Students can access the CBSE Sample Papers for Class 11 Chemistry with Solutions and marking scheme Set 5 will help students in understanding the difficulty level of the exam.
CBSE Sample Papers for Class 11 Chemistry Set 5 with Solutions
Time Allowed : 3 hours
Maximum Marks: 70
General Instructions:
- There are 33 questions in this question paper with internal choice.
- Section – A consists of 16 multiple -choice questions carrying 1 mark each.
- Section – B consists of 5 short answer questions carrying 2 marks each.
- Section – C consists of 7 short answer questions carrying 3 marks each.
- Section – D consists of 2 case – based questions carrying 4 marks each.
- Section – E consists of 3 long answer questions carrying 5 marks each.
- All questions are compulsory.
- Use of log tables and calculators is not allowed.
Section-A
The following questions are multiple-choice questions with one correct answer. Each question carries 1 mark. There is no internal choice in this section.
Question 1.
How many neutrons and protons are there in ^Sr ?
(A) 40
(B) 55
(C) 50
(D) 70
Answer:
(C) 50
Explanation:
38 SR Number of protons = 38 Number of neutrons = 88 – 38 = 50
Question 2.
Which of the following sets of quantum numbers are not possible?
(A) n = 0,1 = 0 ml = 0, ms = + 1/2
(B) n = 1,1 = 0 ml = 0, ms = -1/2
(C) n = 2,1 = 1 ml = 0, ms = – 1/2
(D) n = 3,1 = 1 ml = 0, ms = + 1/2
Answer:
(B) n = 1,1 = 0 ml = 0, ms = -1/2
Explanation:
A hydrocarbon with carbon-carbon single bond is called alkane. It is a saturated hydrocarbon.
Question 3.
Which of the following sets of quantum numbers are correct?

Answer:
![]()
Explanation:
In (A) value of ml of + 2 is not possible as mt is given as 2l + 1. So, ml = +1, 0, -1.
In (D) value of l of 4 is not possible as l is given as (n – 1). So l = 0, 1, 2.
Question 4.
The electronic configuration of gadolinium (Atomic number 64) is:
(A) [Xe] 4f3 5d5 6s2
(B) [Xe] 4f7 5d2 6s1
(C) [Xe] 4f7 5d1 6s2
(D) [Xe] 4f8 5d6 6s2
Answer:
(C) [Xe] 4f7 5d1 6s2
Explanation:
Gadolinium (Gd, Z = 64), belongs to 6th period, f block. The expected electronic configuration of Gadolinium [Xe]4f8 6s2, but the observed electronic configuration is [Xe]4f7 5d1162. This is because half filled orbital is more stable than the partially filled orbital. As 5d and 6f orbitals are close with less energy difference, 1 electron from 4f orbital (expected to have 8e) jumps to 5d orbital making it 5D1 making 4f orbital half filled with high stability (as per Hund’s rule exactly of maximum multiplicity).
Question 5.
The first ionisation enthalpies of Na, Mg, A1 and Si are in the order:
(A) Na < Mg > A1 < Si (B) Na > Mg > A1 > Si
(C) Na < Mg < Al < Si (D) Na > Mg > A1 < Si
Answer:
(A) Na < Mg > A1 < Si
Explanation:
On moving from left to right in a period first ionisation enthalpy increases normally. But, in Mg, as an electron needs to be removed from fully filled s orbital therefore, first ionisation enthalpy of Mg is more than Al.
Question 6.
The electronic configuration of the outer most shell of the most electronegative element is:
(A) 2s22p5
(B) 3s23p5
(C) 4s24p5
(D) 5s25p5
Answer:
(A) 2s22p5
Explanation:
Electronic configuration in (a) is of fluorine while (b), (c) and (d) are of chlorine, bromine and iodine respectively. Fluorine is the most electronegative element.
Question 7.
Catenation, i.e., linking of similar atoms depends on size and electronic configuration of atoms. The tendency of catenation in Group 14 elements follows the order:
(A) C > Si > Ge > Sn
(B) C > Si > Ge = Sn
(C) Si > C > Sn > Ge
(D) Ge > Sn > Si > C
Answer:
(B) C > Si > Ge = Sn
Explanation:
Because the decrease in catenation property is linked with M – M bond energy which decreases from carbon to tin.
Question 8.
The enthalpies of all elements in their standard states are:
(A) unity
(B) zero
(C) < 0
(D) different for each element
Answer:
(B) zero
Explanation:
Enthalpies of Na, K, He etc. in their standard states are zero.
Question 9.
Change in free energy for the equilibrium, gaseous reaction, PC15 → PC13 + Cl2 on addition of an inert gas at constant pressure and at constant volume is respectively:
(A) decrease, no change
(B) increase, no change
(C) no change, no change
(D) no change, decrease
Answer:
(A) decrease, no change
Explanation:
Addition of inert gas at constant volume has no effect on any equilibrium reaction. When inert gas is added at constant pressure in the given reaction, it proceeds in forward direction. ΔG° remains constant but ΔG decreases as the reaction proceeds spontaneously in forward direction to attain equilibrium
Question 10.
The oxidation number of each sulphur atom in Na2S2O3 is:
(A) -2/+6
(B) -41+6
(C) -2/4-8
(D) -l/=5
Answer:
(A) -2/+6
Explanation:
Na2S2O3 Consider the structure of Na2S2O3
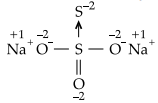
Since, there is a coordinate bond between two S-atoms. So, the oxidation number of acceptor S-atom is -2.
Let, the oxidation number of other S-atom = X
![]()
2 – 6 + x – 2 = 0
X – 6 = 0
X = + 6
Hence, two sulphur atoms in Na2S2O3 have – 2 and +6 oxidation number.
Question 11.
Identify which kind of reaction it is:
CH3Cl + NaOH → CH3OH + NaCl
(A) Substitution
(B) Elimination
(C) Addition
(D) Molecular rearrangement
Answer:
(A) Substitution
Question 12.
Identify which kind of reaction it is:
CH3CONH2 + Br2 + 4KOH -> CH3NH2 + K2CO3 + 2KBr 4-2H2O
(A) Substitution
(B) Elimination
(C) Addition
(D) Molecular rearrangement
Answer:
(B) Elimination
Question 13.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Energy of resonance hybrid is equal to the average of energies of all canonical forms.
Reason (R): Resonance hybrid cannot be presented by a single structure.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(D) A is false but R is true.
Explanation:
Canonical structures always have more energy than resonance hybrid. Resonance hybrids are always more stable than any of the canonical structures. The delocalisation of electrons lowers the orbitals energy and gives stability.
Question 14.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Among the two O-H bonds in H2O molecule, the energy required to break the first O-H bond and the other O-H bond is the same.
Reason (R): This is because the electronic environment around oxygen is the same even after breakage of one O-H bond.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(D) A is false but R is true.
Explanation:
In case of H2O molecule, the enthalpy needed to break the two O—H bonds is not the same. The difference in the ΔaH° value shows that the second O—H bond undergoes some change because of changed chemical environment.
Question 15.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Absolute value of internal energy (U) cannot be determined.
Reason (R): Internal energy is the sum of many types of energies, that depend on several factors.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A
Question 16.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): The increase in energy (ΔU) for the vaporization of one mole in water at 1 atm and 373 K is zero. Reason (R): For gaseous isothermal processes, ΔU = 0.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A
Explanation:
For all isothermal process, internal energy is zero.
Section-B
This section contains 5 questions with internal choice in one question. The following questions are very short answer type and carry 2 marks each.
Question 17.
(A) Cs shows maximum photoelectric effect, why?
(B) What type of metals are used in photoelectric cell? Give one example.
Answer:
(A) Cs is the most electropositive element of all so it has the minimum ionisation energy and contains the maximum capacity to loose electrons. That s why Cs shows maximum photoelectric effect.
(B) The metals having large size and less work function are used in photoelectric cell. e.g., Li
Question 18.
To which orbit the electron in H-atom will jump on absorbing 12.1 eV energy?
Answer:
According to Bohr’s theory, Energy of electron in nth orbital of H-atom with atomic number (Z) = -13.6 \(\frac{\mathrm{Z}^2}{n^2}\)eV
∴ The energy of ground state electron in H-atom = -13.6 eV
Now, it absorbs 12.1 eV energy.
So, its energy increases to -13.6 + 12.1 = – 1.5 eV
∴ E = -13.6 \(\frac{\mathrm{Z}^2}{n^2}\) eV
– 1.5 = -13.6 × \(\frac{(1)^2}{n^2}\) (∴ for hydrogen Z = 1)
∴ n2 = \(\frac{13.6}{1.5}\) = 9.06 ≈ 9
∴ n = 3
Hence, the electron in H-atom jumps to 3rd orbit on absorbing 12.1 eV energy.
Question 19.
Among the elements B, Al, C and Si.
(A) Which element has the highest first ionisation enthalpy?
(B) Which element has the most metallic character? Justify your answer in each case?
Answer:
(A) Since the ionisation enthalpy increases on moving from left to right in a period with decrease in atomic size and decreases down the group with increase in atomic size. So, among B, Al, C and Si, carbon (C) has the highest first ionisation enthalpy.
(B) Metallic character decreases on moving from left to right in a period and increases on moving down the group. So, among B, Al, C and Si, aluminium (Al) has the most metallic character.
Question 20.
(A) Show by a chemical reaction with water that Na2O is a basic oxide and C12O7 is an acidic oxide?
(B) What will happen when Aluminium reacts with cold and hot water?
OR
Find out the value of Kc for each of the following equilibria from thd value of Kp
(i) 2 NOC1 (g) ⇌ 2 NO (g) + Cl2 (g),Kp = 1.8 × 10-2 at 500K
(ii) CaCO3 (s) CaO (s) + CO2 (g), Kp = 167 at 1073 K.
Answer:
(A) Na2O with water forms a strong base whereas Cl2O7 forms strong acid.
Na2O + H2O → 2NaOH
Cl2O7 + H2O → 2HClO4
Their basic or acidic nature can be qualitatively tested with litmus paper
(B) Aluminium do not react with cold and hot water.
It react with steam to give –
2Al(s) + 3H2O(g) → Al2O3(s) + 3H2(g)
Or
(i) ∆ng = 3 – 2 = 1, Kp = Kc × (RT)∆n
or
Kc = \(\frac{\mathrm{K}_p}{\mathrm{RT}}=\frac{1.8 \times 10^{-2}}{0.0831 \times 500}\)
(R = 0.0831 bar liter mol-1K-1
= 4.33 × 10-4
(ii) ∆ng = 1 – 0 = 1,
Kc = \(\frac{K_p}{R T}=\frac{167}{0.0831 \times 1073}\)
= 1.87
Question 21.
(A) Identify the oxidising agent and reducing agent in the following reaction :
2MnO4 + 6I– + 4H2O → MnO2 + 3I2 + 80H+
(B) What do you understand by disproportionation reaction?
Answer:
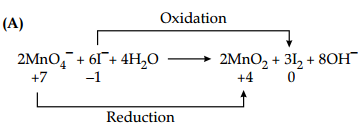
Here, MnO4– is an oxidising reagent as it itself gets reduced and I– is a reducing reagent as it itself gas oxidised. (1)
(B) Disproportionation reaction are special type of redox reactions. In this reactions, an element in one oxidation state is simultaneously oxidized and reduced.
![]()
Section-C
This section contains 7 questions with internal choice in one question. The following questions are short answer type and carry 3 marks each. (No internal choice given in Board SQP)
Question 22.
(A) What are the allowed value for each of the four quantum numbers?
(B) Identify the sub-shell in which electrons with the following quantum numbers are found?
(i) n = 2, l = 1
(ii) n = 4, l = 2
(C) Describe the properties of an electron associated with n and l quantum numbers?
Answer:
(A) n = non-zero integer
l = 0 to n – 1
ml = -l to + l
ms = \(\frac{1}{2}\) or – \(\frac{1}{2}\)
(B) (i) 2p (ii) 4d
(C) The value of ‘n’ describe the range of energy value and possible distance between the nucleus and the electron. ‘l’ describe the shape of the orbital.
Question 23.
C(s) + O2(g) → CO2(g) + 394 kJ
C(s) + 1/2O2(g) → CO(g) + in kJ
(A) In an oven using coal (assume the coal is 80% carbon in weight), insufficient oxygen is supplied such that 60% of carbon is converted to CO2 and 40% carbon is converted to CO. Find out the heat generated when 10 kg of coal is burnt in this fashion.
(B) Calculate the heat generated if a more efficient oven is used so that only CO2 is formed.
(C) Calculate the percentage loss in heating value for the inefficient oven.
Answer:
(A) Weight of pure coal = \(\frac{80}{100}\) × 10 = 8.0 kg
Weight of coal converted into CO2
= 8 × \(\frac{60}{100}\) = 4.8 kg
Weight of coal converted in to CO
= 8 – 4.8 = 3.2 kg
Moles of C in 4.8 kg coal = \(\frac{4800}{12}\) = 400 moles
Moles of C in 3.2 kg coal = \(\frac{3200}{12}\) = 266.67
Now, C + O2 → CO2(g) + 394 KJ
1 mole of C give heat = 394 KJ
∴ 400 moles give heat = 400 × 394 = 157600 kJ
C + 1/2O2 → CO + 111 kJ
1 mole of C gives heat = 111 kJ
∴ \(\frac{3200}{12}\) = 29600 KJ
Total heat generated = 157600 + 29600 = 187200 kJ
(B) C + O2 → CO2 + 394
1 mole of C gives heat = 394 kJ
\(\frac{8000}{12}\) mole of C gives heat
= 394 × \(\frac{8000}{12}\) = 262667 KJ
(C) Heat lost due to inefficient oven
= 262667 – 187200 = 75467 kJ
∴ % loss = \(\frac{75467}{262667}\) × 100 = 28.73%.
Question 24.
A vessel contains 1.6 g of dioxygen at STP (273.15 K, 1 atm pressure). The gas is now transferred to another vessel at constant temperature, where pressure becomes half of the original pressure. Calculate
(A) volume of the new vessel.
(B) number of molecules of dioxygen.
Answer:
(A) p1 = 1 atm.
T1 = 273K
V1 = ?
32 g of oxygen occupies 22.4 L of volume at STP
Hence, 1.6 g of oxygen will occupy,
1.6g oxygen × \(\frac{22.4 \mathrm{~L}}{32 \mathrm{~g} \text { oxygen }}\) = 1.12 L
V1 = 1.12 L
p2 = \(\frac{p_1}{2}=\frac{1}{2}\) = 0.5 atm.
V2 = ?
According to Boyle’s law:
p1V1 = p1V2
or
V2 = \(\frac{p_1 \times V_1}{p_2}\) = \(\frac{1 \mathrm{~atm} \times 1.12 \mathrm{~L}}{0.5 \mathrm{~atm} .}\) = 2.24 L
(B) Number of molecules of oxygen in the vessel
= \(\frac{6.022 \times 10^{23} \times 1.6}{32}\)
= 3.011 × 1022 moleculars
Question 25.
Calculate the standard enthalpy of formation of C2H4(g) from the following thermo-chemical equation:
C2H4(g) + 3O2(g) → 2CO2(g) + 2H2O(g); ∆rHθ = -1323 kj mol-1
Given that ∆fHθ of CO2(g) and H2O(g) as – 393.5 and – 249 kJ mol-1 respectively.
Answer:
∆fHθ of O2(g) = 0 by convention
∆rHθ = Σ∆fHθ(products) – Σ∆fHθ(reactants)
Substituting the given values :
– 1323 = [2 × (-393.5) + 2 × (- 249)] – [∆fHθC2H2]
– 1323 = [-787 – 498] – [∆fHθC2H2]
– 1323 = – 1285 – ∆fHθC2H2
∴ ∆fHθC2H2 = 1323 – 1285
= 38 KJ mol-1
Question 26.
Draw the resonance structures of the following compounds:
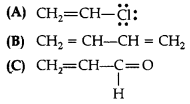
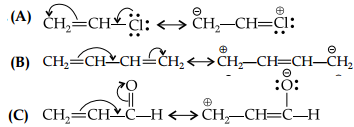
Answer:
Question 27.
Despite their -I effect, halogens are o- and p-direction in haloarenes. Explain?
Answer:
In case of aryl halides, halogens are little deactivating because of their strong -I effect. Therefore, overall electron density on the benzene ring decreases. In other words, halogens are deactivating due to -I effect. However, because of the +R-effect, i.e., participation of lone pairs of electrons on the halogen atom with the p-electrons of the benzene ring, the electron density increases more at O- and p-positions than at m-positions. As a result, halogens are o-, p-directing. The combined result of +R-effect and -I-effect of halogens is that halogens are deactivating but o, p-directing.
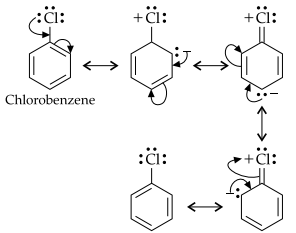
Question 28.
In the alkane H3C – CH2 – C(CH3)2 – CH2 – CH(CH3)2, identify 1°, 2°, 3° carbon atoms and give the number of H atoms bonded to each one of these.
Answer:
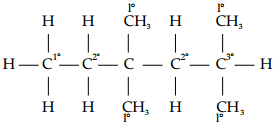
1° carbon atoms are those which are bonded to only one carbon atom, i.e., they have only one carbon atom as their neighbour. The given structure has five 1° carbon atoms and fifteen hydrogen atoms attached to it. 2° carbon atoms are those which are bonded to two carbon atoms, i.e., they have two carbon atoms as their neighbours. The given structure has two 2° carbon atoms and four hydrogen atoms attached to it. 3° carbon atoms are those which are bonded to three carbon atoms, i.e., they have three carbon atoms as their neighbours. The given structure has one 3° carbon atom and only one hydrogen atom is attached to it.
Section-D
The following questions are case-based questions. Each question has an internal choice and carries 4( 1 + 1 + 2 ) marks each.
Question 29.
Read the passage given below and answer the following questions:
Because ethane exists as a gas at normal temperature and pressure, exposure occurs by inhalation. Concentrations of ethane in natural gas range from 5 to 10%. It is also found in the exhaust of diesel (∼1.8%) and gasoline (1.3 – 2.0%) engines. Small amounts of ethane, along with other C1 and C4 alkanes and alkenes, have been detected in mined coal samples. Ethane emissions from cigarettes have been measured at 1600 ug per cigarette. Typical background air concentrations in major US cities range from 0.05 to 0.5 ppm. Because it is lighter than air, a major spill would not be expected to migrate and affect adjacent properties or neighbourhoods. It is possible to spill liquid ethane from a refrigerated tank, causing frostbite upon contact with the skin due to rapid evaporation and loss of heat.
(A) How many moles of carbon atoms are present in 3 moles of ethane?
OR
How many moles of hydrogen atom is present in 3 moles of ethane?
(B) How many molecules of ethane will be there in 3 moles of ethane?
(C) How liquid ethane cause frostbite?
Answer:
(A) 1 mole of ethane (C2H6) contains 2 moles of carbon atoms.
So, 3 moles of C2H6 will contain = 2 × 3 = 6 mol of carbon atoms
OR
1 mole of C2H6 contains 6 moles of hydrogen atoms.
So, 3 moles of C2H6 will contain = 6 × 3 = 18 mol of hydrogen atoms.
(B) 1 mole of C2H6 contains
6.022 × 1023 molecules of ethane.
So, 3 moles of C2H6 will contain
= 3 × 6.022 × 1023
= 18.066 × 1023 molecules
= 1.8066 × 1024 molecules
(C) Liquid ethane is highly vaporisable, so can cause frostbite when comes in contact with skin due to vaporisation and loss of heat from the affected area.
Question 30.
Read the passage given below and answer the following questions:
Le Chatelier’s principle is an observation about chemical equilibria of reactions. It states that changes in the temperature, pressure, volume, or concentration of a system will result in predictable and opposing changes in the system in order to achieve a new equilibrium state. Le Chatelier’s principle can be used in practice to understand reaction conditions that will increased product formation. This idea was discovered and formulated independently by Henri Louis Le Chatelier and Karl Ferdinand Braun.
(A) On increasing the pressure, in which direction will the gas phase reaction proceed to reestablish equilibrium, is predicted by applying the Le Chatelier’s principle. Consider the reaction.
N2(g) + 3H2(g) ⇌ 2NH3(g)
What will happen to the value of K, if the total pressure at which the equilibrium is established, is increased without changing the temperature?
(B) Give two important characteristics of chemical equilibrium.
(C) A liquid is in equilibrium with its vapour in a sealed container at a fixed temperature. The volume of the container is suddenly increased. What is the initial effect of the change on vapour pressure? How do rates of evaporation and condensation change initially?
OR
Explain why pure liquids and solids can be ignored while writing the equilibrium constant expression?
Answer:
(A) According to Le Chatelier’s principle, at constant temperature, the equilibrium composition will change but K will remain same.
(B) Two important characteristics of chemical equilibrium are;
- Chemical equilibrium is dynamic in nature.
- Chemical equilibrium can be achieved from either of the directions, i.e., starting the reaction either by taking the reactants or products.
(C) (i) Initially, the vapour pressure will decrease because the same amount of vapour is now distributed in larger space.
(ii) The rate of evaporation remains constant at constant temperature in a closed vessel. However, the rate of condensation will be low initially because there are fewer molecules per unit volume in the vapour phase and hence the number of collisions per unit time with the liquid surface decreases.
OR
Concentration of pure solid and liquids remain constant as their molar concentration are constant, i.e., independent of the amount present. Therefore, concentration terms of pure solids and pure liquids can be ignored while writing the equilibrium constant expression.
Section-E
The following questions are long answer type and carry 5 marks each. All questions have an internal choice.
Question 31.
Attempt any five questions:
(A) What is uncertain in uncertainty principle?
(B) de Broglie concept cannot be applied to a moving cricket ball. Why?
(C) Write the Schrodinger wave equation.
(D) Which of the following has smallest de-Broglie wavelength ? H2, a proton or an electron.
(E) How much energy is required for the removal of only electron present in the hydrogen atom?
(F) What is the number of waves in nth orbit?
(G) Arrange s, p and d sub-shells of a shell in the increasing order of effective nuclear charge (Zeff) experienced by the electron present in them.
Answer:
(A) Position and momentum (or velocity) of particle are uncertain in uncertainty principle. (1)
(B) The de Broglie equation predicts that a moving ball will behave like a wave, however its wavelength would be exceedingly tiny for ordinary observation. Thus, it may be said that a moving ball won’t possess wavelike characteristics.
λ = \(\frac{h}{m v}=\frac{h}{p}\)
(C) \(\hat{\mathrm{H}} \psi=E \psi\)
where \(\hat{\mathrm{H}}\) = Mathematical operator, called Hamiltonian operator
Ψ = Wave function
E = Energy
(D) According to the de Broglie equation λ = \(\frac{h}{m \times v}\)
for same value of velocity λ = \(\frac{1}{m}\)
∴ O2 molecule has shortest wavelength.
(E) ∆E = E∞ – E1
= 0 – (- 1312 kJ mol-1)
= 1312 kJ mol-1
(F) Number of waves in nth orbit
= \(\frac{\text { Circumference of the orbit }}{\text { Wavelength }}\)
= \(\frac{2 \pi r}{\lambda}=\frac{2 \pi r}{h / m v}=\frac{2 \pi m v r}{h}\)
= \(\frac{2 \pi\left(\frac{n h}{2 \pi}\right)}{h}\) = n
(G) Since the effective nuclear charge decreases as the distance of the sub-shell increases from the nucleus. So, increasing order of effective nuclear charge (Zeff) is d < p < s.
Question 32.
(A) Define hydrogen bond. Is it weaker or stronger than the van der Waals forces?
(B) What is meant by the term bond order?
(C) Calculate the bond order of 02, 02 , and 02 .
OR
(A) Write the important conditions required for the linear combination of atomic orbitals to form molecular orbitals.
(B) Use molecular orbital theory to explain why Be2 molecule does not exist?
Answer:
(A) Hydrogen bond: It is defined as an attractive force which binds hydrogen atom of one molecule with the electronegative atom (F, O or N) of the same or another molecule. e.g., HF
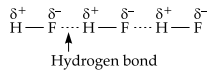
Hydrogen bond is stronger than the van der Waals forces.
(B) Bond order is given by the number of bonds between the two atoms in a molecule. =
[Nb – Na] where Nb = number of electrons occupying the anti-bonding orbitals.
(C) Bond order of N2:
Molecular orbital of N2 : KK(σ2s)2 (σ*2s)2 (π2px)2 = (π2py)2 (σ2pz)2
B.O = \(\frac{N_b-N_a}{2}=\frac{10-4}{2}=\frac{6}{2}\) = 3
Bond order of O2:
Molecular orbital of O2: KK(σ2S)2 (σ*2S)2 (σ2pz)2 (n2px)2 = (π2py)2 (π*2px)1 = (π*2py)1
Bond order of O2 = \(\frac{\mathrm{N} b-\mathrm{N} a}{2}\)
= \(\frac{10-6}{2}=\frac{4}{2}\) = 2
Bond order of O2+:
M.O. of O2+: KK(σ2S)2 (σ*2S)2 (σ2pz)2 (σ2px)2 = (σ2py)2 (σ*2px)1
Bond order of O2+ = [/latex]\frac{\mathrm{N} b-\mathrm{N} a}{2}[/latex]
= \(\frac{10-7}{2}=\frac{3}{2}\) = 1.5
or
(A) The necessary conditions for the linear combination of atomic orbitals to form molecular orbitals are as follows:
- The combining atomic orbitals must have almost equal energies. e.g., 1S – 1S, 2S – 2S, 2pz – 2pz, etc.
- The combining atomic orbitals must have the same symmetry about the molecular axis. e.g., 2pz orbital can combine with 2pz orbital but not with 2px or 2py orbitals.
- The combining atomic orbitals must overlap to the maximum extent.
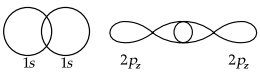
(B) Electronic Configuration:
4Be = 2, 2 = 1s2, 2s2
4Be = 2, 2 = 1s2, 2s2
Molecular Orbital Configuration:
(σ1S)2, (σ*1S)2, (σ2S)2, (σ*2S)2
Molecular Orbital Diagram :
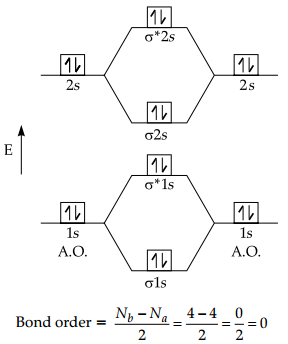
Since, the bond order of Be2 is zero so Be2 molecule does not exist.
Question 33.
How will you estimate the halogens?
OR
(A) Give condensed and bond line structural formulas and identify the functional group(s) present, if any, for:
(i) 2,2,4-Trimethylpentane
(ii) 2-Hydroxy-l, 2, 3-propanetricarboxylic acid
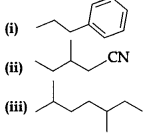
(B) Give the IUPAC names of the following compounds:
Answer:
Carius method:
A known mass of an organic compound is heated with fuming nitric acid in the presence of silver nitrate contained in a hard glass tube known as Carius tube, in a furnace. Carbon and hydrogen present in the compound are oxidised to carbon dioxide and water. The halogen present forms the corresponding silver halide (AgX). It is filtered, washed, dried and weighed. Let the mass of organic compound taken = mg Mass of AgX formed = m1g 1 mol of AgX contains 1 mol of X Mass of halogen in m1 g of AgX = Atomic mass of X × m1 g Molecular mass of AgX 1 Percentage of halogen = Atomic mass of X × m ×100/molecular mass of AgX × m.
OR
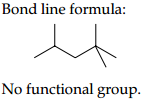
(A) 2, 2 ,4-Trimethylpentane
Condensed formula-(CH3)2CHCH2C(CH3)3
(ii) 2-hydroxy-1, 2, 3-propanetricarboxylic acid Condensed formula-(COOH) CH2C(OH)
(COOH)CH2(COOH)
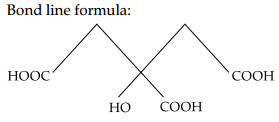
Functional groups present in compound are -COOH and -OH.
(b) (i) 1-phenyl propane
(ii) 3-methylpentane nitrile
(iii) 2, 5-Dimethylheptane