Students can use Curiosity Class 7 Science Book Solutions Chapter 2 Exploring Substances Acidic Basic and Neutral Class 7 Question and Answer as a quick reference guide.
Class 7 Science Chapter 2 Exploring Substances Acidic Basic and Neutral Question Answer
Science Class 7 Chapter 2 Question Answer Exploring Substances Acidic Basic and Neutral
Exploring Substances Acidic Basic and Neutral Class 7 Question Answer (InText)
Question 1.
I am curious to know what are these red and blue litmus paper strips made of? Why do they change colour when drops of some samples are put on them? (Page 10)
Answer:
Litmus paper strips, both red and blue, are made of filter paper soaked with a natural dye derived from lichens. This dye, called litmus, is a pH indicator, meaning it changes colour depending on the acidity or alkalinity of a solution it is in contact with.
Why do they change colour?
- Acids: Blue litmus paper turns red when it comes into contact with an acidic solution, as the dye molecules react with the hydrogen ions (H+) of the acid.
- Bases: Red litmus paper turns blue when it comes into contact with a basic (alkaline) solution, as the dye molecules react with the hydroxide ions (OH-) of the base.
- Neutral solutions: Litmus paper will not change colour in a neutral solution, as there are no acidic or basic components present to react with the dye.
Question 2.
If litmus is not available, are there some other natural substances that can serve as acid- base indicators? (Page 11)
Answer:
Yes, several natural substances can serve as acid-base indicators when litmus is unavailable. These include turmeric, red cabbage, grape juice, hibiscus flowers, and china rose. Turmeric turns yellow in acid and red in base, while red cabbage, grape juice, and China rose exhibit different colour changes depending on the pH of the solution.
![]()
Question 3.
Are there any substances whose odours change on adding acidic or basic substances? (Page 16)
Answer:
Some substances can change their odours when exposed to acidic or basic solutions. These are called olfactory indicators.
| Substances | Odour in Neutral/ Basic condition | Odour in Acidic Condition |
| Garlic, Onion | Strong, Pungent smell | Reduced or altered |
| Cabbage | Sulfurous smell, more in base | Less intense smell |
| Fish (Spoiled fish) | Fishy odour | odour disappear |
| Milk (Spoiled milk) | Sour smell develops over time | More acidic-stronger sour odour |
NCERT Class 7 Science Chapter 2 Question Answer Exploring Substances Acidic Basic and Neutral (Exercise)
Let Us Enhance Our Learning (Pages 20-21)
Question 1.
A solution turns the red litmus paper to blue. Excess addition of which of the following solution would reverse the change?
(i) Lime water
(n) Baking soda
(in) Vinegar
(iv) Common salt solution
Answer:
(iii) Vinegar
Explanation:
If a solution turns red litmus paper blue, it is basic. To reverse this and turn the paper litmus red again, we need to add an acidic solution. Vinegar is acidic.
Question 2.
You are provided with three unknown solutions labelled A, B, and C, but you do not know which of these are acidic, basic, or neutral. Upon adding a few drops of red litmus solution to solution A, it turns blue. When a few drops of turmeric solution are added to solution B, it turns red. Finally, after adding a few drops of red rose extract to solution C, it turns green. Based on the observations, which of the following is the correct sequence for the nature of solutions A, B, and C?
(i) Acidic, acidic, and acidic
(ii) Neutral, basic, and basic
(iii) Basic, basic, and acidic
(iv) Basic, basic, and basic
Answer:
(iv) Basic, basic, and basic
Explanations:
A turns red litmus blue → A is basic
B turns turmeric red → B is basic
C turns red rose extract green → C is basic
Question 3.
Observe and analyse Figs, a, b, and c, in which red rose extract paper strips are used. Label the nature of solutions present in each of the containers.
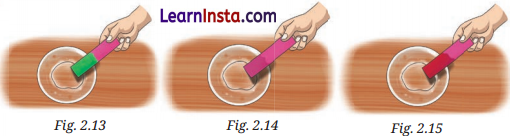
Answer:
Observations:
Fig. (a) shows green colour change → Basic solution
Fig. (b) shows no colour change → Neutral or Acidic solution
Fig. (c) shows bright red colour change → Acidic solution
Conclusion:
Fig.(a): Basic solution
Fig. (b) Neutral or Acidic solution
Fig. (c): Acidic solution
Question 4.
A liquid sample from the laboratory was tested using various indicators:
| Indicator | Red litmus | Blue litmus | Turmeric |
| Change | No change | Turned red | No change in colour |
Based on the tests, identify the acidic or basic nature of the liquid and justify your answer.
Answer:
Conclusion: The liquid is acidic.
Justification:
• Red litmus remains red is an acid.
• Blue litmus turning red indicates an acid.
• Turmeric doesn’t change colour in acid confirms it’s not basic.
Question 5.
Manya is blindfolded. She is given two unknown solutions to test and determine whether they are acidic or basic. Which indicator should Manya use to test the solutions and why?
Answer:
Manya should use olfactory indicators. Onion juice is an olfactory indicator, it changes its smell in basic solutions but retains its characteristic smell in acids and neutral solutions. Since she cannot see the colour change, the change in smell (for visually impaired) may help.
![]()
Question 6.
Could you suggest various materials which can be used for writing the message on the white sheet of paper (given at the beginning of the chapter) and what could be in the spray bottle? Make a table of various possible combinations and the colour of the writing obtained.
Answer:
| Writing Material | Spray Bottle Liquid | Resulting Colour |
| Lime water | Turmeric solution | Reddish brown |
| Soap water | Turmeric solution | Reddish brown |
| Vinegar | Red cabbage extract | Pink |
| Baking soda solution | Red rose extract | Green |
Question 7.
Grape juice was mixed with red rose extract; the mixture got a tint of red colour. What will happen if baking soda is added to this mixture? Justify your answer.
Answer:
Grape juice contains citric acid. Adding baking soda (a base) will make the mixture more basic, turning it green because the red rose extract indicator turns green in basic solution.
Question 8.
Keerthi wrote a secret message to her grandmother on her birthday using orange juice. Can you assist her grandmother in revealing the message? Which indicator would you use to make it visible?
Answer:
Orange juice contains citric acid. We can assist her grandmother in revealing the message by making use of indicator. We can make use of red cabbage leaves extract as it gives a shade of pink or red colour with acids while the background paper will remain purplish or slightly blue.
Question 9.
How can natural indicators be prepared? Explain by giving example.
Answer:
Natural indicators can be made by crushing petals, leaves, or roots using mortar and pestle, then putting them in hot water filtering it and using filtrate as indicator. For example- Collect petals of red roses after crushing, put them in glass tumbler, pour hot water and ensure that they are completely immersed in it. Cover the glass tumbler with the lid. Wait for 5-10 minutes till the water becomes coloured, and filter it. The filtrate is the flower extract that can be used as acid-base indicator.
Question 10.
Three liquids are given to you. One is vinegar, another is a baking soda solution, and the third is a sugar solution. Can you identify them only using turmeric paper? Explain.
Answer:
• Vinegar: No colour change (acidic or neutral)
• Baking soda: Turns turmeric paper reddish-brown(basic).
• Sugar solution: No colour change (Acidic or Neutral) with turmeric paper or reddish brown turned turmeric paper.
![]()
Question 11.
The extract of red rose turns the liquid X to green. What will the nature of liquid X be? What will happen when excess of amla juice is added to liquid X?
Answer:
Liquid X is basic as it turned red rose extract to green. When excess amla juice is added to the solution the solution become acidic and the green colour changes to pink or red.
Question 12.
Observe and analyse the information given in the following flowchart. Complete the missing information.
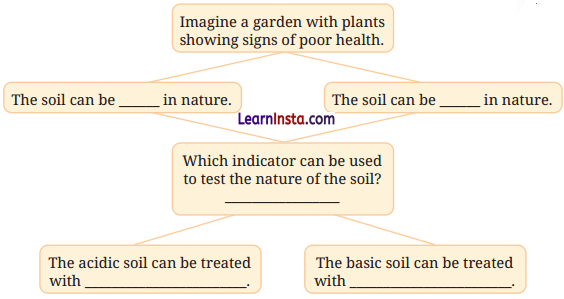
Answer:
• The soil can be acidic in nature.
• The soil can be basic in nature.
• Indicator: Red cabbage extract or litmus can be used.
• The acidic soil can be treated with quick lime or slaked lime.
• The basic soil can be treated with organic compost or manure.
Class 7 Exploring Substances Acidic Basic and Neutral Question Answer (Activities)
Activity 2.1: Let Us Explore (Page 8-9)
Table 2.1: Testing the nature of samples with blue and red litmus papers
| Name of the Sample | Colour of blue litmus paper after putting a drop of sample | Colour of red litmus paper after putting a drop of sample |
| Lemon juice | Red | Red |
| Soap solution | Blue | Blue |
| Amla juice | Red | Red |
| Tamarind water | Red | Red |
| Vinegar | Red | Red |
| Baking soda solution | Blue | Blue |
| Lime water | Blue | Blue |
| Tap water | Blue | Red |
| Washing powder solution | Blue | Blue |
| Sugar solution | Blue | Red |
| Salt solution | Blue | Red |
Table 2.2: Grouping of samples tested in Table 2.1
| Group A (Acidic) | Group B (basic) | Group A (Neutral) |
| Lemon juice, | Soap solution | Tap water |
| Amla juice, | Baking soda solution | Sugar solution |
| Tamarind water, | Lime water | Salt solution |
| Vinegar, | Washing powder solution | Group A (Neutral) |
Observation and conclusion: Substances in Group A, like lemon juice and vinegar, are acidic as they turn blue litmus paper red. Group B substances, like soap and baking soda, are basic as they turn red litmus paper blue. Group C substances, like tap water and sugar solution, are neutral because they do not change the litmus paper colour.
![]()
Activity 2.2: Let Us Relate and Explore (Page 11)
Find out and write the names of the most common acids present in the following substances
(i) Lemon
(ii) Curd
(iii) Tamarind
(iv) Vinegar
Answer:
Lemon: Citric acid
Curd: Lactic acid
Tamarind: Tartaric acid
Vinegar: Acetic acid
Activity 2.4: Let Us Find out (Pages 12-13)
Table 2.3: Testing the nature of samples with red rose extract
| Name of the Sample | The colour of the red rose extract after adding the sample | Nature of substance |
| Lemon juice | Red | Acidic |
| Soap solution | Green | Basic |
| Amla juice | Red | Acidic |