These Class 8 Science Curiosity Worksheet and Class 8 Science Chapter 8 Nature of Matter: Elements, Compounds, and Mixtures Worksheet with Answers are excellent for board exam practice.
Class 8 Science Chapter 8 Nature of Matter: Elements, Compounds, and Mixtures Worksheet
Class 8 Nature of Matter: Elements, Compounds, and Mixtures Worksheet
Worksheet On Nature of Matter: Elements, Compounds, and Mixtures Class 8 – Nature of Matter: Elements, Compounds, and Mixtures Worksheet Class 8
Let’s Begin
A. Multiple-Choice Questions
Question 1.
Look at the chemical equation below, where water breaks down to give two products.
Water → Hydrogen + Oxygen
Based on this information, which of the following statements is correct?
(a) Water is an element, and Hydrogen and Oxygen are compounds.
(b) Water is a compound, and Hydrogen and Oxygen are elements.
(c) Water, Hydrogen, and Oxygen are all compounds. .
(d) Water is a mixture, and Hydrogen and Oxygen are elements.
Question 2.
Which of the following is a mixture?
(a) Water
(b) Air
(c) Gold
(d) Calcite
Question 3.
Which of the following is not made up of matter?
(a) Chair
(b) Heat
(c) Water
(d) Pencil
Question 4.
Which of the following is a pure substance?
(a) Air
(b) Soil
(c) Oxygen
(d) Lemonade
Question 5.
Which of the following is a metal?
(a) Aluminium
(b) Carbon
(c) Sulphur
(d) Hydrogen
Question 6.
This question consists of an Assertion (A) and a Reason (R). Read the Assertion and Reason and choose the appropriate answer.
Assertion (A): Water is an element.
Reason (R): Water is made up of hydrogen and oxygen.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Question 7.
This question consists of an Assertion (A) and a Reason (R). Read the Assertion and Reason and choose the appropriate answer.
Assertion (A): Sodium chloride dissolved in water is a mixture.
Reason (R): Sodium chloride can be separated from water by physical means like evaporation.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
B. State True (T) or False (F).
1. Pyroxene is made up of only one kind of element.
2. Elements and compounds are the building blocks of all matter.
3. The components of a mixture reacts chemically with each other.
4. A compound cannot be separated by physical methods.
5. Nitrogen constitutes about 78% of the air and helps in combustion.
6. All non-metals are gases at room temperature.
![]()
C. Fill in the blanks.
1. Compounds are formed by combination ofelementš in fixed ratios.
2. Elements that have properties between metals and non-metals are called
3. Sugar decomposes on heating to give and water.
4. The gas evolved when dilute hydiochloric acid reacts with iron is gas.
5. At present, there are total ____ elements known.
6. The gas is colourless and has a rotten egg-like odour.
7. is an illegal process that reduces the quality of a product.
D. Assign one word to the following terms.
1. A mixture of two or more metals, or a metal with a non-metal.
2. A gas present in air that supports burning.
3. A substance made of same type of particles.
4. A stable particle formed by the combination of two or more atoms.
5. The lightest material on the Earth made of carbon.
6. A mixture of two or more metals having properties distinct from its constituent metals.
E. Match the elements given in Column I with their correct type given in Column II.
| Column I | Column II |
| (i) Mercury | (a) Native mineral |
| (ii) Gold | (b) Liquid metal |
| (iii) Oxygen | (c) Metalloid |
| (iv) Bromine | (d) Non-metal |
| (v) Silicon | (e) Liquid non-metal |
Wonder Why?
A. Give reasons for the following.
1. Water is considered a pure substance, but milk is not.
________________________________
________________________________
2. Mixtures can be separated by physical methods, but compounds cannot.
________________________________
________________________________
3. We can use magnets to separate iron filings from a mixture of sand and iron.
________________________________
________________________________
4. A compound has different properties than its constituent elements.
________________________________
________________________________
5. We can see the different substances in a non-uniform mixture but not in a uniform.
________________________________
________________________________
6. Silver is known as native mineral, but quartz is not.
________________________________
________________________________
7. Gases are not water vapour.
________________________________
________________________________
B. Mention the type of mixture depending on the physical state of their components and complete the table given below. One example has been done for you.
| Examples | Type of mixture | Uniform/Non-uniform |
| 1. Sugar and water Solid and liquid Uniform | ||
| 2. Oil and water | ||
| 3. Aerated water (soda water) | ||
| 4. Air – | ||
| 5. Sand and water | ||
| 6. Fruit salad | ||
| 7. Oxygen dissolved in water | ||
| 8. Carbon particles in the air | ||
| 9. Baking powder (baking soda and tartaric acid) | ||
| 10. Lemonade |
![]()
C. Read the paragraph and answer the following questions.
Everything around us is made up of matter, but matter does-not always appear the same. Some substances contain only one type of atom, while others.form when different atoms join together. For example, water is formed when hydrogen and oxygen atoms combine in a fixed ratio. Sometimes, different substances are mixed, but they retain their own properties. For example, the air we breathe is a mixture of several gases, each with its own characteristics. Alloys are special mixtures of metals, or metals and non-metals, which can be found naturally or made by humans to improve strength or usefulness, such as brass (copper and zinc). The Earth contains naturally occurring solid materials called minerals. Some minerals are made of a single element, while others are combinations of different elements.
Question 1.
What kind of substances are made of only one type of atom?
________________________________
________________________________
Question 2.
Name a compound mentioned in the paragraph, and its elements.
________________________________
________________________________
Question 3.
What type of mixture is air, and what does it contain?
________________________________
________________________________
Question 4.
Give an example of a mineral that is an element.
________________________________
________________________________
Question 5.
What is an alloy? Give an example.
________________________________
________________________________
D. Look at the picture and answer the following questions.

Question 1.
Name the art given in the picture. Which metals are used to make it?
________________________________
________________________________
Question 2.
Which material is used first to shape the design in this art form?
________________________________
________________________________
Question 3.
From which states of India does this art form belong?
________________________________
________________________________
Question 4.
How does the sculpture get its golden shine and strength?
________________________________
________________________________
E. Look around your home, classroom, or school for different objects and write their correct type and uses in the following table. One example has been done for you.
| Material | Type (Element/Compound/Mixture) | Uses |
| Water | Compound | Drinking, cooking, etc. |
Learn By Doing
A. Take a glass tumbler and fill it halfway with clean water. Slowly add a small amount of calcium oxide (quick lime) and stir well. Calcium oxide reacts with water to form calcium hydroxide (lime water) and releases heat. Filter this solution into a clean beaker and observe its colour. Pour the clear solution into a petri dish and leave it open for a few hours, stirring occasionally. This experiment helps to show the presence of which gas in the air? Write your observations for each step in the given space. Also, write the chemical equation for the reaction.

________________________________
________________________________
________________________________
________________________________
B. Take a teaspoon of sugar and place it in a boiling tube. Heat it gently over the flame using a test tube holder. As the sugar heats, observe the changesin its colour, smell and state. (Perform this activity only under the supervision of a teacher.) Observe/Experiment

(a) Heating of sugar
(b) Colour of the sugar on heating
(c) Charcoal placed on a watch glass
Write your observations in the given table and give conclusion for this experiment.
| Steps | Observations |
| Appearance of sugar before heating | |
| Colour change as it is heated | |
| Appearance of droplets near mouth of tube (Yes/No) | |
| Final residue left behind | |
| Any change in smell |
Conclusion:
________________________________
________________________________
C. Take a glass beaker and fill it two-thirds with clean water. Add a few drops of dilute sulphuric acid and stir gently. Fill two small test tubes completely with this water. Now, place a 9 V battery inside the beaker and invert each test tube over one of its terminals.
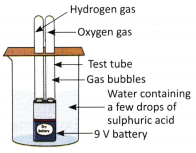
Wait for a few minutes and observe the formation of gas bubbles. After 10-15 minutes, observe the volume of gas collected in each test tube. Remove the test tubes one by one and bring a burning candle near their openings to test the gases. This experiment helps to show the constituents of water.

Write your observations for each step in the given space. Also, write the.chemical equation forthereaction.
| Steps | Observations | Conclusions |
| Burning candle near test tube I | ||
| Burning candle near test tube II |
Chemical equation for the reaction: ________________________________
D. Take 5.6 g of iron filings and 3.2 g of sulphur powder in a watch glass. Mix them thoroughly. This mixture is labelled as Sample A. Take half of Sample A in a China dish and heat it gently while stirring continuously. A black mass will form. Let it cool, grind it, and place it on another watch glass. Label it as Sample B.

This experiment helps to understand the difference between a mixture and a compound formed by the combination of elements.
Write your observations for each step in the table. Also, identify the kind of change that occurre(d)
| Experiment | Observations | Conclusions | ||
| Sample A | Sample B | Sample A | Sample B | |
| 1. Appearance (i) Colour (ii) Texture |
||||
| 2. Magnet test | ||||
| 3. Gas test (with dilute HCI) |
||||
Chemical equation for sample A: ________________________________
Chemical equation for sample B: ________________________________
![]()
Explore with Curiosity
A. During Diwali, Tanya places a clean black paper on her window in the evening. By the next morning, she finds it covered in fine ash and dust. She repeats the same activity after a week and finds the paper mostly clean. Why do you think the results of the experiment changed before and after Diwali?
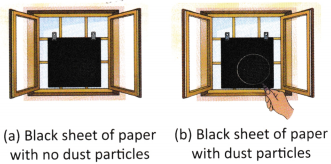
________________________________
________________________________
________________________________
________________________________
B. Kunal is making a model of oxygen using clay balls. He joins two oxygen atoms to form a molecule, just like he saw in his textbook diagram. His cousin asks, “Can oxygen exist as a single atom?” Why do you think oxygen atoms combine in pairs to form molecules? What is the benefit of this pairing?

________________________________
________________________________
________________________________
________________________________
C. Riya attends a science fair where she sees a black, sponge-like object placed gently on a blade of grass. It looks solid but seems extremely light and porous. The teacher tells her, it is made of carbon and can absorb large amounts of oil. What is this material, and what kind of properties do you think this material has? Why might it be helpful in cleaning up oil spills?

________________________________
________________________________
________________________________
________________________________
D. At a science fair, Aman reads a poster that says: “Adding soap to milk increases its foam and makes it look fresh.” He is shocked to learn that some sellers do this to deceive customers.

What is this practice called? What does it mean, and how can it affect human health?
________________________________
________________________________
________________________________
________________________________
E. Kabir hears his grandmother say, “In our time, we used to store water in bronze vessels—it kept us healthy.” When Kabir asks his teacher, she explains that this practice dates back to when alloys like Kamsya were widely use(d) Which two metals are mixed to make Kamsya (bronze)?

________________________________
________________________________
________________________________
________________________________
Word Puzzle
Complete the crossword with the help of the given clues.
ACROSS
1. A uniform mixture of two or more metals
3. A naturally occurring solid substance found on the Earth
7. A combination of two or more substances
8. Unwanted or harmful substances present in the environment
DOWN
1. An illegal process of the addition of low-quality substances to products
2. Anything that has mass and occupies space
4. A pure substance made of only one kind of atom
5. A pure substance formed when two or more elements combine chemically
6. An element that has properties of both metals and non-metals
![]()
Suggested Activities
A. Write a short paragraph starting with “If I were an element, I would be…” Describe where you are found in nature, what you look like, how people uses you, and why you are important in daily life. Present your writing to the class in a fun storytelling format.
B. Observe different objects around your home or school and classify them as elements, compounds, or mixtures. For example, aluminium foil, water, air, and stainless steel. Create a three-column chart to display your observations and classifications.
C. Bring simple mixtures such as salt and sand, iron filings and rice, salt solution, sand and stones. Try to separate them using methods such as filtration, evaporation, magnetic separation, or sieving. Record the method used and the result of each separation. Explain why these methods work only for mixtures and not for compounds in your class.