Students can use Curiosity Class 8 Science Book Solutions Chapter 7 Particulate Nature of Matter Class 8 Question and Answer as a quick reference guide.
Class 8 Science Chapter 7 Particulate Nature of Matter Question Answer
Science Class 8 Chapter 7 Question Answer Particulate Nature of Matter
Particulate Nature of Matter Class 8 Question Answer (InText)
Question 1.
Probe and Ponder (Page 98)
- Why is it possible to pile up stones or sand, but not a liquid like water?
- Why does water take the shape of folded hands but lose that shape when released?
- We cannot see air, so how does it add weight to an inflated balloon?
- Is the air we breathe today the same that existed thousands of years ago?
Share your questions
……………………………………………………..
…………………………………………………….. ?
Answer:
(Questions may vary)
(i) How can small amount of sugar sweeten an entire glass of water?
(ii) Why does perfume spread across a room even when sprayed in one corner?
![]()
Question 2.
Is every speck of fine chalk powder still composed of the same substance, or has it changed into something else on breaking or grinding? (Page 99)
Answer:
Yes, every speck of fine chalk powder is still composed of the same substance. Grinding or breaking chalk is a physical change, so its composition remains the same – only the size of the chalk pieces becomes smaller and its shape gets changed.
Question 3.
Are the units of chalk obtained in this manner (by continuously grinding) considered the smallest units of chalk? (Page 100)
Answer:
Yes. When chalk pieces are continuously ground, a stage is reached where the particles can no longer be broken down any further. The tiny units obtained at this stage are the basic building blocks that make up chalk.
Question 4.
Where did the sugar go in water? (Page 101)
Answer:
When sugar is mixed with water, it dissolves. The tiny sugar particles break apart and occupy the available spaces between the water particles.
![]()
Question 5.
Chalk and sugar can both be broken down into their constituent particles. But how are the constituent particles held together to form the solid pieces we see? (Page 101)
Answer:
The constituent particles of chalk and sugar are held together by interparticle attractions – the forces of attraction between particles. These forces bind the particles and give the definite shape and structure to solids.
Question 6.
In the solid state, is there any way to move their particles apart? (Page 102)
Answer:
Yes, by heating the solid. When a solid is heated, its particles vibrate more vigorously. At a certain temperature (melting point), the vibrations become strong enough to overcome the interparticle forces of attraction. As a result, the solid changes into a liquid, and the particles move farther apart.
Question 7.
Solids have a definite volume; what about liquids and gases? (Page 103)
Answer:
- Liquids have a definite volume but no fixed shape. They take the shape of the container in which they are kept.
- Gases have neither a fixed shape nor a definite volume. They expand to fill the entire space available to them.
Question 8.
Do gases also have a fixed volume? (Page 105)
Answer:
No, gases do not have a fixed volume. They expand to occupy the entire available space in a container.
Question 9.
Sugar and sand are both solids. Why does sugar dissolve in water but sand does not? (Page 108)
Answer:
Sugar being soluble, dissolves in water and its particles interact with water particles and occupy the spaces between them.
Sand being insoluble, however, does not dissolve in water. Its particles are too large and do not interact with water molecules, so they simply settle down at the bottom.
![]()
Question 10.
How can we demonstrate the movement of gas particles that cannot be seen with the naked eye? (Page 110)
Answer:
We can demonstrate the movement of gas particles by burning an incense stick in one corner of the room. After some time, its fragrance spreads throughout the room. This shows that gas particles are in constant motion, helping the gas particles to spread in all directions.
NCERT Class 8 Science Chapter 7 Question Answer Particulate Nature of Matter (Exercise)
Keep the curiosity alive(Pages 113-114)
Question 1.
Choose the correct option.
The primary difference between solids and liquids is that the constituent particles are:
(i) closely packed in solids, while they are stationary in liquids.
(ii) far apart in solids and have fixed position in liquids.
(iii) always moving in solids and have fixed position in liquids.
(iv) closely packed in solids and move past each other in liquids.
Answer:
(iv) In solids, constituent particles are closely packed and in liquids, constituent particles move past each other.
Question 2.
Which of the following statements are true? Correct the false statements.
(i) Melting ice into water is an example of the transformation of a solid into a liquid.
(ii) Melting process involves a decrease in interparticle attractions during the transformation.
(iii) Solids have a fixed shape and a fixed volume.
(iv) The interparticle interactions in solids are very strong, and the interparticle spaces are very small.
(v) When we heat camphor in one corner of a room, the fragrance reaches all corners of the room.
(vi) On heating, we are adding energy to the camphor, and the energy is released as a smell.
Answer:
(i) True
(ii) True
(iii) True
(iv) True
(v) True
(vi) False: On heating, camphor changes into gas directly, and the camphor gas particles spread, carrying the fragrance with them.
![]()
Question 3.
Choose the correct answer with justification. If we could remove all the constituent particles from a chair, what would happen?
(i) Nothing will change.
(ii) The chair will weigh less due to lost particles.
(iii) Nothing of the chair will remain.
Answer:
(iii) Nothing of the chair will remain. Matter is made of particles. If we remove all the constituent particles from a chair, it would no longer exist i.e., there would be nothing left.
Question 4.
Why do gases mix easily, while solids do not?
Answer:
Gases have large interparticle spaces and negligible forces of attraction between particles. This allows the particles to move freely in all directions and mix easily.
While in solids, the interparticle spaces are very small. Their particles are tightly packed and cannot move freely. Hence, solids do not mix as easily as gases.
![]()
Question 5.
When spilled on the table, milk in a glass tumbler, flows and spreads out, but the glass tumbler stays in the same shape. Justify this statement.
Answer:
Milk is a liquid so, it has no fixed shape. It flows and spreads out on the surface of the table, because of weaker interparticle attractions. The glass tumbler is a solid. It has a fixed shape due to strong interparticle attractions that hold the particles in place.
Question 6.
Represent diagrammatically the changes in the arrangement of particles as ice melts and transforms into water vapour.
Answer:
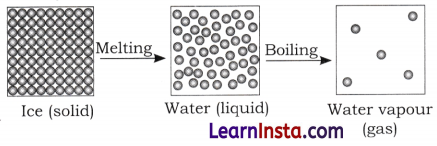
Question 7.
Draw a picture representing particles present in the following:
(i) Aluminium foil
(ii) Glycerin
(iii) Methane gas
Answer:
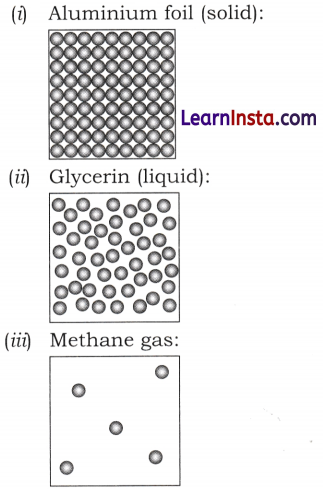
Question 8.
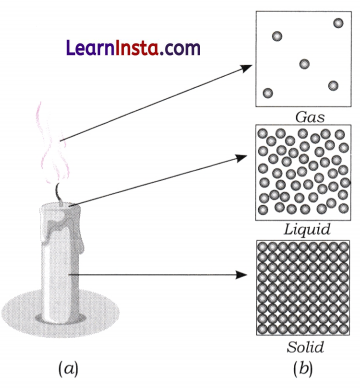
Observe Fig. 7.16a which shows the image of a candle that was just extinguished after burning for some time. Identify the different states of wax in the figure and match them with Fig. 7.16b showing the arrangement of particles.
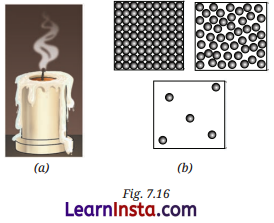
Answer:
States of wax in the given candle:
- Solid wax: The unmelted portion of the candle.
- Liquid wax: The melted wax near the wick.
- Gaseous wax: The wax vapours rising from the flame.
Matching with particle arrangements:
- Solid → Closely packed particles.
- Liquid → Slightly apart, loosely packed.
- Gas → Far apart and freely moving.
Question 9.
Why does the water in the ocean taste salty, even though the salt is not visible? Explain.
Answer:
The salt in ocean water is dissolved. Salt breaks down into its constituent particles, which occupy the interparticle spaces between water particles. These particles are too small to be seen but can be tasted, which gives ocean water a salty flavour.
![]()
Question 10.
Grains of rice and rice flour take the shape of the container when placed in different jars. Are they solids or liquids? Explain.
Answer:
Grains of rice and rice flour are solids. Even though they take the shape of the container, each grain or particle retains its own fixed shape. They behave like solids because their constituent particles are tightly packed and do not flow like the particles in liquids.
Class 8 Particulate Nature of Matter Question Answer (Activities)
Activity 7.1: Let Us Explore (Page 99)
Aim: To explore the physical change in chalk by breaking and grinding it, and to observe that it is made of smaller constituent particles.

Observation:
- The small pieces of chalk are ground into a fine white powder using a mortar and pestle. When observed through a magnifying glass, each small grain of chalk powder still looks like a tiny piece of the original chalk, just much smaller in size.
- Even after grinding, the fine powder is still chalk – it has not changed into any new substance.
Conclusion:
- Grinding of chalk into powder is a physical change. The chalk changes only in size and shape, not in its chemical composition.
- The chalk is made up of many small units, called constituent particles. Even when we break or grind it, these particles remain the same in nature.
Activity 7.2: Let Us Perform (Page 100)
Aim: To observe the process of sugar dissolving in water.
Observation:
- Before stirring, when sugar is added to water, it settles at the bottom, and the top layer of water does not taste sweet.
- After stirring, the sugar dissolves completely, and the top layer of water tastes sweet.
- No sugar particles are visible in the solution after stirring.
Conclusion:
- When sugar dissolves in water, it breaks down into its constituent particles, and spread evenly throughout the solution. Each grain of sugar is made up of millions of such particles.
- The sweet taste after mixing confirms that even though we cannot see the sugar, its constituent particles are still present in the water.
- The sugar particles occupy the spaces between water particles (interparticle spaces).
![]()
Activity 7.3: Let Us Find Out (Page 102)
Aim: To observe that solids have a fixed shape and volume.

Observation:
- All six objects (iron nail, rock salt, stone, wood, key, and aluminium) have a definite shape and size.
When hammered:
- Some objects like the iron nail, key, and aluminium piece are hard to break or change shape.
- Other objects like rock salt, stone and wood may break into smaller pieces but still remain in solid form.
Conclusion:
- All these materials are solids, which have a fixed shape and volume.
- The particles in solids are tightly packed and strongly held together by strong forces of attraction.
Activity 7.4: Let Us Try And Find Out (Page 104)
Aim: To find out that liquids have no fixed shape but have a fixed volume.
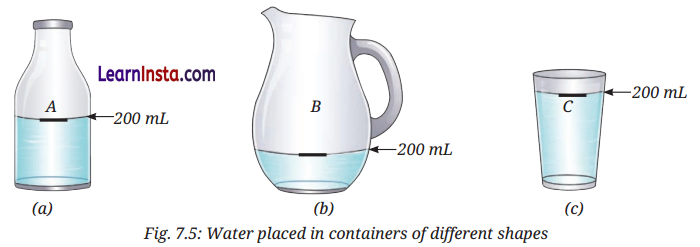
Observation:
- When water is transferred from container A to container B, it reaches 200 mL mark and takes the shape of the container B.
- Similarly, when water is transferred from container B to container C, it again reaches 200 mL mark and takes the shape of the container C .
Conclusion:
- Liquids do not have a fixed shape. They take the shape of the container in which they are kept.
- Liquids have a fixed volume.
![]()
Activity 7.5: let us investigate (pages 105-106)
Aim: To investigate that gases have no fixed shape or volume.
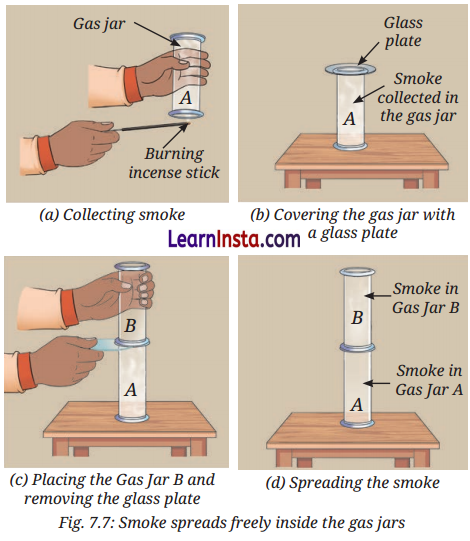
Observation:
When smoke from a burning incense stick is trapped in Gas Jar A and another empty Gas Jar B is placed above it, the smoke fills the entire space in Gas Jar B and takes its shape.
Conclusion:
- Gases do not have a fixed shape or volume. They spread out and occupy the entire available space of the container
they are in. - Gas particles move freely in all directions, with negligible interparticle attraction.
Activity 7.6: Let Us Experiment (Page 107)
Aim: To experiment and find out that gases are compressible, while liquids are not.
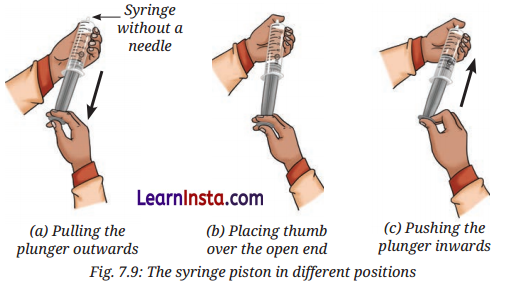
Observation:
- When the plunger of the syringe containing air is pushed inward while the opening is blocked using thumb, the air inside the syringe gets compressed and the volume decreases.
- If the pressure is released, the plunger moves back to its original position as the gas particles expands again.
- When the activity is repeated with water inside the syringe, the plunger does not move inward easily, indicating that water cannot be compressed like air.
Conclusion:
- Gases have large interparticle spaces, which allow their particles to be compressed when pressure is applied.
- When pressure is removed, gas particles move apart again, showing that gases are highly compressible.
- Liquids have much smaller interparticle spaces than gases, so they are almost incompressible.
![]()
Activity 7.7: Let Us Observe (Page 108)
Aim: To observe how the volume of water changes when a soluble solid dissolves in it.

Observation:
- When sugar is added to water, the water level rises from mark A to mark B.
- After stirring and dissolving the sugar, the water level slightly decreases to mark C.
- The final volume of solution is less than the expected sum of the volumes of water and sugar taken.
Conclusion:

- Water has empty spaces between its particles (called interparticle spaces).
- When sugar dissolves, its particles occupy these spaces, so the volume of the solution does not increase much.
- If a similar activity is performed with other soluble solids, like common salt or glucose, their particles also occupy the spaces between water particles, and the volume of the solution does not increase much.
- If a similar activity is performed with insoluble solids, like sand or stone pieces, their particles do not dissolve. Instead, they settle at the bottom and occupy space in the container, causing the total volume of the mixture to increase.
Activity 7.8: let us experiment (pages 109-110)
Aim: To experiment and observe how potassium permanganate particles spread in water.

Observation:
- When a few grains of potassium permanganate are added to water, pink streaks begin to spread out from the grains.
- After some time, the entire water turns uniformly pink without stirring.
Conclusion:
- Water particles are in constant motion.
- First, the water particles pull out the potassium permanganate particles and then they hit these particles so that they spread throughout the liquid.
![]()
Activity 7.9: let us find out (pages 110-111)
Aim: To find out how fragrance particles spread through air.

Observation:
- When an incense stick is burnt in one corner of the room, the fragrance is first felt only near the stick.
- After a few minutes, the fragrance spreads throughout the room, even to areas far from the incense stick.
Conclusion:
- The particles of air are in constant motion.
- These moving air particles hit the fragrance particles and help them spread in all directions.