These Class 8 Science Curiosity Worksheet and Class 8 Science Chapter 9 The Amazing World of Solutes, Solvents, and Solutions Worksheet with Answers are excellent for board exam practice.
Class 8 Science Chapter 9 The Amazing World of Solutes, Solvents, and Solutions Worksheet
Class 8 The Amazing World of Solutes, Solvents, and Solutions Worksheet
Worksheet On The Amazing World of Solutes, Solvents, and Solutions Class 8 – The Amazing World of Solutes, Solvents, and Solutions Worksheet Class 8
Let’s Begin
A. Multiple-Choice Questions
Question 1.
Nitrogen is considered the solvent in air instead of oxygen as it is
(a) Heavier
(b) Present in a larger amount
(c) Able to dissolve oxygen
(d) A gas
Question 2.
What is the solution called when no more solute can dissolve at agivn temperature?
(a) Unsaturated solution
(b) Dilute-solution
(c) Saturated solution
(d) Concentrated solution
Question 3.
Why does oil float on water?
(a) Oil is heavier than water.
(b) Oil dissolves in water.
(c) Oil is less dense than water.
(d) Water is lighter than oil.
Question 4.
The question below consists of an Assertion (A) and a Reason (R). Choose the appropriate answer.
Assertion (A): Gases are more compressible than solids and liquids.
Reason (R): The particles of gases are loosely packed and have large spaces between them.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Question 5.
The question below consists of an Assertion (A) and a Reason (R). Choose the appropriate answer.
Assertion (A): Hydro-alcoholic extracts of herbs are used in traditional medicine.
Reason (R): Water has long been used as a solvent in traditional Indian medicine.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
B. State True (T) or False (F).
1. A solution is a uniform mixture of a solute and a solvent.
2. Concentrated solutions contain more solute than dilute solutions.
3. The solubility of gases increases with rising temperature.
4. A measuring cylinder is used to measure the volume of liquids.
5. Hot air rises because it is denser than cold air.
![]()
C. Fill in the blanks.
1. Applying ___________ has negligible effect on the density of solids.
2. The solubility of most solid solutes ___________ with increasing temperature.
3. Fish can survive underwater because of ___________ gas dissolved in water.
4. The SI unit of density is ___________.
5. The ___________ of an object can be measured using a digital weighing balance.
D. Assign one word to the following terms.
1. A solution that contains a small amount of solute. ___________
2. The force with which the Earth attracts an object towards itself. ___________
3. The comparison of the density of a substance with the density of water. ___________
4. The outermost and lightest layer of the Earth. ___________
5. The temperature at which density of water is highest. ___________
E. Match the Columns.
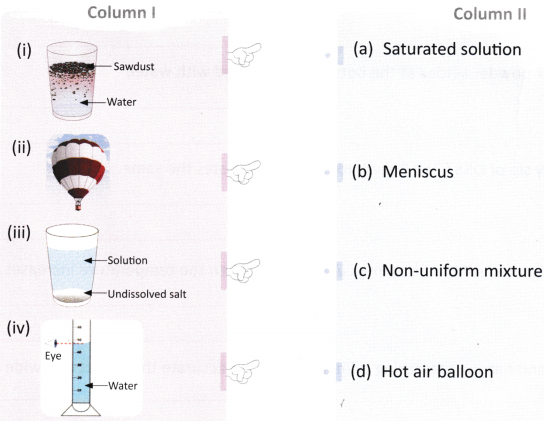
Question 1.
Match the images given in Column I with the terms given in Column II.
Question 2.
Match Column I with Column II.
| Column I | Column II |
| (i) Uniform mixture | (a) Gaseous solution |
| (ii) Air | (b) Measured in newtons |
| (iii) Weight | (c) Measures mass |
| (iv) Two-pan balance | (d) Awarded for Chemical science |
| (v) Asima Chatterjee | (e) Oral Rehydration Solution |
Wonder Why?
A. Give reasons for the following.
1. A raw whole egg sinks in water but floats in salty water.
___________________________________
___________________________________
2. Chalk powder settles at the bottom when mixed with water.
___________________________________
___________________________________
3. Every sip of ORS (Oral Rehydration Solution) tastes the same.
___________________________________
___________________________________
4. A saturated solution becomes unsaturated when the temperature increases.
___________________________________
___________________________________
5. Tall and narrow measuring cylinders are more accurate than short and wide ones.
___________________________________
___________________________________
6. Relative density expressed as a number without any units.
___________________________________
___________________________________
7. Ice floats on water, even though it is solid.
___________________________________
___________________________________
8. Hot air rises above cool air.
___________________________________
___________________________________
![]()
B. Observe the picture and answer the following questions.
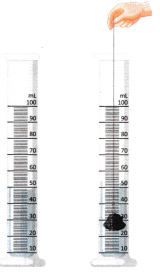
Question 1.
What is being measured using the measuring cylinder in this activity?
___________________________________
___________________________________
Question 2.
This method is suitable for which type of solids?
___________________________________
___________________________________
Question 3.
What does the rise in water level represent?
___________________________________
___________________________________
Question 4.
What should we do if the result is required in cubic centimetres (cm3) instead of millilitres (mL)?
___________________________________
___________________________________
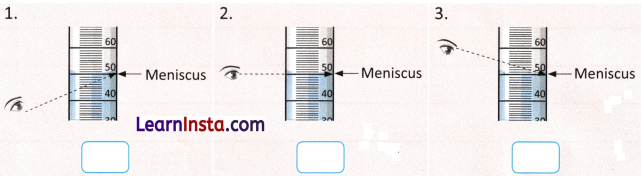
C. Observe the images given below and tick (S) the correct way to measure the volume of the solution.
D. Complete the table below by identifying solute, solvent, and the type of solution. One example has been done for you.
| Solution | Solvent | Solute | Type of Solution |
| Saltwater | Water | Salt | Solid in liquid |
| Carbonated drink | |||
| Air | |||
| Vinegar | |||
| Brass | |||
| Choshni |
E. Read the following paragraph and choose the correct word. Rewrite the paragraph in the space provided.
While shopping for groceries, Anish picked up a 1-litre packet of ghee and noticed that it weighed only 910 grams. He was curious as he remembered that 1 litre of water weighs 1000 grams/1000 kilograms. This reminded him the concept of density/pressure, which is the mass/width of a substance per unit volume/area. Anish realised that this meant ghee is less/more dense than water. His teacher had also explained the concept of relative density, which is the density of a substance compared to the density of oil/water. Since the relative density of ghee is less than 1, it would float/sink in water. Anish now understood that even though both ghee and water packets are the same in volume/weight, they differ in mass/ pressure. Therefore, packet of ghee is felt lighter as it has less mass per unit volume.
___________________________________
___________________________________
___________________________________
___________________________________
Learn By Doing
A. Take a clean glass and fill it halfway (100 mL) with water. Add one teaspoon of salt and stir until it dissolves completely. Keep adding more teaspoons of salt, one at a time, stirring well after each addition. Continue until you notice that some salt no longer dissolves and starts settling at the bottom of the glass. Fill in the observation table below. Then, write a short conclusion based on your results—how many teaspoons of salt dissolved, and what it tells you about solubility and saturation.
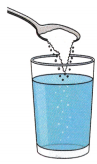
| Amount of Salt Added (Teaspoon) | Observation (Dissolves / Does Not Dissolve) |
| One | |
| Two | |
| Three | |
| Four | |
| Five | |
| – |
Conclusion:
___________________________________
___________________________________
___________________________________
B. Take about 60 mL of water in a glass beaker and measure its temperature using a thermometer (around 30 °C). Add a spoonful of baking soda and stir until it dissolves. Keep adding more baking soda in small amounts while stirring, until some remains undissolved at the bottom. Now gently heat the beaker to around 60 °C while continuing to stir. Observe if the undissolved baking soda dissolves. Add more baking soda again until it stops dissolving. Then heat the mixture further to about 80 °C and observe once more. Write your observations and then conclude how temperature affects the solubility of baking soda. (Perform this activity with the help of your teacher.)

___________________________________
___________________________________
___________________________________
![]()
Explore With Curiosity
A. Riya visits a pond during summer and notices some dead fish floating on the surface. She wonders why the fish might have died. What could be a reason for the death of the fish?

___________________________________
___________________________________
___________________________________
B. In science class, the teacher gave Arjun and Neha a wooden ball and an iron ball of the same size. When both were placed in a bucket of water, Wooden ball floated but the iron ball sank. Why did the iron ball sink and the wooden ball floated, even though they were the same size?

___________________________________
___________________________________
___________________________________
C. During a school trip to a village, the students saw a fisherman using a boat made from bamboo and wooden logs. The guide said, “These boats have been used for centuries.” What properties of bamboo and wood make them suitable materials for building such boats?
___________________________________
___________________________________
___________________________________
D. In the laboratory, Neha observes that the measuring cylinder has 10 small divisions between the 30 mL and 40 mL marks. What volume does each small division represent? What is the smallest volume this cylinder can measure?
___________________________________
___________________________________
___________________________________
E. Read the conversation between Riya and Rahul and fill in the blanks with the words given in the box.
| Mass, Switch ON, Liquid, Zero, Dry and clean; 16.400 g, Density. Reset. Beaker, Tare |

F. Ayan needs to measure exactly 70 mL of water for an experiment. He has the following measuring cylinders: 50 mL (smallest division: 1 mL), 100 mL (smallest division: 1 mL) and 250 mL (smallest division: 2 mL). Which measuring cylinder should Ayan choose and why?
___________________________________
___________________________________
___________________________________
G. A water tank is shaped like a cuboid with dimensions: Length = 100 cm, Width = 40 cm, Height = 50 cm. Calculate its volume in litres.
___________________________________
___________________________________
___________________________________
H. A tank can hold 250 litres of liquid. A factory wants to fill it with a chemical that has a density of 1.4 g/cm3. What will be the total mass of the chemical in kilograms?
___________________________________
___________________________________
___________________________________
Word Puzzle
Complete the crossword with the help of the given clues.
DOWN
1. The space occupied by an object or liquid
2. The maximum solute that can dissolve in a solvent at a given temperature
3. The curved surface of a liquid in a measuring cylinder
4. A mixture in which all the components are evenly distributed
ACROSS
2. The substance that dissolves in a solvent
5. Mass per unit volume of a substance
6. Amount of solute in a fixed quantity of solution
7. Amount of matter in an object

![]()
Suggested Activities
A. Observe different household liquids like oil, water, vinegar, and syrup. Predict which of these will mix to form solutions and which will not. Then, test your predictions by mixing small amounts of each and record your observations in a chart.
B. Find out why there is no aquatic life in the Dead Sea. Investigate whether there are other water bodies around the world with similar conditions. Prepare a short report or presentation summarising your findings and share it with your class.