Students can access the CBSE Sample Papers for Class 11 Chemistry with Solutions and marking scheme Set 1 will help students in understanding the difficulty level of the exam.
CBSE Sample Papers for Class 11 Chemistry Set 1 with Solutions
Time Allowed : 3 hours
Maximum Marks: 70
General Instructions:
- There are 33 questions in this question paper with internal choice.
- Section – A consists of 16 multiple -choice questions carrying 1 mark each.
- Section – B consists of 5 short answer questions carrying 2 marks each
- Section – C consists of 7 short answer questions carrying 3 marks each.
- Section – D consists of 2 case – based questions carrying 4 marks each.
- Section – E consists of 3 long answer questions carrying 5 marks each.
- All questions are compulsory.
- Use of log tables and calculators is not allowed.
Section-A
The following questions are multiple-choice questions with one correct answer. Each question carries 1 mark. There is no internal choice in this section.
Question 1.
A, B and C are three members of a Dobereiner’s triad. If the atomic mass of A is 7 and that of C is 39, what is the atomic mass of B?
(A) 22
(B) 24
(C) 23
(D) 25
Answer:
(C) 23
Explanation: Atomic mass of
B = \(\frac{Atomic mass of A + Atomic mass of C}{2}\)
= \(\frac{7+39}{2}\) = \(\frac{46}{2}\) = 23
Question 2.
The last element of the p-block in 6th period is represented by the outermost electronic configuration.
(A) 7s2 7p6
(B) 5f14 6d10 7s2 7p0
(C) 4f14 5d10 6s2 6p4
(D) 4f14 5d10 6s2 6p4
Answer:
(C) 4f14 5d10 6s2 6p4
Question 3.
In the modern periodic table, the period indicates the value of:
(A) atomic number
(B) atomic mass
(C) principal quantum number
(D) azimuthal quantum number
Answer:
(C) principal quantum number
Explanation:
In the modern periodic table, the period indicates the value of principal quantum number (n).
Question 4.
Arrange the bonds in order of increasing ionic character in the molecules: LiF, KzO, S02 and C1F3:
(A) N2 < SO2 < C1F3 < K2O < LiF
(B) SO2 < N2 < C1F3 < K2O < LiF
(C) K2O < N2 < SO2 < C1F3 < LiF
(D) SO2 < C1F3 < N2 < K2O < LiF
Answer:
(A) N2 < SO2 < C1F3 < K2O < LiF
Question 5.
Match the following processes with entropy change:
| Reaction | Entropy Change |
| (i) A liquid vapourises. | (a) ∆S = 0 |
| (ii) Reaction is non-spontaneous at all temperatures and AH is positive. | (b) ∆S = positive |
| (iii) Reversible expansion of an ideal gas. | (c) ∆S = negative |
Choose the correct option from below.
(A) (i)-(b), (ii)-(c), (iii)-(a)
(B) (ii)-(b), (iii)-(c), (i)-(a)
(C) (i)-(b), (iii)-(c), (ii)-(a)
(D) (iii)-(b), (i)-(c), (ii)-(a)
Answer:
(A) (i)-(b), (ii)-(c), (iii)-(a)
![]()
Question 6.
The entropy change can be calculated by using the expression ∆S = \(\frac{q_{\mathrm{rev}}}{\mathrm{T}}\). When water freezes in a glass beaker,choose the correct statement amongst the following:
(A) ∆S (system) decreases but AS (surroundings) remains the same.
(B) ∆S (system) increases but AS (surroundings) decreases.
(C) ∆S (system) decreases but AS (surroundings) increases.
(D) AS (system) decreases and AS (surroundings) also decreases.
Answer:
(C) ∆S (system) decreases but AS (surroundings) increases.
Explanation:
Freezing is exothermic process. The heat released increases the entropy of surroundings.
Question 7.
What wiU be the value of pH of 0.01 mol dm-3 CH3COOH(Ka = 1.74 × 10-5)?
(A) 3.4
(B) 3.6
(C) 3.9
(d) 3.0
Answer:
(A) 3.4
Explanation:

Question 8.
Which of the following options will be correct for the stage of half completion of the reactions A ⇌ B?
(A) ∆Gθ = 0
(B) ∆Gθ > 0
(C) ∆Gθ < 0
(D) ∆Gθ = -RT In 2
Answer:
(A) ∆Gθ = 0
Explanation:
∆Gθ = -RT ln K
At the stage of half completion of reaction [A] = [B]
Therefore, K = 1.
Thus, ∆Gθ = 0
Question 9.
Identify the correct statement(s) in relation to the following reaction:
Zn + 2HCl → ZnCl2 + H2
(A) Zinc is acting as an oxidant.
(B) Chlorine is acting as a reductant.
(C) Hydrogen ion is acting as an oxidant.
(D) Zinc is acting as a reductant.
Answer:
(C) Hydrogen ion is acting as an oxidant. and
(D) Zinc is acting as a reductant.
Explanation:
![]()
- As the oxidation number increases from 0 to +2, Zn act as reductant not as oxidant.
- As the oxidation number of Cl remains the same, it neither acts as reductant nor as an oxidant.
- As oxidation number decreases from +1 to 0 in H2, it acts as an oxidant.
Question 10.
In the organic compound CH2 = CH – CH2 – CH2 – C = CH, the pair of hybridised orbitals involved in the formation
– of C2 – C2 is:
(A) sp-sp2
(B) sp – sp2
(C) sp2 – sp3
(D) sp3 – sp3
Answer:
(C) sp2 – sp3
Explanation:
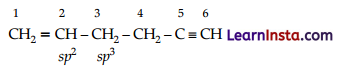
Question 11.
In an electrophilic substitution reaction of nitrobenzene, the presence of nitro group
(A) activates the ring by an inductive effect.
(B) decrease the charge density at ortho- and para-positions of the ring relative to mefa-position by resonance.
(C) increases the charge density at meta-position relative to the ortho and para-positions of the ring by resonance.
(D) None of the above
Answer:
(C) increases the charge density at meta-position relative to the ortho and para-positions of the ring by resonance.
Explanation:
Nitro group by virtue of -I effect withdraws electrons from the ring and increases the charge and destabilises carbocation. In ortho, para- attack of electrophile on nitrobenzene, we are getting two structures in which positive charge is appearing on the carbon atom directly attached to the nitro group. As nitro group is electron withdrawing by nature, it decreases the stability of such product and hence, meta attack is more feasible when electron withdrawing substituents are attached.
Question 12.
Choose the one which is not an example of arenes:
(A) Toluene
(B) Biphenyl
(C) Propanone
(D) Naphthalene
Answer:
(C) Propanone
Explanation: Propanone (CH3COCH3) is not an example of arenes as it does not contain benzene ring.
Question 13.
Given below are two statements labelled as Assertion (A) and Reason (R)
Assertion (A): Among the two O-H bonds in H20 molecule, the energy required to break the first O-H bond and the other O-H bond is the same.
Reason (R): This is because the electronic environment around oxygen is the same even after breakage of one O-H bond.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A.
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(D) A is false but R is true.
Explanation:
In case of H2O molecule, the enthalpy needed to break the two O – H bonds is not the same. The difference in the ∆H° value shows that the second O-H bond undergoes some change because of changed chemical environment.
![]()
Question 14.
Given below are two statements labelled as Assertion (A) and Reason (R)
Assertion (A): Combustion of all organic compounds is an exothermic reaction.
Reason (R): The enthalpies of all elements in their standard state are zero.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A.
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(B) Both A and R are true but R is not the correct explanation of A.
Explanation: Reason (R): Combustion reactions breaks the bonds of organic compound molecules, and the resulting water and carbon dioxide bonds always release more energy than was used to break them originally. That’s why burning organic compounds produces energy and is exothermic.
Question 15.
Given below are two statements labelled as Assertion (A) and Reason (R)
Assertion (A): A liquid crystallises into a solid and is accompanied by decrease in entropy.
Reason (R): In crystals, molecules organise in an ordered manner.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A.
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A.
Explanation:
Entropy of system decreases when the liquid changes into solid. In crystalline state, molecules are ordered having less randomness.
Question 16.
Given below are two statements labelled as Assertion (A) and Reason (R)
Assertion (A): (CH3)3C+ carbocation is more easily formed than (CH3)2 +CH carbocation.
Reason (R): Tertiary butyl carbocation is more stable than isopropyl carbocation.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A.
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A.
Explanation:
(CH3)3C+ carbocation gets the positive charge dispersed by donating three electron donating methyl groups while in (CH3)2CH+ ion, there is lesser of dispersal of charge. So, (CH3)3C+ is more stable than (CH3)3CH+.
Section-B
This section contains 5 questions with internal choice in one question. The following questions are very short answer type and carry 2 marks each.
Question 17.
If ten volumes of dihydrogen gas react with 5 volumes of dioxygen gas.
(A) How many volumes of water vapour would be produced?
(B) Name the associated law with this observation.
Answer:
(A) When 2 volumes of dihydrogen react with 1 volume of oxygen, 2 volumes of water is produced:
![]()
Therefore, when 10 volumes of dihydrogen react with 5 volumes of oxygen, 10 volumes of water is produced.
(B) This observation is associated with the Gay Lu ssac’s law of gaseous volumes. According to this law, the ratio between the volumes of the reactant gases and the products can be expressed in simple whole numbers.
Question 18.
Heisenberg uncertainty principle has no significance in our everyday life. Explain?
Answer:
The effect of Heisenberg uncertainty principle is significant only for motion of microscopic objects and is negligible for that of macroscopic objects. This can be explained by following example – If uncertainty principle is applied to an object of mass, say about a milligram (10-6 kg).
∆v.∆x = \(\frac{h}{4 \pi \cdot m}\)
= \(\frac{6.626 \times 10^{-34} \mathrm{~J} \mathrm{~s}}{4 \times 3.1416 \times 10^{-6} \mathrm{~kg}}\)
= 5.27 × 10-27 m2s-1
∴ The value of AV.AX obtained is extremely small and is insignificant. So, this principle has no significance
in our everyday life.
Question 19.
(A) Describe the hybridisation in case of PC15?
(B) Why are the axial bonds longer as compared to the equatorial bonds?
Answer:
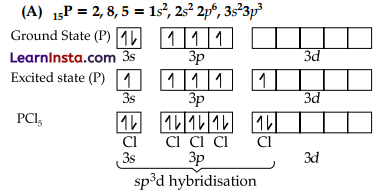
Five orbitals, i.e., one s, three p and one d-orbitals are available for hybridisation to form five sp3d hybrid orbitals in the formation of PCl5. These five sp3d hybrid orbitals are directed towards the five corners of trigonal bipyramidal.
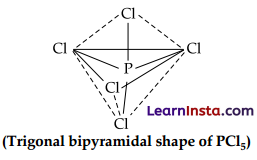
All the bond angles in this structure are not equivalent. In PCl5, three P-Cl bonds lie in one plane with bond angle of 120°. These bonds are called equatorial bonds. The remaining two P-Cl bonds, one lying above and other lying below the equatorial plane form an angle of 90° with the plane. These bonds are called axial bonds.
(B) Axial bonds are longer as compared to the equatorial bonds due to great repulsion on axial bond pair electrons by equatorial bond pair electrons.
Question 20.
(A) What is enthalpy of neutralisation?
(B) Given
N2(g) + 3H2(g) → 2NH3(g); ∆rH° = – 92.4 kj/mol.
What is the standard enthalpy of formation of ammonia gas? At i
OR
Find out the value of equilibrium constant for the following reaction at 298 K.
2NH3(g) + CO2(g) → NH2CONH2 (aq) + H2O(l)
Standard Gibbs energy change, at the given temperature is -13.6 kj mol-1.
Answer:
(a) Enthalpy of Neutralisation (∆nH):
It is the enthalpy change taking place during neutralisation of 1g equivalent of acid with 1g equivalent of a base in dilute aqueous solution
(b) Given: N2(g) + 3H2(g) → 2NH3(g);
∆fH° = -92.4 kJ mol-1
This is heat evolved for 2 moles for NH3(g)
∴ Heat evolved for 1 mole of NH3(g)
= \(\frac{-92.4}{2}\) = – 46.2 KJ
Hence, ∆fH° of NH3 gas = -46.2 KJ mol-1
Or
Given, T = 298 K
-∆rG = -13.6 KJ mol-1
R = 8.314 k-1 mol-1
K = ?
-∆rG = -2.303 RT log
or
log K = -∆rG/2.303 RT
= -(13.6 × 103)/2.303 × 8.314 × 298 = 2.38
K = antilog 2. 38
= 2.4 × 102
![]()
Question 21.
(A) The molar enthalpy of vaporization of acetone is weaker than water. Why?
(B) What will be value of AS for an isolated system if ∆U is zero?
Answer:
(a) Acetone do not have hydrogen bonds which is strong bonding due to presence of high intermolecular forces of attraction. Hence, enthalpy required for vaporization is less as compared to water.
(b) ∆U for an isolated system is always zero because there is no exchange of heat and matter between system and surrounding. DS is entropy which tends to increase in spontaneous reaction i.e., ∆S > 0.
Section-C
This section contains 7 questions with internal choice in one question. The following questions are short answer type and carry 3 marks each. (No internal choice given in Board SQP)
Question 22.
(A) Give the essential postulates of Bohr’s model of an atom.
(B) How did it explain?
(i) The stability of the atom,
(ii) Origin of the spectral lines in H-atom.
(C) What is the value of the Bohr’s radius for the first orbit of hydrogen atom?
Answer:
(A) The electron in the atom can move around the nucleus in a circular path of fixed radius and energy. These paths are called orbits, stationary states or allowed energy states. These orbits are arranged concentrically around the nucleus. Radiation can occur only when the electron jump from one orbit to another. The atom will be completely stable in the state with the smallest orbit, since there is no orbit of lower energy into which the electron can jump.
(B) According to Bohr’s model, radiation (energy) is absorbed if the electron moves from the orbit of smaller Principal quantum number to the orbit of higher Principal quantum number, whereas the radiation (energy) is emitted if the electron moves from higher orbit to lower orbit. It explains the origin of spectral lines in H-atom.
(C) rn = \(\frac{52.9\left(n^2\right)}{Z}\)pm
For first orbit of H-atom, n = 1
Atomic number (Z) of H-atom = 1
∴ r1 = \(\frac{52.9(1)^2}{1}\)
= 52.9 pm = 5.29 × 10-11m
Question 23.
Element Gr-17; Gr-18; Gr-1 in the sequence placed in modern periodic table.
(A) Which has negative electron gain enthalpy?
(B) Which has more metallic behaviour?
(C) Which has zero electronic behaviour?
Answer:
(A) Group 17 element has negative electron gain
enthalpy, because it increases from left to right in a period. However, in Group 18 element, extra electron does not added to neutral gaseous atom due to presence of completely filled valence shell.
(B) Group l element has more metallic behavior because it decreases from left to right in a period and group l element also possesses lowest I.E.
(C) Group 18 element has zero electronic behavior because in this case all electrons are paired.
Question 24.
(A) Distinguish between bonding and anti-bonding molecular orbital.
(B) Write the molecular orbital electronic configuration of N2 molecule. Calculate its bond order and predict its magnetic behaviour?.
Answer:
| Bonding molecular orbital | Anti-bonding molecular orbital |
| (i) Its energy is less than that of parent atomic orbitals. | Its energy is greater than that of parent atomic orbitals. |
| (ii) It is more stable than the parent atomic orbitals. | It is less stable than the parent atomic orbitals. |
| (iii) In bonding molecular orbitals, the probability of finding electrons is maximum. | In anti-bonding mo-lecular orbital, the probability of find¬ing electrons is min¬imum. |
| (iv) It contributes towards the shape of molecule. | It does not contrib¬ute towards the shape of molecule. |
(B) Molecular orbital configuration of N2:
(σls)2 (σ*ls)2 (σ2s)2 (σ*2s)2 (π2Px)2 = (σ2pz)2
Bond order = \(\frac{1}{2}\)(Nb – Na)
= \(\frac{1}{2}\)(10-4) = 3.
Magnetic behaviour – Diamagnetic due to absence of unpaired electrons.
![]()
Question 25.
The combustion of one mole of benzene takes place at 298 K and 1 atm. After combustion, CO2(g) and H2O(1) are produced and 3267.0 kj of heat is liberated. Calculate the standard enthalpy of formation, ∆3Hθ of benzene. Standard enthalpies of formation of CO2(g) and H2O(l) are -393.5 kj mol-1 and – 285.83 kj mol-1 respectively.
Answer:
Reaction for the formation of benzene,
6C(graphite,s) + 3H2(g) → C6H6(l), ∆fH° = ? …….(i)
The enthalpy of combustion of l mol of benzene is:
C6H6(l) + \(\frac{15}{2}\) O2(g) → 6CO2(g) + 3H2O(l); ∆cH°
= -3267 kj mol-1 …….(ii)
The enthalpy of formation of l mol of CO2(g)
C(graphite,s) + O2(g) → CO2(g); ∆fHθ = -393.5 Kj mol-1 …….(iii)
The enthalpy of formation of l mol of H2O(l) is :
H2(g) + \(\frac{1}{2}\)O2(g) → H2O(l); ∆fH°
= -2361 Kj mol-1…….(iv)
3H2(g) + \(\frac{3}{2}\)O2(g) → 3H2O(l); ∆fH°
= -857.49 KJ mol-1
On adding above equations,
6C(graphite/s) + 3H2(g) + \(\frac{15}{2}\)O2(g) → 6CO2(g)
+ 3H2O(l); ∆fH° = – 3218.49 kJ mol-1 …….(v)
On reversing eq. (ii)
6CO2(g) + 3H2O(l) → C6C2(l) + \(\frac{15}{2}\)O2(g); ∆fH° = 3267 Kj mol-1 …….(vi)
On adding equation (v) and (vi), we get 6C(graphite,s) + 3H2(g) → C6H6(l);
∆fHθ = 48.51 Kj mol-1
∴ Standard enthalpy of formation of benzene
(∆fH°) = 48.51 kJ mol-1
Question 26.
At 450 K, Kp = 2.0 × 1010/bar for the given reaction at equilibrium.
2SO2(g) + O2(g) ⇌ 2SO3(g) + 189.4 kj
(A) What is Kc at this temperature?
(B) What is the value of Kc for the reverse reaction at the same temperature?
(C) What would be the effect on equilibrium if more SO2 is added?
Answer:
(A) 2SO2(g) + O2(g) ⇌ 2SO3(g)
For the given reaction
∆ng = np – nr = 2 – 3 = -1
Kp = Kc(RT)∆n
Kp = 2 × 1010bar-1
Kc = \(\frac{\mathrm{K}_p}{(\mathrm{RT})^{\Delta n}}\)
= \(\frac{\mathrm{K}_p}{(\mathrm{RT})^{-1}}\)
= Kp × (RT)
= 2 × 1010 bar-1(0.0831 L bar K-1 mol1 × (450 K)
= 7.48 × 1011 L mol-1
(B) For reverse reaction,
Kc = \(\frac{1}{\mathrm{~K}_c(\text { forward reaction })}\)
= \(\frac{1}{7.48 \times 10^{11}}\)
= 1.34 × 10-12 L mol-1
(C) According to Le-Chatelier’s principle, when concentration of reactants increased the rate
of forward reaction increase. Equilibrium shifts in forward direction. Thus, on adding SO2, forward reaction rate increases and more SO3 is formed
Question 27.
Why does fluorine not show disproportionation reaction?
Answer:
In a disproportionation reaction, the same species is simultaneously oxidised as well as reduced. So, for such a redox reaction to occur, the reacting species must contain an element which has atleast three oxidation states.
The elements, in reacting species, is present in an intermediate state while lower and higher oxidation states are available for reduction and oxidation to occur (respectively).
Fluorine is the strongest oxidising agent. It does not show positive oxidation state. That’s why fluorine does not show disproportionation reaction.
Question 28.
An alkene ‘A’ contains three C – C, eight C – H s bonds and one C – C p bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write IUPAC name of ‘A’.
Answer:
As per the given information, A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. The formation of two moles of an aldehyde indicates the presence of identical structural units on both sides of the double bond containing carbon atoms. Hence, the structure of A’ can be represented as:
XC = CX
There are eight C-H s bonds. Hence, there are 8 hydrogen atoms in A’. Also, there are three C-C bonds. Hence, there are four carbon atoms present in the structure of ‘A’. Combining the inferences, the structure of A’ can be represented as:
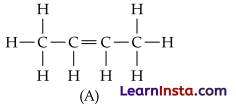
A’ has 3 C-C bonds, 8 C-H π bonds and one C – C π bond. Hence, the IUPAC name of ‘A’ is But-2- ene. Ozonolysis of ‘A’ takes place as:
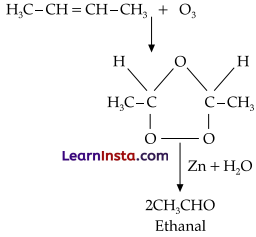
The final product is ethanal with molecular mass = 44u.
Section-D
The following questions are case based questions. Each question has an internal choice and carries 4 ( 1 + 1 + 2 ) marks each.
Question 29.
Read the passage given below and answer the following questions:
James Maxwell (1870) suggested that when electrically charged particle moves under acceleration, alternating electrical and magnetic fields are produced and transmitted. These fields are transmitted in the form of waves called electromagnetic waves or electromagnetic radiation. These waves do not require any medium for their propagation and can move in vacuum.
The increasing order of wavelengths of these radiations is given as below : Cosmic rays < λ – rays < X-rays < Ultraviolet light < Visible light < Infrared rays < Micro waves < Radio waves.
(A) What is the relation between wavelength, wave number, frequency and velocity?
OR
Chlorophyll absorbs radiations of wavelength 700 nm. What is the frequency of this radiation?
(B) The radio 93.5 FM, broadcasts on a frequency of 1,370 kHz (kilo hertz). What will be the wavelength of the
electromagnetic radiation emitted by transmitter. Also, predict which part of the electromagnetic spectrum does it belong to?
(C) When are Paschen and Brackett series displayed? Mention in which portion of electromagnetic spectrum they fall?
Answer:
(A) c = λ × v and \(\frac{1}{\lambda}\) = \(\bar{v}\)
∴ v = c.\(\bar{v}\)
Explanation:
A wave has its wavelength denoted by lambda (l). The number of waves that pass a given point in one second is the frequency denoted by neu (u). The number of wavelengths per unit length is the wave number neu bar (v). The relation can be expressed as, when ‘d is the speed of electromagnetic radiation in vacuum.
OR
Wavelength (λ) = 700 nm = 700 × 10-9 m = 7 × 10-7 m
Velocity of radiation (c) = 3 × 108 m/s velocity
∴ Frequency (v) = \(\frac{\text { velocity }(\mathrm{c})}{\text { wavelength }(\lambda)}\)
= \(\frac{3 \times 10^8}{7 \times 10^{-7}}\)
= 0.42 × 1015 = 4.2 X 1014 Hz
(B) The wavelength, λ = C electromagnetic radiation /v frequency
On substituting the given values, we get λ = c/ v
(3 × 108 m/s) / 1370 × 103/s = 218.9 m
This value wavelength is a characteristic radio wave wavelength.
(C) When an electron transitions from a higher energy level (nh = 4, 5, 6, 7, 8, …) to an energy state with nl = 3, the Paschen series is seen. The Paschen series’ entire wavelength ranges inside the infrared portion of the electromagnetic spectrum. The Brackett series, which has the shortest wavelength, overlaps with the Paschen series.
When an electron transitions from a higher energy level (nh = 5, 6, 7, 8, 9, …) to an energy state with nl = 4, the Brackett series is seen. The electromagnetic spectrum’s infrared region encompasses the entire wavelength range of the Brackett series.
![]()
Question 30.
Read the passage given below and answer the following questions:
According to the major functional group found in the structure, organic molecules are classified into distinct kinds of compounds under the IUPAC naming system. The compound’s identity is determined by the major functional group, which is determined via a system of priorities. Each and every other functional category is viewed as a substitute. The precedence list that follows applies to functional groups with carbon as the main atom. As a general rule, the functional group has a higher priority, the higher the oxidation state of the central carbon. As a result, carboxylic acids are given precedence over alcohols, and so forth.
(A) Which groups are considered as lowest priority groups? Give their descending order of priority.
(B) Mention the precedence of principal functional group series.
(C) What are alkynes? How are they named?
OR
Give the IUPAC names of the following compounds:

Answer:
(A) The groups like alkyl, alkoxy, and halides are considered as lowest priority groups or substituent groups. The descending order of priority is: alkyl > alkoxy > halides.
(B) The precedence of Principal functional group series is as follows:
-COOH > -SO3H > -COOR (R = alkyl group) > -COCl > -CONH2 > -CN > -HC = O > > C = O > -OH > -NH2 > >C = C< > -C = C-
Substituent groups: -R, C6H5-, halogens (F, Cl, Br, I), -NO2, alkoxy (-OR) etc., are always used as prefix substituents.
(C) Alkynes are defined as compounds with double and triple bonds. (Note that the name has a yne at the end). Unless both groups are equally far from the chain ends, in which case the double bond takes precedence and is assigned the lower number, chain numbering begins from the end nearest to either group.
OR
- 2-Bromo-4-methylpentan-3-one
- 4-Methyl-2-nitropentan-3-one
Section-E
The following questions are long answer type and carry 5 marks each. All questions have an internal choice.
Question 31.
Attempt any five questions:
(A) State the law of multiple proportions.
(B) Differentiate between solids, liquids and gases in terms of volume and shapes.
(C) Define the terms:
(i) Empirical formula,
(ii) Molecular formula
(D) A compound made up of two elements A and B has A = 70%, B = 30%. Their relative number of moles in the compound is 1.25 and 1.88, calculate: Atomic masses of the elements A and B.
(E) What is ,the SI unit of mass? How is it defined?
(F) One mole of any substance contains 6.022 × 1023 atoms/molecules. Calculate the number of molecules of H2SO4 present in 100 mL of 0.02 M H2SO4 solution.
Answer:
(A) When two elements combine with each other to form more than one compound, the masses of one element that combine with a fixed mass of the other are in a ratio of small whole numbers. For example: Hydrogen combines with oxygen to form two compounds, namely, water and hydrogen peroxide
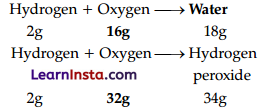
Here, the masses of oxygen (i.e., 16 g and 32 g), which combine with a fixed mass of hydrogen (2 g) bear a simple ratio, i.e., 16 : 32 or 1 : 2.
(B)
| Matter | Volume | Shape |
| Solid | Definite | Definite |
| Liquid | Definite | Indefinite |
| Gas | Indefinite | Indefinite |
(C) (i) E mpirical formula represents the simple whole number ratio of atoms of all elements present in a molecule of the compound.
(ii) Molecular formula gives the actual number of atoms of various elements present in one molecule of compound.
(D) Atomic mass of element A
= \(\frac{\% \text { of element } \mathrm{A}}{\text { Relative number of moles }}\)
= \(\frac{70}{1.25}\)
= 56
Atomic mass of element B
= \(\frac{\% \text { of element B }}{\text { Relative number of moles }}\)
= \(\frac{30}{1.88}\)
= 15.957 ≈ 16.
(E) The S.I. unit of mass is kilogram. The One kilogram is defined as the mass equal to the mass of the international prototype of kilogram.
(F) Number of moles of H2SO4
= molarity × volume in mL
= 0.02 × 100 = 2 milli moles
= 2 × 10-3 mol
Number of molecules of H2SO4
= Number of moles × NA
= 2 × 10-3 × 6.022 × 1023
= 12.044 × 1020 molecules
![]()
Question 32.
(A) Write a short note on differential extraction.
(B) Explain hyperconjugation.
(C) How does hyperconjugation explain the stability of alkenes?
OR
(A) Explain the terms Inductive and Electromeric effects.
(B) Which electron displacement explains the following correct orders of acidity of the carboxylic acids?
(i) Cl3CCOOH > Cl2CHCOOH > ClCH2COOH
(ii) CH3CH2COOH > (CH3)2CHCOOH > (CH3)3CCOOH
Answer:
(A) When an organic compound is present in an aqueous medium, it is separated by shaking it with organic solvent in which it is more soluble than in water. The aqueous solution is mixed with organic solvent in a separating funnel and shaken for sometimes and then allowed to stand for some time. When organic solvent and water form two separate layers, the lower layer is run out by opening the tap of funnel and organic layer is separated. The process is repeated several times and pure organic compound is separated.
(B) Hyperconjugation is a permanent general stabilising interaction which involves delocalisation of s electrons of C—H bond of an alkyl group directly attached to an atom of an unsaturated system or to an atom with an unshared p orbital. The S electrons of C—H bond of the alkyl group enter into partial conjugation with the attached unsaturated system or with the unshared p orbital.
(C) The greater the hyperconjugation, the greater will be the stability of the compound. The increasing order of stability can be shown as :
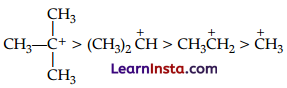
(A) Inductive effect :
The permanent displacement of sigma (a) electrons along a saturated chain, whenever an electron withdrawing or electron donating group is present is called inductive effect.
Inductive effect could be +I effect or -I effect. When an atom or group attracts electrons
towards itself more strongly than hydrogen, it is said to possess -I effect. When an atom or group attracts electrons towards itself less strongly than hydrogen, it is said to possess +I effect.
Electromeric effect:
It involves the complete transfer of the shared pair of electrons to either of the two atoms linked by multiple bonds in the presence of an attacking agent. For example, electromeric effect could be +E effect or -E effect. It is a temporary effect, exist only in the presence of the reagent. It results in complete charge.
+ E effect: When the electrons are transferred towards the attacking reagent.
-E effect: When the electrons are transferred away from the attacking reagent.
(B) (i) Cl3CCOOH > Cl2CHCOOH > ClCH2COOH The order of acidity can be explained on the basis of inductive effect (-I effect). As the number of chlorine atoms increases, the -I effect increases. With the increase in -I effect, the acid strength also increases accordingly.
(ii) CH3CH2COOH > (CH3)2CHCOOH > (CH3)3CCOOH The order of acidity can be explained on the basis of inductive effect (+I effect). As the number of alkyl groups increases, the +I effect also increases. With the increase in +I effect, the acid strength also increases accordingly.
Question 33.
Write down the preparation of alkanes from carboxylic acid by the following methods-
(A) Decarboxylation
(B) Kolbe’s Electrolysis
OR
(A) (i) Sodium salt of which acid will be needed for the preparation of propane? Write chemical equation for the
reaction?
(ii) What happen in incomplete combustion of hydrocarbon?
(B)(i) What is isomerization?
(ii) What is controlled oxidation?
(iii) What is pyrolysis?
Answer:
(A) Decarboxylation:
Sodium salts of carboxylic acids on heating with soda lime (mixture of sodium hydroxide and calcium oxide) give alkanes containing one carbon atom less than the carboxylic acid. This process of elimination of carbon dioxide from a carboxylic acid is known as decarboxylation.
![]()
(B) An aqueous solution of sodium or potassium salt of a carboxylic acid on electrolysis gives alkane containing even number of carbon atoms at the anode.
2CH3COO–Na+ + 2H2O > CH3 – CH3 + 2CO2 + H2 + 2NaOH
The reaction is supposed to follow the following path :
At Anode:
![]()
At Cathode:
H2O + e– → OH– + H+
2H+ → H2
Methane cannot be prepared by this method
OR
(A)![]()
(ii) During incomplete combustion of alkanes with insufficient amount of air or dioxygen, carbon black is formed which is used in the manufacture of ink, printer ink, black pigments and as filters.
(B) (i) n-Alkanes on heating in the presence of anhydrous aluminium chloride and hydrogen chloride gas
isomerise to branched chain alkanes
(ii) Alkanes on heating with a regulated supply of dioxygen or air at high pressure and in the presence of suitable catalysts give a variety of oxidation products
(iii) Higher alkanes on heating to higher temperature decompose into lower alkanes, alkenes etc. Such a decomposition reaction into smaller fragments by the application of heat is called pyrolysis or cracking.