Students rely on Class 8 Science Extra Questions and Class 8th Curiosity Chapter 8 Nature of Matter: Elements, Compounds, and Mixtures Extra Question Answer to improve their grades.
Class 8 Science Chapter 8 Nature of Matter: Elements, Compounds, and Mixtures Extra Questions
Class 8 Science Chapter 8 Extra Questions on Nature of Matter: Elements, Compounds, and Mixtures
Nature of Matter: Elements, Compounds, and Mixtures Class 8 Very Short Question Answer
Question 1.
What is the ratio of hydrogen and oxygen in a water molecule?
Answer:
2: 1 (two hydrogen atoms and one oxygen atom)
![]()
Question 2.
What are alloys?
Answer:
The homogeneous mixture of two or more metals or a metal and a non-metal is called an alloy. For example, stainless steel is an alloy of iron, nickel, chromium and a small amount of carbon.
Question 3.
Give one example of a gas in a liquid solution.
Answer:
Cold drinks are made by mixing carbon dioxide gas with water.
Question 4.
Classify brass and diamond as elements and mixtures.
Answer:
Brass – Mixture.
Diamond – Element
Question 5.
Name two metals and two non-metals.
Answer:
Metals: Iron, Aluminium Non-metals: Oxygen, Sulphur
Question 6.
What is formed when iron and sulphur are heated together?
Answer:
Iron sulphide (a compound)
![]()
Question 7.
Which gas makes a candle flame glow brighter- hydrogen or oxygen?
Answer:
Oxygen
Question 8.
Which gas released from water produces a ‘pop’ sound on burning?
Answer:
Hydrogen gas
Question 9.
Sucrose (sugar) crystals obtained from sugarcane and beetroot are mixed together. Will it be a pure substance or a mixture? Give reasons for the same. (NCERT Exemplar)
Answer:
It will be a pure substance because, irrespective of the source, the chemical composition of sugar crystals would be the same.
Question 10.
Classify the following into element or compound:
(a) Carbon
(b) Carbon dioxide.
Answer:
(a) Carbon – Element
(b) Carbon dioxide – Compound
![]()
Question 11.
What happens to sugar when it is heated?
Answer:
It decomposes to form carbon (charcoal) and water.
Question 12.
What are minerals?
Answer:
Minerals are naturally occurring substances, often compounds, found in rocks.
Question 13.
What are metalloids?
Answer:
Elements with properties intermediate between metals and non-metals.
Question 14.
How is Dhokra art related to elements and mixtures?
Answer:
Dhokra art uses mixtures of metals like brass or bronze, which are alloys (mixtures) of different elements.
Nature of Matter: Elements, Compounds, and Mixtures Class 8 Short Question Answer
Question 1.
Why is a mixture called an impure substance?
Answer:
Mixture consists of different components which retain their properties and can be easily separated by physical processes, hence it is called an impure substance.
Question 2.
Why is air considered a mixture and not a compound?
Answer:
Air is considered a mixture because it exhibits the following properties:
- Each component present in the air retains its properties.
- The components do not have any fixed proportion. All gases are present in different amounts. For example, in a greener area-more oxygen and water vapour is present; near an industrial area-air consists of a lot of impurities and smoke suspended in it.
![]()
Question 3.
How can you prove that water is a compound?
Answer:
When electricity is passed through water at two different electrodes, we get two different gases, i.e., oxygen and hydrogen. The ratio of hydrogen : oxygen is 2: 1 by number of molecules.
- The properties of oxygen and hydrogen gases are entirely different from that of liquid water.
- The ratio of hydrogen to oxygen combination is always constant, i.e., 2: 1 by volume.
- To separate the components of water, we need a special setup that uses electricity, and it is not a simple process. This proves that water is a compound.
Question 4.
What are native minerals? Provide two examples and explain their significance.
Answer:
Native minerals are those that occur naturally in the earth in their pure, elemental form without being chemically combined with other elements. Examples include gold and sulphur. Native minerals are important because they are sources of pure metals and elements used in various applications, such as gold in jewellery and sulphur in fertilisers and medicines.
![]()
Question 5.
How can iron be separated from a mixture of iron filings and sulphur? Explain the method.
Answer:
Iron can be separated from a mixture of iron filings and sulphur using a magnet. Since iron is magnetic, a magnet can attract and pull out the iron filings from the mixture, leaving the non-magnetic sulphur powder behind. This is a physical method of separation because the substances are not chemically bonded in the mixture.
Nature of Matter: Elements, Compounds, and Mixtures Class 8 Long Question Answer
Question 1.
What is meant by a ‘pure substance’ in science? How does this meaning differ from its use in everyday language? Give examples.
Answer:
In science, a pure substance refers to a material that consists of only one kind of particles (atoms or molecules). It has definite properties such as boiling point, melting point, and composition, and cannot be separated by physical methods.
In everyday language, “pure” means something that is clean or unadulterated. For example, “pure milk” refers to milk that is not diluted with water or other substances, but scientifically, milk is not a pure substance-it is a mixture.
Examples:
Everyday Pure (But Mixtures): Milk, Ghee, Honey
Scientifically Pure: Distilled water, Oxygen, Sodium chloride
Question 2.
Iron filings and sulphur were mixed together and divided into two parts, ‘ A ‘ and ‘ B ‘. Part ‘ A ‘ was heated strongly while Part ‘ B ‘ was not heated. Dilute hydrochloric acid was added to both the parts and evolution of gas was seen in both the cases. How will you identify the gases evolved?
Answer:
The following reaction takes place when part A is heated:
Iron + Sulphur → Iron sulphide
When dil. hydrochloric acid is added to iron sulphide, following reaction takes place and hydrogen sulphide gas is evolved.
Iron sulphide + Dilute hydrochloric acid → Iron chloride + Hydrogen sulphide
Hydrogen sulphide is a foul smelling gas and smells like rotten egg.
When dil. hydrochloric acid is added to the mixture of iron and sulphur, following reaction takes place and hydrogen gas is evolved.
Iron + Dilute hydrochloric acid → Iron chloride + Hydrogen gas
In this case, sulphur does not participate in the reaction.
When a burning matchstick is brought near the evolved gas, the matchstick burns with a pop sound. This confirms the evolution of hydrogen gas.
![]()
Question 3.
Define an element. How are elements classified? Give suitable examples.
Answer:
An element is a pure substance made up of only one kind of atoms. It cannot be broken down into simpler substances by physical or chemical methods.
Classification of elements:
- Metals-Shiny, conduct heat and electricity, malleable. For example, Iron, Copper and Aluminium
- Non-metals – Dull, poor conductors, brittle. For example, Sulphur, Oxygen and Carbon
- Metalloids – Show properties of both metals and non-metals. For example, Silicon and Arsenic
Nature of Matter: Elements, Compounds, and Mixtures Class 8 Skill-Based Questions
Question 1.
Observe the given figure and answer the following questions.

(a) Which property is shown by this figure?
(b) What type of mixture is shown in the figure-uniform or non-uniform?
(c) Name the method used in the figure to separate the components of the mixture.
Answer:
(a) The property of magnetism is shown iron is magnetic and gets attracted to the magnet, whereas sulphur does not.
(b) Non-uniform mixture as the components can be seen separately.
(c) Magnetic separation.
Question 2.
Observe the given figure and answer the following questions.
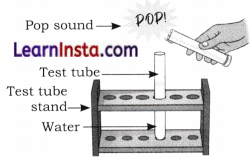
(a) What is the name of the gas collected in the test tube?
(b) What is the characteristic test for gas shown in the figure?
Answer:
(a) Hydrogen gas
(b) It burns with a pop sound when a burning candle is brought near it.
Nature of Matter: Elements, Compounds, and Mixtures Class 8 Quiz Questions
Question 1.
What is the smallest unit of an element?
Answer:
Atom
![]()
Question 2.
Which element is used to make graphene?
Answer:
Carbon
Question 3.
What is formed when two or more elements are chemically combined?
Answer:
Compound
Question 4.
Which method is used to separate iron filings from a mixture?
Answer:
Magnetic separation
Question 5.
What type of mixture has a uniform composition throughout?
Answer:
Uniform-mixture
Question 6.
What do we call a substance made of only one kind of atoms?
Answer:
Element
Question 7.
What type of change occurs when new substances are formed?
Answer:
Chemical change
Question 8.
What do we call the smallest unit of a compound?
Answer:
Molecule
Question 9.
What do we call substances like water, oxygen, and carbon dioxide?
Answer:
Pure substances
![]()
Question 10.
What is the term used to define a mixture that does not look the same throughout?
Answer:
Non-uniform mixture
Nature of Matter: Elements, Compounds, and Mixtures Class 8 Case Based Questions
Question 1.
Matter can be classified into two categories: pure substances and mixtures. Pure substances are made up of a single type of particles and cannot be separated into other substances by physical methods. They have definite and constant properties. On the other hand, mixtures are made up of two or more substances that are physically combined and can be separated into their individual components.
![]()
Mixtures can be further classified into uniform and non-uniform mixtures. Uniform mixtures are uniform in composition, meaning the components are evenly distributed throughout the mixture. Non-uniform mixtures, on the other hand, have non-uniform composition with visible different parts. It is important to understand the nature of matter around us and differentiate between pure substances and mixtures to comprehend their properties and behaviour.
Answer the following questions based on the above information.
I. Give two examples of pure substances.
II. Which type of mixture does not have fixed composition?
III. What is the main characteristic of a pure substance?
OR
How are mixtures different from pure substances?
Answer:
I. Examples of pure substances include water and oxygen.
II. A non-uniform mixture does not have fixed composition.
III. The main characteristic of a pure substance is that it is made up of a single type of particles and has definite and constant properties.
OR
Mixtures are made up of two or more substances physically combined and can be separated into their individual components, whereas pure substances consist of a single type of particles and cannot be separated by physical methods.
Nature of Matter: Elements, Compounds, and Mixtures Extra Questions for Practice
Multiple Choice Questions
Question 1.
Which of the following is a compound?
(a) Iron
(b) Sulphur
(c) Water
(d) Brass
![]()
Question 2.
Which of these is a uniform mixture?
(a) Sand and salt
(b) Salt solution
(c) Iron and sulphur
(d) Oil and water
Question 3.
Graphene is made up of
(a) Iron
(b) Sulphur
(c) Carbon
(d) Copper
Question 4.
Read the given statements and select the correct option:
Assertion (A): Elements cannot be broken down into simpler substances.
Reason (R): Elements are made of identical atoms.
(a) Both A and R are true and R is the correct explanation of A .
(b) Both A and R are true but R is not the correct explanation of A .
(c) A is true but R is false.
(d) A is false but R is true.
![]()
Question 5.
Match the items in Column A to those in Column B.
Column A — Column B
(i) Iron sulphide — (p) Uniform mixture
(ii) Salt solution — (q) Non-uniform mixture
(iii) Iron — (r) Compound
(ii) Sprout salad — (s) Pure element
(a) (i)-(s), (ii)-(p), (iii)-(q), (iv)-(r)
(b) (i)- (r), (ii)-(p), (iii)-(s), (iv)-(q)
(c) (i)- ( r), (ii)-(s), (iii)-(p), (iv)-(q)
(d) (i)-(r), (ii)-(p), (iii)-(q), (iv)-(s)
Very Short Answer Type Questions
Question 1.
Define an element.
Question 2.
Name one use of graphene.
Question 3.
What is the smallest unit of a compound?
Question 4.
Write one property of a non-uniform mixture.
![]()
Question 5.
What is the method used to separate iron from a mixture?
Short Answer Type-I Questions
Question 1.
What is a compound? Give one example.
Question 2.
Differentiate between atoms and elements.
Question 3.
Define pollutant and give one example.
Question 4.
State two differences between uniform and non-uniform mixtures.
Question 5.
Why is air called a mixture?
Short Answer Type-II Questions
Question 1.
Describe the method of magnetic separation with an example.
Question 2.
Explain why iron sulphide is considered a compound and not a mixture.
Question 3.
What are the properties of compounds? (Any three)
![]()
Question 4.
What changes occur when sugar is heated, and what do they indicate about its nature?
Question 5.
Give three examples each of elements, compounds, and mixtures.
Long Answer Type Questions
Question 1.
Compare and contrast elements, compounds, and mixtures based on their composition, separation methods, properties and uses. Also give examples.
Question 2.
Explain with diagrams how magnetic separation can be used to separate a mixture of sulphur and iron filings. State the principle involved.
![]()
Question 3.
A candle makes a pop sound when brought near a gas in a test tube. Which gas is it? How is it produced in the lab? What does the pop sound indicate? What safety precautions should be taken?
Suggested Projects And Activities
Question 1.
Design and create comic strips using real-life examples to differentiate between elements, compounds, and mixtures. Use characters like “Atom Annie” (element), “Molecule Max” (compound), and “Mixie the Mixture” in daily settings such as kitchens or labs. Illustrate their properties, structures, and uses through visuals and dialogues in the comic.
Question 2.
Read labels on items like packaged snacks, toothpaste, or detergents. List out the mixtures and compounds present in them. Classify the ingredients into pure substances or mixtures and create a table showing your findings.