Students rely on Class 8 Science Extra Questions and Class 8th Curiosity Chapter 7 Particulate Nature of Matter Important Extra Question Answer to improve their grades.
Class 8 Science Chapter 7 Particulate Nature of Matter Extra Questions
Class 8 Science Chapter 7 Extra Questions on Particulate Nature of Matter
Particulate Nature of Matter Class 8 Very Short Question Answer
Question 1.
What are constituent particles of matter?
Answer:
Constituent particles are the basic units that make up a substance.
Question 2.
Is grinding chalk a physical or chemical change?
Answer:
Physical change.
![]()
Question 3.
Why do solids have a fixed volume?
Answer:
Solids have a fixed volume because their particles are closely packed.
Question 4.
How do sugar particles behave when dissolved in water?
Answer:
Sugar particles break into constituent particles and spread evenly in water.
Question 5.
What is meant by ‘interparticle space’?
Answer:
The gaps or spaces between particles of a substance.
Question 6.
What force holds particles together in matter?
Answer:
Interparticle forces of attraction.
Question 7.
Who first proposed the idea of the atom (Parmanu)?
Answer:
Acharya Kanad.
![]()
Question 8.
Do liquids have a fixed shape?
Answer:
No, liquids take the shape of their container.
Question 9.
Which state of matter has the strongest interparticle attraction?
Answer:
Solid.
Question 10.
Name the process by which a solid changes into a liquid.
Answer:
Melting.
Particulate Nature of Matter Class 8 Short Question Answer
Question 1.
Write the melting point of the following substances.
(i) Ice
(ii) Urea
(iii) Iron
Answer:
(i) Ice -0°C
(ii) Urea -133°C
(iii) Iron – 1538°C
![]()
Question 2.
What is evaporation? How is it different from boiling?
Answer:
Evaporation is the slow formation of vapour from the surface of a liquid at all temperatures. Boiling occurs rapidly at the boiling point only and throughout the liquid.
Question 3.
Explain why water takes the shape of its container but retains the same volume.
Answer:
Water is a liquid. Its particles are loosely packed and have weaker interparticle forces of attraction compared to solids. This allows them to move freely, but only within a limited space. As a result, water does not have a fixed shape, but it maintains a fixed volume.
Question 4.
What happens to the volume of the mixture when sand is added to water?
Answer:
Sand is insoluble solid that does not dissolve in water. When added to water, the sand particles settle down and occupy some space in the container, causing the total volume to increase.
Question 5.
How does soap help in removing oil stains from clothes?
Answer:
When we wash oily clothes with soap, many tiny soap particles surround the oil particles on the fabric. Each soap particle has two ends. One end of each soap particle sticks to the oil, while the other end mixes with water. This allows the soap to lift the oil from the fabric and wash it away.
Particulate Nature of Matter Class 8 Long Question Answer
Question 1.
What are interparticle forces? How do they influence the physical states of matter?
Answer:
Interparticle forces are the attractive forces that hold the constituent particles of matter together. In solids, these forces are very strong, keeping the particles tightly packed and fixed in position. In liquids, the forces are weaker, allowing particles to move around but still stay close. In gases, the forces are negligible, so particles move freely and are far apart. The strength of these forces determines whether matter exists as a solid, liquid, or gas.
![]()
Question 2.
What happens to the movement of particles in each case when potassium permanganate is added to hot water, water at room temperature, and cold water?
Answer:
The movement of water particles depends on the temperature. It increases when heat is provided. In hot water, the particles move faster because they have more heat energy. In water at room temperature, the particles move at a medium speed, and in cold water, they move slowly due to less heat energy. When potassium permanganate is added:
- It spreads the fastest in hot water.
- It spreads slowly in water at room temperature.
- It spreads the slowest in cold water.
Thus, faster the water particles move, the quicker they help spread the potassium permanganate throughout the water.
Question 3.
Explain how the thermal energy of particles affects the physical state of matter.
Answer:
The physical state of matter depends on the thermal (heat) energy of its particles.
- In the solid state, the thermal energy of particles is very low. As a result, particles remain tightly packed and attractive forces between them are very strong. They only vibrate in their fixed positions.
- At the melting point, when heat is supplied, the thermal energy of the particles increases. This extra energy helps the particles overcome the attractive forces between particles, allowing them to move slightly apart. At this stage, the solid changes into a liquid, where the particles are still close but can move around each other.
- In the gaseous state, the particles have high thermal energy to overcome the forces of attraction between them and they can move freely in all directions.
Particulate Nature of Matter Class 8 Skill-Based Questions
Question 1.
Observe the given image and answer the following questions.
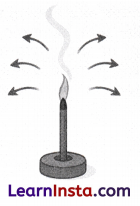
(i) What is shown in the image?
(ii) Will the fragrance of the incense stick spread throughout the room?
(iii) What property of matter does this demonstrate, and which state of matter is involved?
Answer:
(i) The image shows a burning incense stick.
(ii) Yes, the fragrance of the incense stick will spread throughout the room after some time.
(iii) This demonstrates the property of diffusion in gases. The state of matter involved is gas, as gas particles move randomly in all directions due to negligible interparticle forces of attraction.
![]()
Question 2.
Observe the given image and answer the following questions.

(i) What is shown in the image?
(ii) What can you conclude about the state of water?
Answer:
(i) The image shows water poured into containers of different shapes.
(ii) Water is in the liquid state. This activity demonstrates that liquids do not have a fixed shape but have a definite volume. Liquids take the shape of the container they are poured into but their volume remains unchanged.
Particulate Nature of Matter Class 8 Quiz Questions
Question 1.
What process is involved when a solid changing into a liquid upon heating?
Answer:
Melting
Question 2.
What is the process of a liquid changing into gas at the boiling point called?
Answer:
Boiling
![]()
Question 3.
What is the state of matter in which particles are tightly packed?
Answer:
Solid
Question 4.
What is the full form of SPM?
Answer:
Suspended Particulate Matter
Question 5.
Which physical state of matter has maximum interparticle spacing?
Answer:
Gas
Question 6.
Do liquids have a fixed volume?
Answer:
Yes, liquids have a fixed volume
Question 7.
What kind of space exists between particles of matter?
Answer:
Interparticle space
Question 8.
What is the term used for substances like liquids and gases that can flow?
Answer:
Fluids
![]()
Question 9.
Name the stable particle formed when the two atoms of hydrogen combine.
Answer:
A molecule of hydrogen gas
Question 10.
What happens to particles of a solid when heated?
Answer:
They vibrate more vigorously
Particulate Nature of Matter Class 8 Case Based Questions
Question 1.
Aman poured 200 mL of water into a glass. Then he transferred the water into a bowl, and finally into a flat dish. He noticed that the water took the shape of each container but the amount of water remained the same.
I. Which state of matter is being described here?
II. Why did the volume of the water not change in different containers?
III. What would happen if this experiment were done with a solid object like a stone?
OR
If this experiment is repeated with honey in place of water, then what would be the observations?
Answer:
I. Liquid
II. This is because the interparticle force of attraction in liquids is strong enough to keep the volume fixed.
III. The stone would retain its own shape and would not change according to the container.
OR
If this experiment is repeated with honey then observations will be the same as that of water as honey is also a liquid.
![]()
Question 2.
Sonal lit an incense stick in one corner of the room. After some time, her sister, sitting in another corner, also started smelling the fragrance. Sonal wondered how the fragrance travelled across the room even though she did not see anything moving.
I. Which state of matter is involved in the spreading of fragrance?
II. Why is the fragrance able to travel across the room?
III. What does this observation tell us about properties of gas particles?
OR
Gas particles flow from one place to another, what is this property called?
Answer:
I. Gas
II. This is because gas particles move freely and quickly in all directions.
III. Gas particles are in constant motion and have very weak interparticle forces of attraction.
OR
This property is called fluidity.
Particulate Nature of Matter Extra Questions for Practice
Multiple Choice Questions
Question 1.
Liquids have
(a) a fixed shape and fixed volume
(b) no fixed shape but fixed volume
(c) no fixed shape and no fixed volume
(d) a fixed shape but no fixed volume.
![]()
Question 2.
Which of the following statements is true about interparticle attractions?
(a) They are strongest in gases.
(b) They do not affect the state of matter.
(c) They are strongest in solids and weakest in gases.
(d) They are the same in solids, liquids, and gases.
Question 3.
In which state of matter do particles vibrate but do not move freely?
(a) Solid
(b) Liquid
(c) Gas
(d) All of these
Question 4.
Read the given statements and select the correct option.
Assertion (A): Gases are highly compressible.
Reason (R): Gases have large interparticle spaces between their particles.
(a) Both A and R are true and R is the correct explanation of A .
(b) Both A and R are true but R is not the correct explanation of A .
(c) A is true but R is false.
(d) A is false but R is true.
![]()
Question 5.
Match the terms given in Column A with their correct descriptions given in Column B.
Column A — Column B
(i) Solid — (p) Definite volume but no fixed shape
(ii) Liquid — (q) Do not retain their fixed shape
(iii) Gas — (r) Particles can only move to and fro about their positions
(iv) Fluids — (s) Particles are free to move
(a) (i)-(s), (ii)-(r), (iii)-(q), (iv)-(p)
(b) (i)-(s), (ii)-(p), (iii)-(q), (iv)-(r)
(c) (i) -(r),(ii)-(p),(iii)-(s),(iv)-(q)
(d) (i) -(q),(ii)-(r),(iii)-(s),(iv)-(p)
Very Short Answer Type Questions
Question 1.
What is the name of the force that holds the particles of matter together?
Question 2.
Which solid dissolves in water: sugar or sand?
Question 3.
What is matter made up of?
![]()
Question 4.
Name the state of matter having maximum interparticle spaces.
Question 5.
Is grinding chalk into fine powder a physical or chemical change?
Short Answer Type-I Questions
Question 1.
Why are solids hard and rigid?
Question 2.
What are constituent particles of matter?
Question 3.
Describe the following terms:
(a) Melting point
(b) Boiling point
Question 4.
Explain how heating affects the movement of particles in a solid.
Question 5.
Why does potassium permanganate spread faster in hot water compared to cold water?
Short Answer Type-II Questions
Question 1.
If you place a burning incense stick in one corner of a room, why does its smell spread everywhere?
Question 2.
Write true (T) or false (F) against the following statements. Rewrite the correct statement.
(i) Water has a definite shape and a definite volume.
(ii) Evaporation happens only at the boiling point of a liquid.
(iii) Particles of solids can move freely in all directions.
![]()
Question 3.
Draw diagrams representing the particles present in the following substances:
(i) Gold
(ii) Water
(iii) Oxygen gas
Question 4.
During an experiment, a student observes that after sugar dissolves in water, the water level does not increase significantly. Why?
Question 5.
What was the belief of the ancient Indian philosopher Acharya Kanad about “Parmanu”?
Long Answer Type Questions
Question 1.
Differentiate between the particulate nature of the three states of matter.
![]()
Question 2.
Describe an activity to show that gases do not have a fixed volume.
Question 3.
Explain the changes that occur in the movement of particles during the conversion of a liquid into a gas.
Suggested Projects And Activities
Question 1.
Take three beakers and fill each with the same amount of water – one cold, one at room temperature, and one hot. Add an equal amount of sugar to each beaker and observe how long it takes to dissolve in each case. Record your observations and prepare a short report based on your findings.
![]()
Question 2.
Interview a chemistry teacher, lab technician, or science professional to understand how the knowledge of states of matter is useful in everyday life (For example, cooking, weather forecasting, or industrial design). Based on the interview, create a short write-up, video presentation, or poster to share what you learned.