Students can access the CBSE Sample Papers for Class 11 Chemistry with Solutions and marking scheme Set 3 will help students in understanding the difficulty level of the exam.
CBSE Sample Papers for Class 11 Chemistry Set 3 with Solutions
Time Allowed : 3 hours
Maximum Marks: 70
General Instructions:
- There are 33 questions in this question paper with internal choice.
- Section – A consists of 16 multiple -choice questions carrying 1 mark each.
- Section – B consists of 5 short answer questions carrying 2 marks each.
- Section – C consists of 7 short answer questions carrying 3 marks each.
- Section – D consists of 2 case – based questions carrying 4 marks each.
- Section – E consists of 3 long answer questions carrying 5 marks each.
- All questions are compulsory.
- Use of log tables and calculators is not allowed.
Section-A
The following questions are multiple-choice questions with one correct answer. Each question carries 1 mark. There is no internal choice in this section.
Question 1.
Electromagnetic radiations differ from each other in ……… and wavelength.
(A) frequency
(B) time period
(C) wave velocity
(D) none of the above
Answer:
(A) frequency
Explanation:
Electromagnetic radiations differ from each other in frequency and wavelength.
Question 2.
Which of the following statements about the electron is incorrect?
(A) It is a negatively charged particle.
(B) The mass of electron is equal to the mass of neutron.
(C) It is a basic constituent of all atoms.
(D) It is a constituent of cathode rays.
Answer:
(B) The mass of electron is equal to the mass of neutron.
Explanation:
The mass of electron is very small in comparison to the mass of neutron.
![]()
Question 3.
Which of the following is responsible to rule out the existence of definite paths or trajectories of electrons?
(A) Pauli’s exclusion principle.
(B) Heisenberg’s uncertainty principle.
(C) Hund’s rule of maximum multiplicity.
(D) Aufbau principle.
Answer:
(B) Heisenberg’s uncertainty principle.
Explanation:
According to Heisenberg’s uncertainty principle, the position and velocity of an electron cannot be determined simultaneously with accuracy which rules out the existence of fixed paths.
Question 4.
Which of the following conclusions could not be derived from Rutherford’s a-particle scattering experiment?
(A) Most of the space in the atom is empty.
(B) The radius of the atom is about 10 ® m while that of nucleus is 10 15 m.
(C) Electrons move in a circular path of fixed energy called orbits.
(D) Electrons and the nucleus are held together by electrostatic forces of attraction.
Answer:
(C) Electrons move in a circular path of fixed energy called orbits.
Explanation:
The concept of movement of electrons in circular path of fixed energy called orbit was given by Bohr.
Question 5.
The enthalpy of combustion of methane, graphite and dihydrogen at 298 K are – 890.3 kj mol-1, -393.5 kj mol-1 and -285.8 kj mol-1 respectively. Enthalpy of formation of CH4(g) will be:
(A) – 74.8 kj mol-1
(C) – 52.27 kj -1
(B) + 74.8 kj mol-1
(D) + 52.26 kj mol-1
Answer:
(A) – 74.8 kj mol-1
Explanation:
CH4(g) + 2O2(g) → CO2(g) + 2H2O; ∆H = -890.3 kJ/mol
C(s) + O2(g) → CO2(g); ∆H = -393.5 kJ/mol
H2(g) + 1/2O2(g) → H2O(g); ∆H = -285.8 kJ/mol
Therefore, for eqn. C(s) + 2H2(g) → CH4(g); ∆H = ?
– 393.5 + 2(-285.8) – (-890.3); ∆H = -74.8 kJ/mol
Question 6.
A reaction A + B → + D + q is found to have a entropy change. The reaction will be:
(A) possible at high temperature.
(B) possible only at low temperature.
(C) not possible at any temperature.
(D) possible at any temperature.
Answer:
(D) possible at any temperature.
Explanation:
Since, the reaction is exothermic
(∆H < 0) and has ∆S > 0, therefore, it is feasible at all temperatures because both the factors favour spontaneity.
![]()
Question 7.
For an ideal gas, the work of reversible expansion under isothermal condition ‘can be calculated by using the expression
w = -nRT In — \(\frac{\mathrm{V}_f}{\mathrm{~V}_i}\)
A sample containing 1.0 mol of an ideal gas is expanded isothermally and reversibly to ten times of its original volume, in two separate experiments. The expansion is carried out at 300 K and at 600 K respectively. Choose the correct option.
(A) Work done at 600 K is 20 times the work done at 300 K.
(B) Work done at 300 K is twice the work done at 600 K.
(C) Work done at 600 K is twice the work done at 300 K.
(D) Work done at 300 K is 20 times the work done at 600 K.
Answer:
(C) Work done at 600 K is twice the work done at 300 K.
Explanation:
\(\frac{w_{600 K}}{w_{300 K}}\) = \(\frac{1 \times \mathrm{R} \times 600 \mathrm{~K} \ln \frac{10}{1}}{1 \times \mathrm{R} \times 300 \mathrm{~K} \ln \frac{10}{1}}\)
= \(\frac{600}{300}\) = 2
For isothermal expansion of ideal gases, ∆U = 0 Since, temperature is constant this means there is no change in internal energy. Therefore, ∆U = 0
Question 8.
Which of the following has S = – ve?
(A) Adsorption
(B) Dissolution of NH4C1 in water
(C) H2 → 2H
(D) 2NaHCO3,(s) → Na2CO3 + CO2 + H2O
Answer:
(A) Adsorption
Explanation:
In case of adsorption, entropy decreases due to attraction between adsorbent and adsorbate.
Question 9.
Non-metallic elements have:
(A) positive oxidation number
(B) negative oxidation number
(C) positive or negative oxidation number
(D) no defined oxidation number
Answer:
(C) positive or negative oxidation number
Explanation:
Non-metallic elements have both positive and negative oxidation numbers.
![]()
Question 10.
The equilibrium Solid → Liquid → Gas will shift in forward direction when:
(A) temperature is increased.
(B) temperature is lowered.
(C) pressure is increased.
(D) pressure is lowered.
Answer:
(A) temperature is increased.
Explanation:
Increase in temperature will favour the forward reaction which is endothermic.
Question 11.
Ethyne on passing through red hot iron tube at 873K undergoes cyclic polymerisation. Three molecules of ethyne polymerise to form:
(A) ethane
(B) ethene
(C) benzene
(D) ethanol
Answer:
(C) benzene
Question 12.
The addition of HC1 to an alkene proceeds in two steps. The first step is the attack of H+ ion to ![]() portion which can be shown as
portion which can be shown as

Answer:
![]()
Explanation:
Since double bond is a source of electrons and the charge flows from source of more electron density. Therefore, electrons of the double bond attack the proton.
Question 13.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Generally, ionisation enthalpy increases from left to right in a period.
Reason (R): When successive electrons are added to the orbitals in the same principal quantum level, the shielding effect of inner core of electrons does not increase very much to compensate for the increased attraction of the electron to the nucleus.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A
Question 14.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): The increase in energy (AU) for the vaporization of one mole in water at 1 atm and 373 K is zero. Reason (R): For gaseous isothermal processes, AU = 0.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A
Explanation:
For all isothermal process, internal energy is zero.
![]()
Question 15.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): A solution containing a mixture of acetic acid and sodium acetate maintains a constant value of pH on addition of small amounts of acid or alkali.
Reason (R): A solution containing a mixture of acetic acid and sodium acetate acts as a buffer solution around pH 4.75.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A
Question 16.
Given below are two statements labelled as Assertion (A) and Reason (R)
Assertion (A): Pent-l-ene and pent-2-ene are position isomers.
Reason (R): Position isomers differ in the position of functional group or substituent.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A ,
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A ,
Section-B
This section contains 5 questions with internal choice in one question. The following questions are very short answer type and carry 2 marks each.
Question 17.
How many significant figure are present in the folloing
(A) 0.0123 + 0.8250 + 0.025
(B)\( \frac{0.025 \times 298.15 \times 0.1155}{0.5785}\)
Answer:
17. (A) 0.0125 + 0.8250 + 0.025 = 0.8625 = 0.863
∵ 0.025 has three digits including zero after the decimal point so, the result should be reported only upto three digits after the decimal point. Therefore, there are three significant figures in the answer.
(B)\(\frac{0.025 \times 298.15 \times 0.1155}{0.5785}\) = \(\frac{0.860908125}{0.5785}\)
= 1.48817 = 1.49
∵ 298.15 has two digits after the decimal point so, the result should be reported only upto two digits after the decimal point. So, there are three significant figures.
Question 18.
Cations are smaller than neutral atom whereas anions are larger in size than neutral atom. Explain.
Answer:
Cations are smaller than neutral atom due to presence of more number of protons than number of electrons in cations. So, coulombic force increases and size decreases.
Whereas anions are larger than neutral atom due to more number of electrons than number of protons in anions, coulombic force decreases and size increases.
Question 19.
How many electrons in an atom may have the following quantum numbers?
Answer:
(A) For n = 4
Total number of electrons
= 2n2 = 2(4)2 = 32
Half of these electrons will have
ms = -1/2
∴ Total electrons with ms (-1/2) = 16
(B) For n = 3
l = 0
ml = 0
ms = +1/2, -1/2 (two electrons)
There are two electrons for this set of quantum number.
![]()
Question 20.
Write the name of the isomerism shown by the following pairs:
(A) Buta-1,3-diene and But-l-yne (C4H6)
(B) Ethoxy butane and Propoxy propane (C6H14O)
OR
Draw possible functional isomers for the following molecular formula:
(A) C3H6O (B) C2H6O
Answer:
(A) Functional isomerism
H2C = CH – CH = CH2 and HC = C – CH2CH3
(B) Metamerism
CH3 – CH2 – CH2 – CH2 – O – CH2CH3 and C3H7OC3H7
Question 21.
(A) How will you convert: ethyne to but-2- yne
(B) Draw the geometrical isomers of 2, 3-dichlorobut-2-ene
Answer:
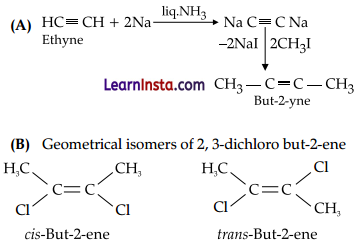
Section-C
This section contains 7 questions with internal choice in one question. The following questions are short answer type and carry 3 _ marks each. (No internal choice given in Board SQP)
Question 22.
What are:
(A) representative elements
(B) transition elements
(C) Lanthanoid and actinoids.
Give their position in modern periodic table.
Answer:
(A) Representative Elements: Group 1, 2, 13, 14, 15, 16, 17 and 18 exhibit the main groups of the periodic table. So, the elements of these groups are collectively called representative elements.
These elements belong to s-block and p-block in modern periodic table.
(B) Transition Elements: d-block transition metals form a bridge between the chemically active metals of s-block elements and the less active elements of Groups 13 and 14 are known as transition elements. (1)
(C) Lanthanoids and Actinoids: The elements of 4f series [i.e., from Ce (Z = 58) to Lu (Z = 71)] are called lanthanoids and the elements of 5f series [i.e., from Th (Z = 90) to Lr (Z = 103)] are called actinoids. These elements belong to f-block elements in the modern periodic table which lie at the bottom of the periodic table.
Question 23.
(A) A neutral atom has 2 K, 8L and 15 M electrons. Find the total number of electrons in s, p, d and/sub-shell.
(B) How many unpaired electrons are present in Al+ ion.
(Atomic number: A1 =13)
(C) One electron is present in 4f sub-shell. What is the sum of n, l, m, and ms values assuming f sub-shell follows -3 to +3 order of filling electron.
Answer:
(A) The electronic configuration of neutral atom having 2 K, 8L and 15 M electrons is 1S2, 2S2 2p6, 3s23p63d7
∴ Total number of electrons in s-subshell = 6
Total number of electrons in p-subshell = 12
Total number of electrons in D-subshell = 7
Total number of electrons in /-subshell = 0
(B) Al+ : Number of electrons = 13 – 1 = 12
Electronic configuration = 2, 8, 2
= 1s2, 2s22p6, 3s2
∴ No. of unpaired electrons in Al+ = 0
(C) For 4f subshell having one electron,
n = 4
l = 3
ml = – 3
ms = ±1/2
Sum = 4 + 3+ (- 3) + 1/2
= 41/2
Question 24.
(A) Explain molecular orbital?
(B) Write the molecular orbital electronic configuration of H2 molecule. Calculate its bond order and predict its magnetic behaviour.
Answer:
(A) Molecular orbital is an orbitals or wave function of a molecules electron. It tells us about the probability of finding an electron within a specified location.
(B) Electronic configuration of H2 is: σ1s22
(C) It is diamagnetic in nature
Bond order
= \(\frac{\text { No. of } e^{-} \text {in bonding }- \text { No. of } e^{-} \text {in antibonding }}{2}\)
= \(\frac{2-0}{2}\) = 1
Hence, bond order in H2 is 1.
![]()
Question 25.
Whenever a reaction between an oxidizing agent and an reducing agent is carried out, a compound of lower oxidation state is formed if the reducing agent is in excess and a compound of higher oxidation state is formed if the oxidising agent is in excess. Justify this statement giving three illustrations.
Answer:
C = Reducing agent
O2 = Oxidising agent
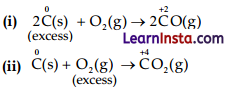
So, when carbon (reducing agent) is taken in excess, a compound, i.e., CO of lower oxidation state is obtained, if O2 (oxidising agent) is taken in excess, a compound (CO2) of higher oxidation state is formed.
(B) P4 = Reducing agent
Cl2 = Oxidising agent

(C) Na = Reducing agent
O2 = Oxidising agent
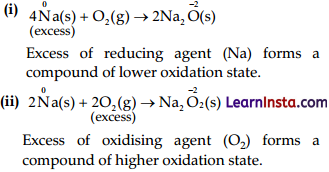
Question 26.
Explain why the following systems are not π aromatic?
![]()
Answer:
(A) For the given compound, the number of π-electrons is 6.
By Huckel’s rule,
4n + 2 = 6
4n = 4
n = 1
For a compound to be aromatic, the value n must be an integer (n = 0, 1, 2…). Since, the value of n is an integer, it should be aromatic. However, for a compound to be aromatic p electron cloud should be equally distributed on all the ring, hence, it is non-aromatic in nature.
(B) For the given compound, the number of p-electrons is 4.
By Huckel’s rule,
4n + 2 = 4
4n = 2
n = 1/2
For a compound to be aromatic, the value of n must be an integer (n = 0, 1, 2…), which is not true for the given compound. Hence, it is not aromatic in nature.
(C) For the given compound, the number of π-electrical is 8.
By Huckel’s rule,
4n + 2 = 8
4n = 6
n = 3/2
For a compound to be aromatic, the value of n must be an integer (n = 0, 1, 2…). Since the value of n is not an integer, the given compound is not aromatic in nature.
![]()
Question 27.
Discuss the chemistry of Lassaigne’s test.
Answer:
This test is employed to detect the presence of nitrogen, sulphur, halogens and phosphorus in an organic compound. These elements are present in the covalent form in an organic compound. These are converted into the ionic form by fusing the compound with sodium metal.
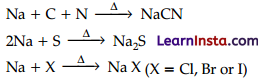
The cyanide, sulphide, and halide of sodium formed are extracted from the fused mass by boiling it in distilled water. The extract so obtained is called Lassaigne’s or sodium fusion extract. This Lassaigne’s or sodium fusion extract is then tested for the presence of nitrogen, sulphur, halogens, and phosphorus.
Question 28.
In the alkane H3C – CH2 – C(CH3)2 – CH2 – CH(CH3)2. Identify r,2°,3°carbon atoms and give the number of H atoms bonded to each one of these.
Answer:
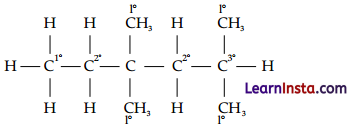
1° carbon atoms are those which are bonded to only one carbon atom, i.e., they have only one carbon atom as their neighbour. The given structure has five 1° carbon atoms and fifteen hydrogen atoms attached to it.
2° carbon atoms are those which are bonded to two carbon atoms, i.e., they have two carbon atoms as their neighbours. The given structure has two 2° carbon atoms and four hydrogen atoms attached to it.
3° carbon atoms are those which are bonded to three carbon atoms, i.e., they have three carbon atoms as their neighbours. The given structure has one 3° carbon atom and only one hydrogen atom is attached
Section-D
The following questions are case-based questions. Each question has an internal choice and carries 4 ( 1 + 1 + 2) marks each.
Question 29.
Read the passage given below and answer the following questions:
The hydrogen atom makes a weak connection with the electronegative atom of the other molecule when it is connected to an atom of a strongly electronegative element, such as fluorine, oxygen, or nitrogen. The hydrogen atom in the molecule of hydrogen fluoride, HF, establishes a weak link with the fluorine atom while still being attached to its own fluorine atom. The hydrogen bond is represented by a dotted line (…). Each water molecule in the ice crystal structure forms an H-bond with four other water molecules to form a cage-like tetrahedral structure.
(A) Why water has highest density at 4°C?
OR
What are the different types of hydrogen bonds?
(B) Name any two compounds that contain inter-molecular hydrogen bonding other than water.
(C) What is the strength of hydrogen bond? What determines the dissociation energy of the hydrogen bond?
Answer:
(A) H-bonds in the ice are broken during melting, resulting in smaller spaces between water molecules and an up to 4°C rise in water density. More H-bonds are broken above 4°C. As the water molecules separate from one another, the density once more drops. So, at 4°C, water has its highest density.
OR
Hydrogen bonds can be classified into two types:
(i) Intermolecular hydrogen bond:
Intermolecular hydrogen bond is formed between different molecules of the same or different substances.
(ii) Intramolecular hydrogen bond:
Intramolecular hydrogen bond is formed when the hydrogen atom is between the two highly electronegative atom (F, O or N) present within the same molecule.
(B) Ammonia and ethanol
(C) The hydrogen bond is a weak bond. The strength
of the hydrogen bond lies between the covalent bonds and the weak vander Waals forces. The attraction of the shared pair of electrons and, consequently, the atom’s electronegativity determine the dissociation energy of the hydrogen bond.
![]()
Question 30.
Read the passage given below and answer the following questions:
Entropy is a measure of degree of randomness. Entropy is directly proportional to temperature. Every system tries to acquire maximum state of randomness or disorder. Entropy is a measure of unavailable energy. Unavailable energy = Entropy x Temperature. The ratio of Entropy of Vaporization and boiling point of a substance remains almost constant.
(A) The entropy of steam is more than that of water at its boiling point?
(B) The standard free energy of reaction is found to be zero. What is the value of equilibrium constant K?
(C) Water decomposes by absorbing 286.2 kj of electrical energy per mole. When H2 and 02 combine to form one mole of H20,286.2 kj of heat is produced, Which thermodynamic law is proved? Write its statement.
OR
Define standard enthalpy of solution and enthalpy of hydration.
Answer:
(A) Entropy of steam is more than that of water at its boiling point because in steam, the molecules can move freely so there is more disorder.
(B) ∵ ΔG° = – 2.303 RT log K
Given ΔG° = 0
∴ 2.303 RT log K = 0
or log K = 0
or K = 1
(C) H2O(g) → H2(g) + 1/2O2(g); ΔrHθ = 286.2 kJ mol-1
H2(g) + 1/2O2(g) → H2O(s); ΔrHθ = – 286.2 kJ mol-1
It is referred to as first law of thermodynamics. According to this law, “Energy can neither be created nor be destroyed but can be converted from one form to another”.
OR
Enthalpy of Solution (ΔsolH°):
It is the enthalpy change taking place when 1 mole of the solute is dissolved in specified amount of solvent. The enthalpy of solution at infinite dilution is the enthalpy change observed on dissolving the substance in an infinite amount of solvent when the interactions between the ions (or solute molecules) are negligible.
Enthalpy of Hydration (ΔhydH°):
It is the enthalpy change occurring during the hydration of 1 mole of anhydrous salt by combining with specific number of moles of water.
Section-E
The following questions are long answer type and carry 5 marks each. All questions have an internal choice.
Question 31.
Attempt any five questions:
(A) What is meant by homogeneous equilibrium? Give an example.
(B) For which equilibrium, the vapour pressure is constant at a given temperature.
(C) At 450 K, KP = 2.0 x 1010/bar for the given reaction at equilibrium.
2SO2(g) + O2(g) ⇌ 2SO3(g) + 189.4 kj What is Kc at this temperature?
(D) Give the direction in which the reaction would proceed if Qp = Kc.
(E) The value of ∆Gθ for the phosphorylation of glucose in glycolysis is 13.8 kj/mol. Find the value of Kc at 298 K.
(F) Hydrolysis of sucrose gives,
Sucrose + H2O ⇌ Glucose + Fructose
Equilibrium constant Kc for the reaction is 2 × 1013 at 300 K. Calculate AGe at 300 K.
(G) What is the effect of increasing pressure on the equilibrium state of the reaction?
N2(g) + 3H2(g) → 2NH3(g) + Heat
Answer:
(A) Homogeneous Equilibrium:
Equilibrium in a system having only one phase is called homogeneous equilibrium.
(B) For liquid ⇌ vapour equilibrium, the vapour pressure is constant at a given temperature.
e.g. N2(g) + 3H2(g) ⇌ 2NH3(g)
(C) 2SO2(g) + O2(g) ⇌ 2SO3(g)
For the given reaction
∆ng = np – nr
= 2 – 3 = – 1
Kp = Kc (RT)∆n
Kp = 2 × 1010 bar-1
Kc = \(\frac{\mathrm{K}_p}{(\mathrm{RT})^{\Delta n}}\)
= \(\frac{\mathrm{K}_p}{(\mathrm{RT})^{-1}}\)
= Kp × (RT)
= 2 × 1010 bar-1
(0.0831 L bar K-1 mol-1) × (450 K)
= 7.48 × 1011 L mol-1
(D) If Qc = Kc, the reaction is in state of equilibrium.
(E) Given,
∆Gθ = 13.8 Kj/mol
13.8 × 103 J/mol
T = 298 K
R = 8.314 J mol-1 K-1
Kc = ?
∵ ∆Gθ = -RT in Kc
In Kc = – \(\frac{\Delta \mathrm{G}^{\circ}}{\mathrm{RT}}\)
= \(\frac{-13.8 \times 10^3 \mathrm{~J} / \mathrm{mol}}{8.314 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1} \times 298 \mathrm{~K}}\)
= – 5.569
or
Kc = e-5.569
= 3.81 × 10-3
(F) Given, Kc = 2 × 1013, T = 300K, ∆G° = ?
∵ ∆G° = – RT ln Kc
= – 8.314 J mol-1 K-1 × 300 K × ln (2 × 1013)
= – 7.64 × 104 J mol-1
(G) For the reaction,
N2(g) + 3H2(g) → 2NH3(g) + Heat
On increasing the pressure, the reaction will proceed in forward direction. This results in increase in yield of ammonia.
![]()
Question 32.
(A) Define the terms:
(i) Empirical formula
(ii) Molecular formula
(B) An organic compound on analysis gave the following data: C = 57.82%, H = 3.6% and the rest oxygen. If the molecular mass was found to be 166 u then calculate its empirical formula and molecular formula.
(C) Calculate the mole fraction of ethanol if 9.2 g of it is mixed with 1.8 g water.
Or
(A) What is the difference between molarity and molality?
(B) The molarity of a solution of sulphuric acid is 1.35 M. Calculate its molality. (The density of acid solution is 1.02 g-3 cm.
Answer:
(A) (i) Empirical formula represents the simple whole number ratio of atoms of all elements present in a molecule of the compound.
(ii) Molecular formula gives the actual number of atoms of various elements present in one molecule of compound.
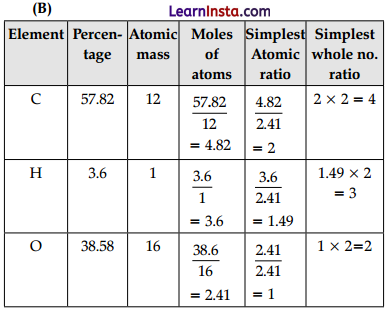
Empirical formula = C4H3O2 (1)
Empirical formula mass = 12 × 4 + 1 × 3 + 16 × 2 = 83
Molecular mass = 166 U
n = \(\frac{\text { Molecular mass }}{\text { Empirical formula mass }}\)
= \(\frac{166}{83}\) = 2
Molecular formula = (C4H3O2)2
= C8H6O4
(C) Mass of ethanol = 9.2 g
Molar mass of C2H5OH
= 2 × 12 + 5 × 1 + 16 + 1
= 24 + 5 + 16 + 1 = 46
Mass of H2O = 1.8 g
Molar mass of H2O = 2 × 1 + 16 = 18
Number of moles of ethanol = \(\frac{9.2}{46}\) = 0.2
Number of moles of water = \(\frac{1.8}{18}\) = 0.1
Mole fraction of ethanol = \( \frac{n_{\text {ethanol }}}{n_{\text {water }}+n_{\text {ethanol }}}\)
= \(\frac{0.2}{0.1+0.2}\) = \(\frac{0.2}{0.3}\)
= 0.666 ≈ 0.67
Mole fraction of water = 1 – 0.67 = 0.33
Or
(A)
| Molarity | Molality |
| It is defined as the number of moles of the solute in 1 litre of the solution. | It is defined as the number of moles of solute present in 1 kg of solvent. |
| It is affected by the change in temperature. | It is not affected by the change in temperature. |
(B) Molarity = 1.35 M, Molecular mass of H2SO4 = 2 + 32 + 64 = 98
Mass of H2SO4 in 1 L solution = 1.35 × 98 = 132.3 g
Mass of 1 L solution = 1000 × 1.02
= 1020 g (∵ Density = 1.02 g cm-3)
Mass of water in solution = 1020 – 132.3
= 887.7 g = 0.888 kg
Molality = \(\frac{\text { Number of moles of solute }}{\text { Mass of solvent in } \mathrm{kg}}\)
= \(\frac{1.35}{0.888}\)
= 1.52 m
![]()
Question 33.
An organic compound contains 69% carbon and 4.8% hydrogen, the remainder being oxygen. Calculate the masses of carbon dioxide and water produced when 0.20 g of this substance is subjected to complete combustion.
OR
(A) (i) What is retardation factor?
(ii) Which elements are detected by Lassaigne’s test?
(B) Give condensed and bond line structural formulas and identify the functional group(s) present, if any, for:
(i) 2,2,4-Trimethylpentane
(ii) 2-Hydroxy-1,2, 3-propanetricarboxylic acid
(iii) Hexanedial
Answer:
Percentage of carbon in organic compound = 69 % That is i.e., 100 g of organic compound contains 69 g of carbon.
0.2 g of organic compound will contain
\(\frac{69 \times 0.2}{100}\) = 0.138 g of C
Molecular mass of carbon dioxide, CO2 = 44 g
i. e., 12 g of carbon is contained in 44 g of CO2. Therefore, 0.138 g of carbon will be contained in \(\frac{44 \times 0.138}{12}\) = 0.506 g of CO2
Thus, 0.506 g of CO2 will be produced on complete combustion of 0.2 g of organic compound.
Percentage of hydrogen in organic compound is 4.8. i.e., 100 g of organic compound contains 4.8 g of hydrogen.
Therefore, 0.2 g of organic compound will contain
\(\frac{4.8 \times 0.2}{100}\) = 0.0096 g of H
It is known that molecular mass of water (H2O) is 18 g.
Thus, 2g of hydrogen is present in 18 g of water.
0.0096 g of hydrogen will be contained in \(\frac{18 \times 0.0096}{2}\) = 0.0864 g of water
Thus, 0.0864 g of water will be produced on complete combustion of 0.2 g of the organic compound. (5)
OR
(A) (i) The relative adsorption of each component of the mixture is expressed in terms of its retardation factor i.e.,
Rf value = Distance moved by the substance from base line (x)/Distance moved by the solvent from base line (y)
(ii) Nitrogen, sulphur, halogens and phosphorus present in organic compound are detected by ‘Lassaigne’s test.
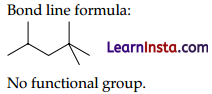
(B) (i) 2, 2 ,4- Trimethylpentane Condensed formula-(CH3)2CHCH2C(CH3)3 Bond line formula:
(ii) 2-hydroxy-1, 2, 3-propanetricarboxylic acid
Condensed formula-(COOH) CH2C(OH) (COOH)CH2(COOH)
Bond line formula:
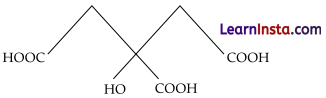
Functional groups present in the given compound are -COOH and – OH.
(iii) Hexanedial:
Condensed formula-(CHO)(CH2)4(CHO) Bond line formula:
![]()
Functional group present in the given compound is – CHO.