modern periodic law states that “the properties of elements are the periodic function of their atomic number. Repetition of properties is due to the same outer electronic configuration.”
Question 13.
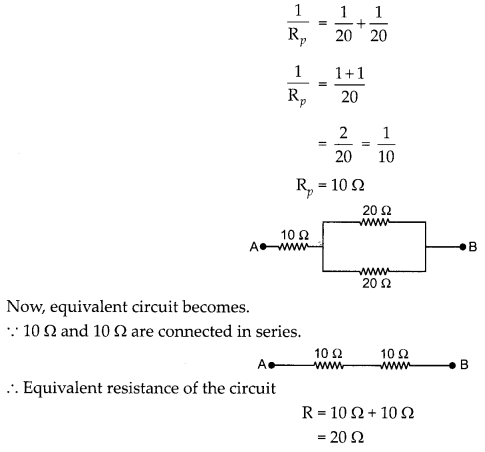
Calculate the equivalent resistance of the following network: (3)
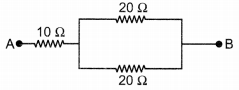
Answer:
Let Rp is the equivalent resistance of resistors connected in parallel.
∴ Equivalent resistance of the circuit
Section – C
This section has 02 case-based questions (14 and 15). Each case is followed by 03 sub-questions (a, b and c). Parts a and b are compulsory. However, an internal choice has been provided in part c.
Question 14.
(a) The current flowing through a resistor connected in a circuit and the potential difference developed across its ends are as shown in the diagram by milliammeter and voltmeter readings respectively: (4)
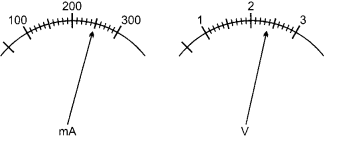
(i) What are the least counts of these meters?
(ii) What is the resistance of the resistor?
(b) Why is the tungsten metal more coiled in the bulb and not installed in straight parallel wire form?
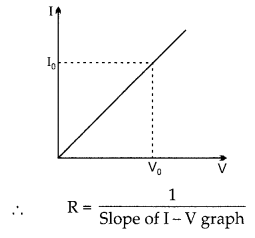
(c) While studying the dependence of potential difference (V) across a resistor on the current (I) passing through it, in order to determine the resistance of the resistor, a student took 5 readings for different values of current and plotted a graph between V and I. He got a straight line graph passing through the origin. What does the straight line signify? Write the method of determining resistance of the resistor using this graph.
OR
What would you suggest to a student if while performing an experiment he finds that the pointer/needle of the ammeter and voltmeter do not coincide with the zero marks on the scales when circuit is open? No extra ammeter/voltmeter is available in the laboratory.
Answer:
(a) (i) 10 mA and 0.1 V
(ii) V = 2.4 volt, I = 250 mA = 0.25 A
From Ohm’s law. R = \(\frac { V }{ I }\) = \(\frac { 2.4 }{ 0.25 }\) = 9.6 Ω
(b) The coiled wire of tungsten increases the surface area of the wire in very less space so as to emit more light and helps in glowing with more intensity.
(c) The straight line in the graph signify that potential difference and current are directly proportional to each other.
The method of determining resistance of resistor using the graph is by Ohm’s law,
V = IR and by calculating the slope from the points mentioned on the graph.
OR
This is called the zero error of the scale of ammeter or voltmeter. If there is a zero error then this error is subtracted from the value that depicts when the circuit is closed otherwise accurate reading will not be recorded.
Question 15.
A student noticed that an organism by mistake was cut in two parts. After sometime both the parts developed into new individuals. (4)
(a) Name the mode of reproduction used by the organism.
(b) State the type of cells which carry this process.
(c) Write examples of two organisms which multiply by this process.
OR
Ram and Shyam went for a trip to Botanical garden. They saw some plants with beautifully coloured and scented flowers. They wondered why some flowers were beautifully coloured and scented. Then they saw in a flower bed, rose plants with same coloured flowers and of same size. Next day when they went to school they asked teacher about that.
(i) Why flowers are beautifully coloured and scented?
(ii) Why all the flowers in the flower bed were of same size and colour?
Answer:
(a) Regeneration method of asexual mode of reproduction.
(b) Specialised regenerative cells.
(c) Planaria and Hydra multiply by this process.
OR
(i) Flowers are beautifully coloured and scented to attract insects for pollination. Pollination would lead to fertilisation and finally formation of fruits and seeds.
(ii) Rose plants might have propagated by vegetative propagation so, they resemble their parents i.e., all the rose plants are of same size and of same colour.