Practicing Class 8 Science MCQ and Class 8 Science Chapter 8 Nature of Matter: Elements, Compounds, and Mixtures MCQ Questions Online Test with Answers daily helps in time management.
Class 8 Nature of Matter: Elements, Compounds, and Mixtures MCQ with Answers
MCQ on Nature of Matter: Elements, Compounds, and Mixtures Class 8
Class 8 Science Chapter 8 Nature of Matter: Elements, Compounds, and Mixtures MCQ Questions with Answers
Question 1.
Which of the following statements are true for pure substances? (NCERT Exemplar)
(i) Pure substances contain only one kind of particles
(ii) Pure substances may be compounds or mixtures
(iii) Pure substances have the same composition throughout
(iv) Pure substances can be exemplified by all elements other than nickel
(a) (i) and (ii)
(b) (i) and (iii)
(c) (iii) and (iv)
(d) (ii) and (iii)
Answer:
(b) (i) and (iii)
![]()
Question 2.
Pick up the odd one out.
(a) Brass
(b) Air
(c) Sand
(d) Graphite
Answer:
(d) Graphite
Question 3.
Graphene aerogel is used to clean oil spills because it is
(a) a liquid
(b) reactive
(c) highly porous and absorbent
(d) a compound of nitrogen
Answer:
(c) highly porous and absorbent
Question 4.
Match the following types of solutions with examples and select the correct option.
Column A — Column B
(i) Solid in liquid — (a) Air
(ii) Gas in liquid — (b) Vinegar
(iii) Liquid in liquid — (c) Soda water
(iv) Gas in gas — (d) Salt in water
(a) (i)-(a), (ii)-(b), (iii)-(c), (iv)-(d)
(b) (i)-(c), (ii)-(d), (iii)-(a), (iv)-(b)
(c) (i)-(b), (iz)-(a), (iii)-(d), (iv)-(c)
(d) (i)-(d), (ii)-(c), (iii)-(b), (iv)-(a)
Answer:
(d) (i)-(d), (ii)-(c), (iii)-(b), (iv)-(a)
![]()
Question 5.
A student is asked to make a uniform mixture. He is provided with the following substances.
A. Water
B. Soil
C. Milk
D. Chocolate powder
E. Salt
Which two substances should the student mix to form a uniform mixture?
(a) A and B
(b) B and D
(c) B and E
(d) A and E
Answer:
(d) A and E
Question 6.
Which of the following statements is not true about an atom? (NCERT Exemplar)
(a) Atoms are not able to exist independently.
(b) Atoms are the basic units from which molecules and ions are formed.
(c) Atoms are always neutral in nature.
(d) Atoms aggregate in large numbers to form the matter that we can see, feel or touch.
Answer:
(a) Atoms are not able to exist independently.
Question 7.
Observe the properties of substance X.
(i) It has a fixed composition throughout the substance.
(ii) Its properties are different from its constituents.
(iii) It can be divided into smaller parts by chemical methods.
(iv) It is made of two different kinds of atoms.
What can substance X be classified as?
(a) Element, because it has different properties.
(b) Element, because it can be divided into smaller parts.
(c) Compound, because it can change from one form to another.
(d) Compound, because it is made of two kinds of atoms joined in a definite proportion.
Answer:
(d) Compound, because it is made of two kinds of atoms joined in a definite proportion.
![]()
Question 8.
Match the types of pure substances with their examples and select the correct option.
Column A — Column B
(i) Mixture — (a) Iron
(ii) Compound — (b) Brass
(iii) Element — (c) Air
(iv) Alloy — (c) Water
(a) (i)-(a), (ii)-(b), (iii)-(c), (iv)-(d)
(b) (i)-(b),(i)-(c),(ii)-(d),(iv)-(a)
(c) (i)-(c), (ii)-(d), (iii)-(a), (iv)-(b)
(d) (i)-(d), (ii)-(a), (iii)-(b),(iv)-(c)
Answer:
(c) (i)-(c), (ii)-(d), (iii)-(a), (iv)-(b)
Question 9.
Two chemical species X and Y combine together to form a product P which contains both X and Y.
X+Y → P
X and Y cannot be broken down into simpler substances by simple chemical reactions. Which of the following concerning the species X, Y and P are correct? (NCERT Exemplar)
(i) P is a compound.
(ii) X and Y are compounds.
(iii) X and Y are elements.
(iv) P has a fixed composition.
(a) (i), (ii) and (iii)
(b) (i), (ii) and (iv)
(c) (ii), (iii) and (iv)
(d) (i), (iii) and (iv)
Answer:
(d) (i), (iii) and (iv)
![]()
Question 10.
A student made models using gumballs. Which model would represent an element?
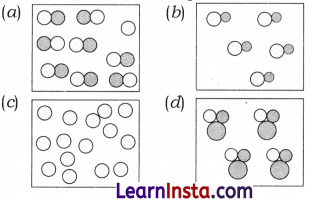
Answer:
(c)
Nature of Matter: Elements, Compounds, and Mixtures Class 8 Assertion-Reason Type Questions
Directions (Q. No. 1 to 4): The question below consists of an Assertion (A) and a Reason (R).
Use the following key to choose the appropriate answer.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Question 1.
Assertion (A): Air is a pure substance.
Reason (R): Air is a uniform mixture of gases like oxygen, nitrogen, and carbon dioxide
Answer:
(d) A is false but R is true
![]()
Question 2.
Assertion (A): A compound is a pure substance that consists of two or more elements chemically combined.
Reason (R): The properties of a compound are the same as the properties of its constituent elements.
Answer:
(c) A is true but R is false
Question 3.
Assertion (A): Graphene is useful in cleaning oil spills.
Reason (R): Graphene aerogel is highly absorbent and very light.
Answer:
(a) Both A and R are true and R is the correct explanation of A
![]()
Question 4.
Assertion (A): All mixtures can be separated into their components by physical methods. Reason (R): Mixtures are formed by chemical combination between substances.
Answer:
(c) A is true but R is false
Nature of Matter: Elements, Compounds, and Mixtures Class 8 Fill in the blanks
Question 1.
A substance that consists of only one kind of particles is called a ………..
Answer:
pure substance
Question 2.
Brass is a solid-solid ……….mixture of copper and zinc.
Answer:
uniform
Question 3.
A mixture of sand and salt is ………..mixture.
Answer:
non-uniform
![]()
Question 4.
The gas that makes a burning splinter burn more brightly is …………
Answer:
oxygen
Question 5.
Silicon is an example of a …………
Answer:
metalloid
Nature of Matter: Elements, Compounds, and Mixtures Class 8 True or False
Question 1.
The components of a mixture can be separated by physical methods.
Answer:
True
Question 2.
Milk is considered a pure substance in science.
Answer:
False
![]()
Question 3.
Compounds have different properties than the elements they are made of.
Answer:
True
Question 4.
A molecule of hydrogen is made up of two hydrogen atoms.
Answer:
True
Question 5.
Iron sulphide reacts with dilute hydrochloric acid to form hydrogen gas.
Answer:
False
![]()
Nature of Matter: Elements, Compounds, and Mixtures Class 8 Match the following
Question 1.
| Column A | Column B |
| (i) Not a matter | (a) Bihar and Odisha |
| (ii) Mixture | (b) Electricity |
| (iii) Element | (c) Made of two or more elements |
| (iv) Dhokra art | (d) Lemon juice |
| (v) Compound | (e) Cannot be broken down |
Answer:
| Column A | Column B |
| (i) Not a matter | (b) Electricity |
| (ii) Mixture | (d) Lemon juice |
| (iii) Element | (e) Cannot be broken down |
| (iv) Dhokra art | (a) Bihar and Odisha |
| (v) Compound | (c) Made of two or more elements |
![]()
Question 2.
| Column A | Column B |
| (i) Graphene | (a) Mixture with same composition throughout |
| (ii) Atom | (b) One-atom thick sheet of carbon atoms |
| (iii) Mineral | (c) Substance that causes harm to environment |
| (iv) Pollutant | (d) Found naturally in Earth’s crust |
| (v) Uniform mixture | (e) Smallest unit of an element |
Answer:
| Column A | Column B |
| (i) Graphene | (b) One-atom thick sheet of carbon atoms |
| (ii) Atom | (e) Smallest unit of an element |
| (iii) Mineral | (d) Found naturally in Earth’s crust |
| (iv) Pollutant | (c) Substance that causes harm to environment |
| (v) Uniform mixture | (a) Mixture with same composition throughout |